

Table of SynGAP1 Isoform α2 (UniProt Q96PV0-1) Missense Variants.
| c.dna | Variant | SGM Consensus | Domain and Structure information: based on WT protein | Annotated databases | Deep learning-based pathogenicity predictions | Folding stability-based pathogenicity predictions | Sequence/structure-based pathogenicity predictions | Phase Separation | Evolutionary/physical properties | Molecular Dynamics-based analysis | DOI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Domain | IUPred2 | ANCHOR2 | AlphaFold | MobiDB | PhosphoSitePlus | ClinVar | gnomAD | ESM1b | AlphaMissense | FoldX | Rosetta | Foldetta | PremPS | REVEL | PROVEAN | PolyPhen-2 HumDiv | PolyPhen-2 HumVar | FATHMM | SIFT | PSMutPred | PAM | Physical | SASA | Normalized B-factor backbone | Normalized B-factor sidechain | SynGAP Structural Annotation | |||||||||||||||||||||||||||||||||||||||||||||
| Score | Prediction | Score | Prediction | pLDDT | disorder | disorder | LTP | HTP | KL | PTM | Clinical Status | Review | Subm. | ID | Allele count | Allele freq. | LLR score | Prediction | Pathogenicity | Class | Optimized | Average ΔΔG | Prediction | StdDev | ΔΔG | Prediction | ΔΔG | Prediction | ΔΔG | Prediction | Score | Prediction | Score | Prediction | pph2_prob | Prediction | pph2_prob | Prediction | Nervous System Score | Prediction | Prediction | Status | Conservation | Sequences | IP RF | SP RF | Prediction | PAM250 | PAM120 | Hydropathy Δ | MW Δ | Average | Δ | Δ | StdDev | Δ | StdDev | Secondary | Tertiary bonds | Inside out | GAP-Ras interface | At membrane | No effect | MD Alert | Verdict | Description | |||||
| c.819G>T | E273D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant E273D is listed in ClinVar (ID 1471608) with an “Uncertain” status and is present in gnomAD (variant ID 6‑33437724‑G‑T). Prediction tools that report a benign effect include REVEL, FoldX, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score. Only FATHMM predicts a pathogenic outcome, while premPS is inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also predicts benign. Overall, the preponderance of evidence indicates the variant is most likely benign, and this conclusion does not contradict the current ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.071867 | Structured | 0.398918 | Uncertain | 0.863 | 0.196 | 0.125 | Conflicting | 2 | 6-33437724-G-T | 2 | 1.24e-6 | -1.811 | Likely Benign | 0.058 | Likely Benign | Likely Benign | 0.26 | Likely Benign | 0.1 | -0.48 | Likely Benign | -0.11 | Likely Benign | -0.63 | Ambiguous | 0.092 | Likely Benign | 1.99 | Neutral | 0.004 | Benign | 0.010 | Benign | 2.00 | Pathogenic | 1.00 | Tolerated | 3.38 | 18 | 0.1711 | 0.1859 | 3 | 2 | 0.0 | -14.03 | 223.1 | 22.1 | 0.2 | 0.0 | 0.0 | 0.1 | X | Potentially Benign | The negatively charged residue Glu273, located in a β hairpin loop (res. Glu273-Lys278) that connects two anti-parallel β sheet strands, is replaced with another negatively charged residue, aspartate. Because the C2 domain loop faces the membrane surface, the potentially crucial role of the carboxylate group of Glu273 or Asp273 on SynGAP-membrane association cannot be fully explored via solvent-only simulations.As a minor note, the neighboring residue Arg272, which stacks with the indole ring of the Trp362 side chain and directly faces RasGTPase, forms a salt bridge more often with Asp273 than with the non-mutated Glu273 in the simulations. Regardless, due to the similar physicochemical properties of the WT and variant residues at the membrane interface, the residue swap is likely to be well tolerated. | |||||||||||||
| c.964G>T | A322S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A322S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. Only FATHMM predicts a pathogenic outcome. Grouping by consensus, the benign‑predicting tools outnumber the single pathogenic prediction. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) reports “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also indicates a benign effect. No prediction or stability result is missing or inconclusive. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.175930 | Structured | 0.425745 | Uncertain | 0.938 | 0.334 | 0.000 | -1.424 | Likely Benign | 0.058 | Likely Benign | Likely Benign | 0.41 | Likely Benign | 0.2 | 0.38 | Likely Benign | 0.40 | Likely Benign | -0.44 | Likely Benign | 0.127 | Likely Benign | 1.09 | Neutral | 0.010 | Benign | 0.010 | Benign | 2.00 | Pathogenic | 0.67 | Tolerated | 0.2696 | 0.5253 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.107A>T | H36L 2D  AIThe SynGAP1 missense variant H36L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus, SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, indicates a likely benign classification. AlphaMissense‑Optimized also reports benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, was not available for this variant. Overall, the majority of evidence points to a benign effect. Thus, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar classification exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -2.403 | Likely Benign | 0.129 | Likely Benign | Likely Benign | 0.095 | Likely Benign | -1.73 | Neutral | 0.010 | Benign | 0.011 | Benign | 4.19 | Benign | 0.00 | Affected | 0.1310 | 0.6111 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.1120T>G | S374A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S374A is reported in gnomAD (6‑33438025‑T‑G) and has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments—AlphaMissense‑Optimized, SGM‑Consensus, and Foldetta (combining FoldX‑MD and Rosetta outputs)—all indicate a benign effect. Based on the collective predictions, the variant is most likely benign, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.642678 | Disordered | 0.428948 | Uncertain | 0.333 | 0.812 | 0.625 | 6-33438025-T-G | -4.245 | Likely Benign | 0.082 | Likely Benign | Likely Benign | -0.08 | Likely Benign | 0.1 | 0.41 | Likely Benign | 0.17 | Likely Benign | 0.10 | Likely Benign | 0.122 | Likely Benign | -0.53 | Neutral | 0.012 | Benign | 0.011 | Benign | 5.32 | Benign | 0.04 | Affected | 4.32 | 13 | 0.5091 | 0.5774 | Strenghten | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||
| c.1210G>A | A404T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A404T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: SGM‑Consensus (Likely Benign), REVEL, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only FoldX and premPS are inconclusive, so they are treated as unavailable. High‑accuracy methods corroborate this: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign; and Foldetta (combining FoldX‑MD and Rosetta) is benign. Overall, the evidence strongly supports a benign classification, with no conflict with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.232838 | Structured | 0.415505 | Uncertain | 0.965 | 0.355 | 0.000 | -6.674 | Likely Benign | 0.279 | Likely Benign | Likely Benign | 0.74 | Ambiguous | 0.1 | -0.01 | Likely Benign | 0.37 | Likely Benign | 0.56 | Ambiguous | 0.060 | Likely Benign | -1.74 | Neutral | 0.049 | Benign | 0.011 | Benign | 4.12 | Benign | 0.09 | Tolerated | 0.1710 | 0.7367 | 1 | 0 | -2.5 | 30.03 | |||||||||||||||||||||||||||||
| c.1312G>A | A438T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A438T is listed in ClinVar with an “Uncertain” status and is present in the gnomAD database (gnomAD ID 6‑33438217‑G‑A). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is benign. Based on the collective predictions, the variant is most likely benign, and this conclusion does not contradict the current ClinVar status of “Uncertain.” Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.147574 | Structured | 0.290154 | Uncertain | 0.929 | 0.293 | 0.000 | Conflicting | 3 | 6-33438217-G-A | 16 | 9.91e-6 | -5.339 | Likely Benign | 0.085 | Likely Benign | Likely Benign | 0.21 | Likely Benign | 0.0 | -0.07 | Likely Benign | 0.07 | Likely Benign | 0.36 | Likely Benign | 0.021 | Likely Benign | -0.81 | Neutral | 0.300 | Benign | 0.011 | Benign | 4.18 | Benign | 0.14 | Tolerated | 3.38 | 26 | 0.0999 | 0.5574 | 1 | 0 | -2.5 | 30.03 | 214.2 | -42.7 | -0.3 | 0.1 | -0.4 | 0.1 | X | Potentially Benign | The methyl group of Ala438, located in a four-residue loop connecting two α helices (res. Asn440-Thr458 and Pro413-Glu436), packs against hydrophobic residues from a nearby α helix or loop residues (e.g., Leu703, Val699). In the variant simulations, the methyl group of Thr438 is able to establish similar hydrophobic packing. Moreover, the hydroxyl group also H-bonds with nearby residues, such as the carbonyl group of the neighboring loop residue Pro437. Accordingly, the residue swap does not generate an apparent negative effect on the protein structure based on the simulations. | |||||||||||||
| c.1328G>C | G443A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G443A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that reach a consensus all indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the substitution as tolerated. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized classifies the variant as benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports it as Likely Benign. The protein‑folding stability predictor Foldetta, which integrates FoldX‑MD and Rosetta outputs, yields an uncertain result and is therefore treated as unavailable evidence. Overall, the collective predictions strongly suggest that the variant is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.250310 | Structured | 0.258623 | Uncertain | 0.935 | 0.206 | 0.000 | -0.332 | Likely Benign | 0.101 | Likely Benign | Likely Benign | -0.59 | Ambiguous | 0.1 | -1.61 | Ambiguous | -1.10 | Ambiguous | -0.52 | Ambiguous | 0.033 | Likely Benign | -1.04 | Neutral | 0.022 | Benign | 0.011 | Benign | 3.41 | Benign | 0.54 | Tolerated | 0.3539 | 0.3192 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||
| c.1888A>G | I630V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I630V is listed in ClinVar as Benign and is present in gnomAD (variant ID 6‑33440940‑A‑G). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome; all other tools (FoldX, Rosetta, Foldetta, premPS, ESM1b) return uncertain or inconclusive results. The high‑accuracy consensus (SGM Consensus) derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN yields a benign majority (2 benign vs. 1 pathogenic, 1 uncertain). AlphaMissense‑Optimized also predicts benign. Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, is uncertain. Taken together, the overwhelming majority of predictions support a benign effect, and this conclusion aligns with the ClinVar designation. Thus, the variant is most likely benign, with no contradiction to the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.040537 | Structured | 0.036106 | Uncertain | 0.966 | 0.236 | 0.000 | Benign/Likely benign | 4 | 6-33440940-A-G | 59 | 3.66e-5 | -7.264 | In-Between | 0.145 | Likely Benign | Likely Benign | 1.33 | Ambiguous | 0.0 | 0.94 | Ambiguous | 1.14 | Ambiguous | 0.64 | Ambiguous | 0.143 | Likely Benign | -0.38 | Neutral | 0.018 | Benign | 0.011 | Benign | -1.37 | Pathogenic | 0.35 | Tolerated | 3.37 | 34 | 0.0960 | 0.2891 | 4 | 3 | -0.3 | -14.03 | 235.0 | 26.2 | -0.1 | 0.0 | -0.3 | 0.1 | X | Potentially Benign | The sec-butyl side chain of Ile630, located in an α helix (res. Glu617-Asn635), packs with hydrophobic residues (e.g., Phe594, Leu633, Ile626, Ile602) in the hydrophobic inter-helix space between two α helices (res. Glu617-Asn635 and res. Glu582-Met603).In the variant simulations, the iso-propyl side chain of Val630, which shares a similar size and physicochemical properties with Ile630 in the WT, maintains similar interactions in the inter-helix space. Although no negative structural effects are observed during the simulations, the implications of the residue swap on the complex formation with the GTPase, due to its location, cannot be investigated using solvent-only simulations. | ||||||||||||||
| c.1924A>G | K642E 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant K642E is not reported in ClinVar and has no entries in gnomAD. Functional prediction tools cluster into two groups: benign predictions (REVEL, FoldX, Rosetta, Foldetta, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM) and pathogenic predictions (SGM‑Consensus, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized). High‑accuracy methods give a mixed signal: AlphaMissense‑Optimized predicts pathogenic, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also indicates likely pathogenic, whereas Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign stability. No prediction is inconclusive. Overall, the majority of tools lean toward pathogenicity, and this is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.061840 | Structured | 0.181468 | Uncertain | 0.806 | 0.289 | 0.000 | -11.287 | Likely Pathogenic | 0.968 | Likely Pathogenic | Likely Pathogenic | 0.26 | Likely Benign | 0.1 | -0.13 | Likely Benign | 0.07 | Likely Benign | 0.40 | Likely Benign | 0.404 | Likely Benign | -3.92 | Deleterious | 0.116 | Benign | 0.011 | Benign | 2.96 | Benign | 0.03 | Affected | 0.3865 | 0.1013 | 0 | 1 | 0.4 | 0.94 | |||||||||||||||||||||||||||||
| c.1975T>A | S659T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S659T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: SIFT, PolyPhen‑2 (HumDiv and HumVar), REVEL, PROVEAN, premPS, FoldX, AlphaMissense‑Default, AlphaMissense‑Optimized, ESM1b, FATHMM, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all classify the change as benign. Only Rosetta reports an uncertain outcome, which is treated as unavailable evidence. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized is benign, the SGM Consensus is “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. No tool predicts pathogenicity. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.067594 | Structured | 0.154597 | Uncertain | 0.954 | 0.283 | 0.000 | -5.693 | Likely Benign | 0.157 | Likely Benign | Likely Benign | -0.15 | Likely Benign | 0.0 | -0.74 | Ambiguous | -0.45 | Likely Benign | 0.42 | Likely Benign | 0.106 | Likely Benign | -2.04 | Neutral | 0.069 | Benign | 0.011 | Benign | 3.41 | Benign | 0.39 | Tolerated | 0.1505 | 0.5976 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.2101C>G | P701A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant P701A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; all of these classify the substitution as benign. No tool predicts a pathogenic outcome. Predictions that are inconclusive are FoldX (uncertain) and Foldetta (uncertain). High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also predicts benign, while Foldetta remains uncertain. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.074921 | Structured | 0.404318 | Uncertain | 0.918 | 0.345 | 0.000 | -6.836 | Likely Benign | 0.217 | Likely Benign | Likely Benign | 1.35 | Ambiguous | 0.0 | 0.23 | Likely Benign | 0.79 | Ambiguous | -0.16 | Likely Benign | 0.058 | Likely Benign | -0.55 | Neutral | 0.002 | Benign | 0.011 | Benign | 3.50 | Benign | 0.82 | Tolerated | 0.3143 | 0.3765 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||
| c.212A>C | D71A 2D  AIThe SynGAP1 D71A missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that indicate a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign (three benign votes versus one pathogenic). High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this assessment does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.575842 | Disordered | 0.456046 | Uncertain | 0.350 | 0.799 | 0.375 | -3.422 | Likely Benign | 0.619 | Likely Pathogenic | Likely Benign | 0.077 | Likely Benign | -1.89 | Neutral | 0.092 | Benign | 0.011 | Benign | 4.06 | Benign | 0.00 | Affected | 0.3876 | 0.6040 | 0 | -2 | 5.3 | -44.01 | |||||||||||||||||||||||||||||||||||||||
| c.236A>G | N79S 2D  AIThe SynGAP1 missense variant N79S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.618285 | Disordered | 0.457064 | Uncertain | 0.290 | 0.876 | 0.375 | -2.311 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.017 | Likely Benign | -0.31 | Neutral | 0.113 | Benign | 0.011 | Benign | 4.29 | Benign | 0.00 | Affected | 0.3155 | 0.4740 | 1 | 1 | 2.7 | -27.03 | |||||||||||||||||||||||||||||||||||||||
| c.2383C>G | P795A 2D  AIThe SynGAP1 missense variant P795A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; no tool predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly suggests that P795A is most likely benign, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.972450 | Disordered | 0.410339 | Uncertain | 0.457 | 0.903 | 0.875 | -5.348 | Likely Benign | 0.049 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -0.52 | Neutral | 0.016 | Benign | 0.011 | Benign | 4.32 | Benign | 0.16 | Tolerated | 0.3672 | 0.5371 | 1 | -1 | 3.4 | -26.04 | ||||||||||||||||||||||||||||||||||||||
| c.253A>T | T85S 2D  AIThe SynGAP1 missense variant T85S is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Pathogenic, while the SGM‑Consensus remains Likely Benign; Foldetta results are unavailable. Overall, the majority of conventional predictors lean toward a benign impact, but the high‑accuracy AlphaMissense‑Optimized tool suggests pathogenicity, creating a conflict. No ClinVar entry exists, so there is no reported status to contradict. Based on the collective evidence, the variant is most likely benign, though the high‑accuracy prediction indicates uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.680603 | Disordered | 0.542004 | Binding | 0.288 | 0.888 | 0.500 | -4.171 | Likely Benign | 0.973 | Likely Pathogenic | Likely Pathogenic | 0.103 | Likely Benign | -1.37 | Neutral | 0.113 | Benign | 0.011 | Benign | 3.87 | Benign | 0.00 | Affected | 0.2590 | 0.3569 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2806G>C | A936P 2D  AIThe SynGAP1 missense variant A936P is not reported in ClinVar and is absent from gnomAD. Consensus from multiple in‑silico predictors indicates a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score all classify the change as benign or likely benign. Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is also benign. No Foldetta stability prediction is available. Overall, the computational evidence overwhelmingly points to a benign effect, and this is consistent with the absence of a ClinVar pathogenic classification. Thus, the variant is most likely benign, and this does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.812494 | Disordered | 0.973218 | Binding | 0.319 | 0.874 | 0.625 | -2.782 | Likely Benign | 0.191 | Likely Benign | Likely Benign | 0.014 | Likely Benign | -1.28 | Neutral | 0.012 | Benign | 0.011 | Benign | 2.46 | Pathogenic | 0.06 | Tolerated | 0.2043 | 0.6109 | 1 | -1 | -3.4 | 26.04 | |||||||||||||||||||||||||||||||||||||||
| c.2854G>C | G952R 2D  AIThe SynGAP1 missense variant G952R is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it as pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence indicates that G952R is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.985964 | Disordered | 0.910621 | Binding | 0.341 | 0.926 | 0.750 | -5.974 | Likely Benign | 0.295 | Likely Benign | Likely Benign | 0.139 | Likely Benign | -0.93 | Neutral | 0.077 | Benign | 0.011 | Benign | 3.20 | Benign | 0.02 | Affected | 0.1145 | 0.4933 | -3 | -2 | -4.1 | 99.14 | |||||||||||||||||||||||||||||||||||||||
| c.2914C>G | P972A 2D  AIThe SynGAP1 missense variant P972A is listed in ClinVar with an uncertain significance (ClinVar ID 3172763.0) and is present in the gnomAD database (gnomAD ID 6‑33443466‑C‑G). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” The Foldetta protein‑folding stability analysis is unavailable for this variant. Overall, the computational evidence strongly suggests the variant is most likely benign, and this conclusion does not contradict the current ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.891961 | Disordered | 0.954150 | Binding | 0.472 | 0.904 | 0.625 | Uncertain | 1 | 6-33443466-C-G | 1 | 6.20e-7 | -0.167 | Likely Benign | 0.045 | Likely Benign | Likely Benign | 0.046 | Likely Benign | -0.89 | Neutral | 0.016 | Benign | 0.011 | Benign | 4.29 | Benign | 0.07 | Tolerated | 4.32 | 2 | 0.3212 | 0.4753 | -1 | 1 | 3.4 | -26.04 | ||||||||||||||||||||||||||||||||
| c.320G>T | R107M 2D  AIThe SynGAP1 missense variant R107M is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, and Foldetta results are unavailable. Overall, the balance of evidence favors a benign interpretation, and this conclusion does not conflict with the ClinVar status, which currently contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.666105 | Disordered | 0.663448 | Binding | 0.331 | 0.863 | 0.875 | -4.873 | Likely Benign | 0.995 | Likely Pathogenic | Likely Pathogenic | 0.233 | Likely Benign | -2.65 | Deleterious | 0.028 | Benign | 0.011 | Benign | 2.96 | Benign | 0.00 | Affected | 0.1605 | 0.4415 | 0 | -1 | 6.4 | -24.99 | ||||||||||||||||||||||||||||||||||||||||
| c.3344T>A | I1115N 2D  AIThe SynGAP1 missense variant I1115N is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.889439 | Disordered | 0.892339 | Binding | 0.308 | 0.912 | 0.750 | -4.018 | Likely Benign | 0.118 | Likely Benign | Likely Benign | 0.095 | Likely Benign | -0.60 | Neutral | 0.009 | Benign | 0.011 | Benign | 2.77 | Benign | 0.08 | Tolerated | 0.1299 | 0.1270 | -2 | -3 | -8.0 | 0.94 | |||||||||||||||||||||||||||||||||||||||
| c.352A>G | M118V 2D  AIThe SynGAP1 missense variant M118V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The prediction is consistent with the lack of ClinVar annotation, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.694846 | Disordered | 0.676867 | Binding | 0.330 | 0.883 | 0.500 | -2.322 | Likely Benign | 0.122 | Likely Benign | Likely Benign | 0.158 | Likely Benign | -1.23 | Neutral | 0.012 | Benign | 0.011 | Benign | 3.98 | Benign | 0.02 | Affected | 0.3326 | 0.3630 | 2 | 1 | 2.3 | -32.06 | |||||||||||||||||||||||||||||||||||||||
| c.3607C>T | H1203Y 2D 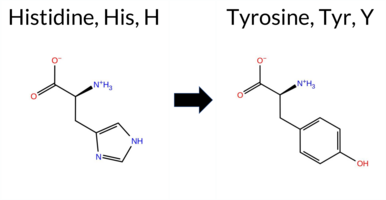 AIThe SynGAP1 missense variant H1203Y is listed in ClinVar with an “Uncertain” status and is present in the gnomAD database (ID 6‑33446599‑C‑T). Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the variant as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also yields a benign prediction. Foldetta results are unavailable. Overall, the evidence strongly supports a benign impact for H1203Y, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.618285 | Disordered | 0.527023 | Binding | 0.892 | 0.589 | 0.250 | Uncertain | 1 | 6-33446599-C-T | 2 | 1.24e-6 | -6.834 | Likely Benign | 0.149 | Likely Benign | Likely Benign | 0.233 | Likely Benign | -1.52 | Neutral | 0.006 | Benign | 0.011 | Benign | 5.55 | Benign | 0.10 | Tolerated | 3.77 | 5 | 0.0588 | 0.2379 | 2 | 0 | 1.9 | 26.03 | |||||||||||||||||||||||||||||||
| c.3791G>A | G1264D 2D  AIThe SynGAP1 missense variant G1264D is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign, while only AlphaMissense‑Default predicts it as pathogenic. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote) is also benign; Foldetta results are not available. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.429200 | Structured | 0.762385 | Binding | 0.897 | 0.579 | 0.000 | -4.725 | Likely Benign | 0.648 | Likely Pathogenic | Likely Benign | 0.097 | Likely Benign | -1.29 | Neutral | 0.012 | Benign | 0.011 | Benign | 2.72 | Benign | 0.23 | Tolerated | 0.1658 | 0.1754 | 1 | -1 | -3.1 | 58.04 | ||||||||||||||||||||||||||||||||||||||
| c.4018A>G | T1340A 2D  AIThe SynGAP1 missense variant T1340A is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the consensus of the majority of tools, including the high‑accuracy methods, supports a benign classification. This prediction is consistent with the lack of ClinVar evidence and does not contradict any existing database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.852992 | Disordered | 0.977899 | Binding | 0.444 | 0.697 | 0.750 | -3.192 | Likely Benign | 0.078 | Likely Benign | Likely Benign | 0.116 | Likely Benign | -1.01 | Neutral | 0.010 | Benign | 0.011 | Benign | 4.20 | Benign | 0.02 | Affected | 0.3984 | 0.4288 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||||||||||||
| c.4022C>G | A1341G 2D  AIThe SynGAP1 missense variant A1341G is not reported in ClinVar and is absent from gnomAD. Consensus predictions from multiple in silico tools overwhelmingly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also reports it as likely benign. Only the SIFT algorithm predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Taken together, the preponderance of evidence points to a benign effect for A1341G, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Thus, the variant is most likely benign, and this assessment does not contradict the ClinVar record, which contains no pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.980111 | Binding | 0.383 | 0.696 | 1.000 | -3.334 | Likely Benign | 0.092 | Likely Benign | Likely Benign | 0.049 | Likely Benign | -0.84 | Neutral | 0.006 | Benign | 0.011 | Benign | 4.12 | Benign | 0.05 | Affected | 0.2026 | 0.4541 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.4022C>T | A1341V 2D  AIThe SynGAP1 missense variant A1341V is catalogued in gnomAD (ID 6‑33451896‑C‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign or tolerated outcomes, while the single pathogenic signal comes from SIFT. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates a benign likelihood. Foldetta results are unavailable, so they do not influence the assessment. Overall, the preponderance of evidence points to a benign impact for A1341V, and this conclusion is not contradicted by any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.980111 | Binding | 0.383 | 0.696 | 1.000 | 6-33451896-C-T | 1 | 6.29e-7 | -3.687 | Likely Benign | 0.131 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -1.90 | Neutral | 0.006 | Benign | 0.011 | Benign | 4.04 | Benign | 0.02 | Affected | 3.77 | 5 | 0.1398 | 0.6557 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||
| c.503A>G | H168R 2D 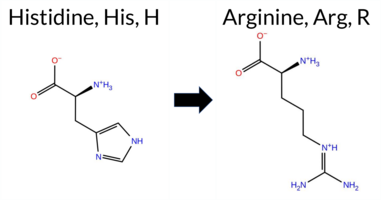 AIThe SynGAP1 missense variant H168R is reported in gnomAD (ID 6‑33432800‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized all classify the change as benign, whereas SIFT predicts it to be pathogenic. Two tools (ESM1b and AlphaMissense‑Default) return uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive because it contains two benign and two uncertain calls, and Foldetta data are not available. Overall, the preponderance of evidence points to a benign impact. This conclusion does not contradict ClinVar, which currently has no classification for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.433034 | Structured | 0.502450 | Binding | 0.402 | 0.678 | 0.125 | 6-33432800-A-G | 1 | 6.20e-7 | -7.334 | In-Between | 0.395 | Ambiguous | Likely Benign | 0.153 | Likely Benign | -1.08 | Neutral | 0.016 | Benign | 0.011 | Benign | 4.26 | Benign | 0.02 | Affected | 4.32 | 3 | 0.1752 | 0.2499 | 0 | 2 | -1.3 | 19.05 | |||||||||||||||||||||||||||||||||||
| c.520A>C | M174L 2D  AIThe SynGAP1 missense variant M174L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Taken together, the majority of evidence points to a benign impact for M174L. This conclusion is consistent with the lack of ClinVar annotation and does not contradict any existing clinical classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.485854 | Uncertain | 0.373 | 0.620 | 0.375 | -5.839 | Likely Benign | 0.608 | Likely Pathogenic | Likely Benign | 0.192 | Likely Benign | -0.82 | Neutral | 0.055 | Benign | 0.011 | Benign | 4.21 | Benign | 0.46 | Tolerated | 0.1335 | 0.4545 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.520A>T | M174L 2D  AIThe SynGAP1 missense variant M174L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Taken together, the majority of evidence points to a benign impact for M174L. This conclusion is consistent with the lack of ClinVar annotation and does not contradict any existing clinical classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.485854 | Uncertain | 0.373 | 0.620 | 0.375 | -5.839 | Likely Benign | 0.608 | Likely Pathogenic | Likely Benign | 0.192 | Likely Benign | -0.82 | Neutral | 0.055 | Benign | 0.011 | Benign | 4.21 | Benign | 0.46 | Tolerated | 0.1335 | 0.4545 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.70G>C | V24L 2D  AIThe SynGAP1 missense variant V24L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect for V24L, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.438970 | Uncertain | 0.382 | 0.890 | 0.500 | -2.590 | Likely Benign | 0.320 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -0.62 | Neutral | 0.043 | Benign | 0.011 | Benign | 3.94 | Benign | 0.00 | Affected | 0.1102 | 0.5226 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.70G>T | V24L 2D  AIThe SynGAP1 missense variant V24L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect for V24L, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.438970 | Uncertain | 0.382 | 0.890 | 0.500 | -2.590 | Likely Benign | 0.320 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -0.62 | Neutral | 0.043 | Benign | 0.011 | Benign | 3.94 | Benign | 0.00 | Affected | 0.1102 | 0.5226 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.1494G>A | M498I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M498I has no ClinVar record (ClinVar ID None) and is not reported in gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are FoldX, SIFT, FATHMM, and AlphaMissense‑Default. Two tools, Rosetta and premPS, give uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as pathogenic, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split. Overall, the majority of predictions lean toward a benign impact, and this is not contradicted by ClinVar status, which lacks an entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.092881 | Structured | 0.399612 | Uncertain | 0.932 | 0.158 | 0.000 | -4.796 | Likely Benign | 0.760 | Likely Pathogenic | Likely Benign | 2.56 | Destabilizing | 0.3 | 1.65 | Ambiguous | 2.11 | Destabilizing | 0.88 | Ambiguous | 0.418 | Likely Benign | -1.09 | Neutral | 0.018 | Benign | 0.012 | Benign | -1.28 | Pathogenic | 0.04 | Affected | 0.1184 | 0.2537 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.1494G>C | M498I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M498I has no ClinVar record (ClinVar ID None) and is not reported in gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are FoldX, SIFT, FATHMM, and AlphaMissense‑Default. Two tools, Rosetta and premPS, give uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as pathogenic, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split. Overall, the majority of predictions lean toward a benign impact, and this is not contradicted by ClinVar status, which lacks an entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.092881 | Structured | 0.399612 | Uncertain | 0.932 | 0.158 | 0.000 | -4.796 | Likely Benign | 0.760 | Likely Pathogenic | Likely Benign | 2.56 | Destabilizing | 0.3 | 1.65 | Ambiguous | 2.11 | Destabilizing | 0.88 | Ambiguous | 0.418 | Likely Benign | -1.09 | Neutral | 0.018 | Benign | 0.012 | Benign | -1.28 | Pathogenic | 0.04 | Affected | 0.1184 | 0.2537 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.1494G>T | M498I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M498I has no ClinVar record (ClinVar ID None) and is not reported in gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are FoldX, SIFT, FATHMM, and AlphaMissense‑Default. Two tools, Rosetta and premPS, give uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as pathogenic, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split. Overall, the majority of predictions lean toward a benign impact, and this is not contradicted by ClinVar status, which lacks an entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.092881 | Structured | 0.399612 | Uncertain | 0.932 | 0.158 | 0.000 | -4.796 | Likely Benign | 0.760 | Likely Pathogenic | Likely Benign | 2.56 | Destabilizing | 0.3 | 1.65 | Ambiguous | 2.11 | Destabilizing | 0.88 | Ambiguous | 0.414 | Likely Benign | -1.09 | Neutral | 0.018 | Benign | 0.012 | Benign | -1.28 | Pathogenic | 0.04 | Affected | 0.1184 | 0.2537 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.1536A>C | E512D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant E512D is not reported in ClinVar or gnomAD. Across a broad panel of in silico predictors, the majority indicate a benign effect: REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score. Only the ESM1b model assigns a pathogenic label, while Rosetta and Foldetta provide uncertain results. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) resolves to benign, and Foldetta remains inconclusive. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.092881 | Structured | 0.247079 | Uncertain | 0.923 | 0.273 | 0.000 | -8.354 | Likely Pathogenic | 0.198 | Likely Benign | Likely Benign | 0.12 | Likely Benign | 0.3 | 1.05 | Ambiguous | 0.59 | Ambiguous | 0.00 | Likely Benign | 0.259 | Likely Benign | -2.10 | Neutral | 0.016 | Benign | 0.012 | Benign | 3.34 | Benign | 0.23 | Tolerated | 0.2055 | 0.3089 | 3 | 2 | 0.0 | -14.03 | |||||||||||||||||||||||||||||
| c.1536A>T | E512D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense change E512D is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Across the broad panel of in‑silico predictors, the majority (REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus) all classify the variant as benign, whereas only ESM1b predicts it as pathogenic. The high‑accuracy tools give a consistent benign signal: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also favors benign. Foldetta, which integrates FoldX‑MD and Rosetta outputs, is inconclusive. Overall, the evidence strongly supports a benign effect; this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.092881 | Structured | 0.247079 | Uncertain | 0.923 | 0.273 | 0.000 | -8.354 | Likely Pathogenic | 0.198 | Likely Benign | Likely Benign | 0.12 | Likely Benign | 0.3 | 1.05 | Ambiguous | 0.59 | Ambiguous | 0.00 | Likely Benign | 0.259 | Likely Benign | -2.10 | Neutral | 0.016 | Benign | 0.012 | Benign | 3.34 | Benign | 0.23 | Tolerated | 0.2055 | 0.3089 | 3 | 2 | 0.0 | -14.03 | |||||||||||||||||||||||||||||
| c.218G>C | R73T 2D 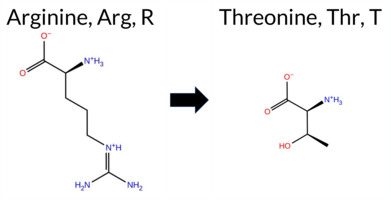 AIThe SynGAP1 missense variant R73T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, and the SGM‑Consensus also indicates Likely Benign. Foldetta results are unavailable and therefore not considered. Overall, the preponderance of evidence from multiple prediction algorithms and consensus methods indicates that R73T is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.501700 | Disordered | 0.453164 | Uncertain | 0.332 | 0.826 | 0.375 | -4.061 | Likely Benign | 0.343 | Ambiguous | Likely Benign | 0.070 | Likely Benign | -1.05 | Neutral | 0.115 | Benign | 0.012 | Benign | 4.05 | Benign | 0.00 | Affected | 0.1980 | 0.4439 | -1 | -1 | 3.8 | -55.08 | |||||||||||||||||||||||||||||||||||||||
| c.2632A>T | T878S 2D  AIThe SynGAP1 missense variant T878S is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.622677 | Disordered | 0.628767 | Binding | 0.288 | 0.878 | 0.250 | -2.493 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.073 | Likely Benign | -0.06 | Neutral | 0.009 | Benign | 0.012 | Benign | 2.82 | Benign | 0.03 | Affected | 0.3557 | 0.4867 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2642T>C | L881S 2D 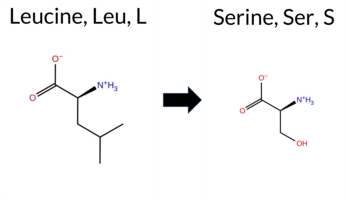 AIThe SynGAP1 missense variant L881S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact for the L881S variant, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.629350 | Binding | 0.299 | 0.874 | 0.250 | -3.063 | Likely Benign | 0.169 | Likely Benign | Likely Benign | 0.051 | Likely Benign | -0.49 | Neutral | 0.068 | Benign | 0.012 | Benign | 2.49 | Pathogenic | 0.00 | Affected | 0.3363 | 0.0850 | -3 | -2 | -4.6 | -26.08 | |||||||||||||||||||||||||||||||||||||||
| c.2819G>C | G940A 2D  AIThe SynGAP1 missense variant G940A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion is consistent with the lack of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.889439 | Disordered | 0.920635 | Binding | 0.383 | 0.902 | 0.625 | -5.356 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.163 | Likely Benign | 0.64 | Neutral | 0.004 | Benign | 0.012 | Benign | 2.85 | Benign | 0.89 | Tolerated | 0.3627 | 0.4831 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.281C>G | P94R 2D  AIThe SynGAP1 missense variant P94R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta results are not available for this variant. Overall, the majority of evidence points to a benign effect, and this assessment does not contradict any ClinVar annotation (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.570978 | Binding | 0.350 | 0.869 | 0.625 | -3.272 | Likely Benign | 0.281 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -2.31 | Neutral | 0.110 | Benign | 0.012 | Benign | 4.11 | Benign | 0.00 | Affected | 0.1445 | 0.2780 | 0 | -2 | -2.9 | 59.07 | |||||||||||||||||||||||||||||||||||||||
| c.2902G>C | G968R 2D  AIThe SynGAP1 missense variant G968R is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the evidence strongly supports a benign classification, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.978316 | Disordered | 0.961360 | Binding | 0.327 | 0.896 | 0.750 | -4.492 | Likely Benign | 0.332 | Likely Benign | Likely Benign | 0.140 | Likely Benign | -0.63 | Neutral | 0.005 | Benign | 0.012 | Benign | 4.19 | Benign | 0.08 | Tolerated | 0.0972 | 0.5017 | -3 | -2 | -4.1 | 99.14 | |||||||||||||||||||||||||||||||||||||||
| c.292C>A | H98N 2D 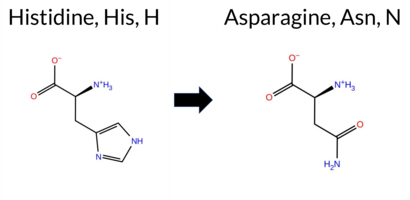 AIThe SynGAP1 missense variant H98N is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.733139 | Disordered | 0.631713 | Binding | 0.348 | 0.872 | 0.625 | -3.855 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.103 | Likely Benign | -0.35 | Neutral | 0.115 | Benign | 0.012 | Benign | 4.24 | Benign | 0.00 | Affected | 0.2003 | 0.3759 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.292C>G | H98D 2D 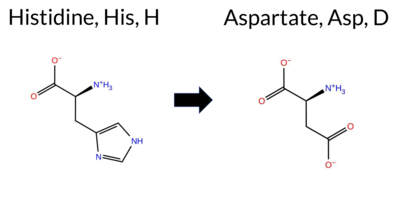 AIThe SynGAP1 missense variant H98D is reported in gnomAD (variant ID 6‑33425900‑C‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) all classify the change as benign. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is “Likely Benign.” No Foldetta stability result is available, so it does not influence the assessment. Overall, the consensus of the available predictions indicates that the variant is most likely benign, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.733139 | Disordered | 0.631713 | Binding | 0.348 | 0.872 | 0.625 | 6-33425900-C-G | 1 | 6.20e-7 | -1.739 | Likely Benign | 0.167 | Likely Benign | Likely Benign | 0.140 | Likely Benign | -0.42 | Neutral | 0.115 | Benign | 0.012 | Benign | 4.24 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2732 | 0.3018 | -1 | 1 | -0.3 | -22.05 | ||||||||||||||||||||||||||||||||||
| c.293A>T | H98L 2D  AIThe SynGAP1 H98L missense variant is reported in gnomAD (ID 6‑33425901‑A‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it as pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments further support this: AlphaMissense‑Optimized predicts benign, SGM‑Consensus is likely benign, and Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign impact for H98L, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.733139 | Disordered | 0.631713 | Binding | 0.348 | 0.872 | 0.625 | 6-33425901-A-T | 1 | 6.32e-7 | -1.804 | Likely Benign | 0.113 | Likely Benign | Likely Benign | 0.194 | Likely Benign | -0.51 | Neutral | 0.115 | Benign | 0.012 | Benign | 4.24 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1125 | 0.6291 | -3 | -2 | 7.0 | -23.98 | ||||||||||||||||||||||||||||||||||
| c.2951A>G | K984R 2D  AIThe SynGAP1 missense variant K984R is reported in gnomAD (ID 6‑33443503‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it to be pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of tools indicates that the variant is most likely benign, and this conclusion is not contradicted by any ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.951648 | Binding | 0.288 | 0.895 | 0.750 | 6-33443503-A-G | 2 | 1.24e-6 | -2.044 | Likely Benign | 0.104 | Likely Benign | Likely Benign | 0.082 | Likely Benign | -0.41 | Neutral | 0.012 | Benign | 0.012 | Benign | 2.67 | Benign | 0.00 | Affected | 4.32 | 1 | 0.5098 | 0.1558 | Weaken | 2 | 3 | -0.6 | 28.01 | |||||||||||||||||||||||||||||||||
| c.3075G>C | Q1025H 2D  AIThe SynGAP1 missense variant Q1025H is not reported in ClinVar and is absent from gnomAD, indicating no known population frequency data. Prediction tools largely agree on a benign effect: SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence supports a benign impact, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.993410 | Binding | 0.363 | 0.746 | 0.500 | -4.976 | Likely Benign | 0.405 | Ambiguous | Likely Benign | 0.051 | Likely Benign | -1.43 | Neutral | 0.014 | Benign | 0.012 | Benign | 2.68 | Benign | 0.05 | Affected | 0.1332 | 0.3852 | 3 | 0 | 0.3 | 9.01 | |||||||||||||||||||||||||||||||||||||||
| c.3075G>T | Q1025H 2D  AIThe SynGAP1 missense variant Q1025H is not reported in ClinVar and is absent from gnomAD, indicating no known population frequency data. Prediction tools largely agree on a benign effect: SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence supports a benign impact, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.993410 | Binding | 0.363 | 0.746 | 0.500 | -4.976 | Likely Benign | 0.405 | Ambiguous | Likely Benign | 0.051 | Likely Benign | -1.43 | Neutral | 0.014 | Benign | 0.012 | Benign | 2.68 | Benign | 0.05 | Affected | 0.1332 | 0.3852 | 3 | 0 | 0.3 | 9.01 | |||||||||||||||||||||||||||||||||||||||
| c.3250C>A | P1084T 2D  AIThe SynGAP1 missense variant P1084T is reported in ClinVar as “Not submitted” and is present in gnomAD (ID 6‑33443802‑C‑A). Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it as pathogenic, but this is the sole discordant call. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign; Foldetta results are not available. Overall, the preponderance of evidence indicates the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which currently contains no pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.842060 | Disordered | 0.979020 | Binding | 0.348 | 0.889 | 1.000 | 6-33443802-C-A | -4.665 | Likely Benign | 0.089 | Likely Benign | Likely Benign | 0.127 | Likely Benign | -2.14 | Neutral | 0.025 | Benign | 0.012 | Benign | 4.11 | Benign | 0.01 | Affected | 3.77 | 5 | 0.1546 | 0.6793 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||||
| c.3511G>T | A1171S 2D  AIThe SynGAP1 missense variant A1171S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta results are not available, so they do not influence the assessment. Overall, the variant is most likely benign, and this conclusion is consistent with the lack of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.599170 | Disordered | 0.702689 | Binding | 0.472 | 0.775 | 0.500 | -2.875 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.156 | Likely Benign | -0.24 | Neutral | 0.009 | Benign | 0.012 | Benign | 5.54 | Benign | 0.08 | Tolerated | 0.2536 | 0.4676 | 1 | 1 | -2.6 | 16.00 | ||||||||||||||||||||||||||||||||||||||
| c.358G>A | G120S 2D  AIThe SynGAP1 missense variant G120S is reported in gnomAD (variant ID 6-33432223‑G‑A) but has no ClinVar entry. Functional prediction tools uniformly classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability predictor, was not available for this variant. Overall, the evidence strongly supports a benign classification, and there is no conflict with ClinVar status because no ClinVar assertion exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.659993 | Binding | 0.359 | 0.887 | 0.750 | 6-33432223-G-A | 1 | 6.20e-7 | -3.558 | Likely Benign | 0.082 | Likely Benign | Likely Benign | 0.034 | Likely Benign | 0.07 | Neutral | 0.020 | Benign | 0.012 | Benign | 4.29 | Benign | 0.91 | Tolerated | 3.61 | 5 | 0.2841 | 0.4713 | 0 | 1 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||||
| c.380G>T | R127L 2D  AIThe SynGAP1 missense variant R127L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as Likely Benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. There is no ClinVar annotation to contradict this conclusion. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.497853 | Structured | 0.711716 | Binding | 0.333 | 0.870 | 0.625 | -0.626 | Likely Benign | 0.643 | Likely Pathogenic | Likely Benign | 0.127 | Likely Benign | -2.02 | Neutral | 0.080 | Benign | 0.012 | Benign | 3.92 | Benign | 0.01 | Affected | 0.1965 | 0.3834 | -3 | -2 | 8.3 | -43.03 | |||||||||||||||||||||||||||||||||||||||
| c.3901C>G | P1301A 2D  AIThe SynGAP1 missense variant P1301A is catalogued in gnomAD (ID 6‑33451775‑C‑G) but has no ClinVar entry. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the substitution as tolerated or benign. No tool in the dataset predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as likely benign. Foldetta, a protein‑folding stability predictor, was not available for this residue. Overall, the consensus of all available predictions is benign, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.728858 | Disordered | 0.885064 | Binding | 0.447 | 0.841 | 0.875 | 6-33451775-C-G | 1 | 6.20e-7 | -4.290 | Likely Benign | 0.055 | Likely Benign | Likely Benign | 0.088 | Likely Benign | 0.66 | Neutral | 0.003 | Benign | 0.012 | Benign | 3.05 | Benign | 0.88 | Tolerated | 3.77 | 5 | 0.2850 | 0.3087 | -1 | 1 | 3.4 | -26.04 | ||||||||||||||||||||||||||||||||||
| c.3961C>A | P1321T 2D  AIThe SynGAP1 missense variant P1321T is catalogued in gnomAD (ID 6‑33451835‑C‑A) but has no ClinVar entry. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the variant as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and this conclusion is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.933505 | Binding | 0.463 | 0.828 | 0.875 | 6-33451835-C-A | -4.946 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.058 | Likely Benign | 1.07 | Neutral | 0.115 | Benign | 0.012 | Benign | 4.25 | Benign | 1.00 | Tolerated | 3.77 | 5 | 0.2016 | 0.5901 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||||
| c.4028A>T | H1343L 2D  AIThe SynGAP1 missense variant H1343L is reported in gnomAD (ID 6‑33451902‑A‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign status. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the available predictions points to a benign effect, and this is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.910643 | Disordered | 0.983646 | Binding | 0.350 | 0.677 | 0.875 | 6-33451902-A-T | -1.552 | Likely Benign | 0.132 | Likely Benign | Likely Benign | 0.058 | Likely Benign | -1.28 | Neutral | 0.053 | Benign | 0.012 | Benign | 4.07 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1222 | 0.5869 | -3 | -2 | 7.0 | -23.98 | ||||||||||||||||||||||||||||||||||||
| c.558G>C | L186F 2D  AIThe SynGAP1 missense variant L186F is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), and FATHMM. In contrast, tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Pathogenic.” High‑accuracy assessments further support a deleterious impact: AlphaMissense‑Optimized predicts pathogenicity, and the SGM‑Consensus (majority vote) also indicates likely pathogenic. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence points to a pathogenic effect, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.458154 | Structured | 0.428613 | Uncertain | 0.397 | 0.617 | 0.500 | Uncertain | 1 | -11.861 | Likely Pathogenic | 0.993 | Likely Pathogenic | Likely Pathogenic | 0.132 | Likely Benign | -3.03 | Deleterious | 0.009 | Benign | 0.012 | Benign | 3.50 | Benign | 0.00 | Affected | 0.0524 | 0.3177 | 2 | 0 | -1.0 | 34.02 | |||||||||||||||||||||||||||||||||||||
| c.558G>T | L186F 2D  AIThe SynGAP1 missense variant L186F has no ClinVar entry (ClinVar status: not reported) and is absent from gnomAD (gnomAD ID: none). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. High‑accuracy assessments further support a deleterious impact: AlphaMissense‑Optimized predicts pathogenic, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Pathogenic.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence indicates that the variant is most likely pathogenic, and this conclusion does not contradict any ClinVar status because no ClinVar classification exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.458154 | Structured | 0.428613 | Uncertain | 0.397 | 0.617 | 0.500 | -11.861 | Likely Pathogenic | 0.993 | Likely Pathogenic | Likely Pathogenic | 0.132 | Likely Benign | -3.03 | Deleterious | 0.009 | Benign | 0.012 | Benign | 3.50 | Benign | 0.00 | Affected | 0.0524 | 0.3177 | 2 | 0 | -1.0 | 34.02 | |||||||||||||||||||||||||||||||||||||||
| c.581A>C | E194A 2D  AIThe SynGAP1 missense variant E194A is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. The high‑accuracy assessment shows AlphaMissense‑Optimized predicts pathogenicity. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default (pathogenic), ESM1b (uncertain), FATHMM (benign), and PROVEAN (pathogenic), also indicates pathogenicity. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of predictions—including the high‑accuracy tools—suggest that E194A is likely pathogenic, and this conclusion does not contradict any ClinVar annotation because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.418646 | Structured | 0.430723 | Uncertain | 0.346 | 0.551 | 0.125 | -7.101 | In-Between | 0.958 | Likely Pathogenic | Likely Pathogenic | 0.235 | Likely Benign | -3.75 | Deleterious | 0.009 | Benign | 0.012 | Benign | 3.99 | Benign | 0.01 | Affected | 0.4331 | 0.5079 | 0 | -1 | 5.3 | -58.04 | ||||||||||||||||||||||||||||||||||||||||
| c.613A>C | I205L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I205L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Functional prediction tools uniformly indicate a benign effect: REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all classify the variant as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, the SGM Consensus predicts likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign. No contradictory evidence exists. Based on the collective predictions, the variant is most likely benign, and this assessment does not conflict with the absence of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.264545 | Structured | 0.409933 | Uncertain | 0.821 | 0.414 | 0.125 | -5.474 | Likely Benign | 0.105 | Likely Benign | Likely Benign | 0.07 | Likely Benign | 0.2 | 0.02 | Likely Benign | 0.05 | Likely Benign | 0.30 | Likely Benign | 0.112 | Likely Benign | -0.86 | Neutral | 0.004 | Benign | 0.012 | Benign | 4.17 | Benign | 0.34 | Tolerated | 0.0682 | 0.2702 | 2 | 2 | -0.7 | 0.00 | |||||||||||||||||||||||||||||
| c.616A>C | I206L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I206L is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and Foldetta. Those that predict a pathogenic effect are ESM1b and AlphaMissense‑Default. Uncertain or inconclusive results come from premPS, AlphaMissense‑Optimized, and Rosetta. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑vs‑2 split, and Foldetta predicts a benign outcome. Overall, the majority of tools (seven benign vs two pathogenic) support a benign classification. Thus, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.298791 | Structured | 0.405123 | Uncertain | 0.863 | 0.391 | 0.125 | -9.488 | Likely Pathogenic | 0.790 | Likely Pathogenic | Ambiguous | 0.05 | Likely Benign | 0.1 | 0.75 | Ambiguous | 0.40 | Likely Benign | 0.78 | Ambiguous | 0.080 | Likely Benign | -1.72 | Neutral | 0.004 | Benign | 0.012 | Benign | 3.78 | Benign | 0.08 | Tolerated | 0.0632 | 0.3608 | 2 | 2 | -0.7 | 0.00 | ||||||||||||||||||||||||||||||
| c.1027G>C | V343L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V343L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote) all predict benign or likely benign. Only FATHMM predicts a pathogenic outcome, while FoldX and Rosetta provide uncertain results and are therefore not considered decisive. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign, the SGM‑Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also indicates benign. Overall, the majority of evidence supports a benign classification, and this is consistent with the absence of ClinVar reporting. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.291804 | Structured | 0.383911 | Uncertain | 0.882 | 0.497 | 0.250 | -6.268 | Likely Benign | 0.310 | Likely Benign | Likely Benign | -0.93 | Ambiguous | 0.2 | 0.66 | Ambiguous | -0.14 | Likely Benign | 0.16 | Likely Benign | 0.033 | Likely Benign | -1.09 | Neutral | 0.005 | Benign | 0.013 | Benign | 2.12 | Pathogenic | 0.64 | Tolerated | 0.1361 | 0.5008 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||
| c.1211C>G | A404G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A404G is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are premPS and SIFT. FoldX, Rosetta, and Foldetta give uncertain or inconclusive results and are treated as unavailable. The high‑accuracy consensus (SGM‑Consensus) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, remains uncertain. Overall, the majority of evidence points to a benign impact for A404G, and this assessment does not contradict any ClinVar status, as none is available. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.232838 | Structured | 0.415505 | Uncertain | 0.965 | 0.355 | 0.000 | -5.277 | Likely Benign | 0.137 | Likely Benign | Likely Benign | 0.92 | Ambiguous | 0.0 | 1.88 | Ambiguous | 1.40 | Ambiguous | 1.18 | Destabilizing | 0.099 | Likely Benign | -2.34 | Neutral | 0.045 | Benign | 0.013 | Benign | 4.03 | Benign | 0.00 | Affected | 0.2370 | 0.5054 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||
| c.14G>C | R5P 2D  AIThe SynGAP1 missense variant R5P is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote) is likely benign; Foldetta results are unavailable. Overall, the preponderance of evidence indicates that R5P is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar classification exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.547847 | Binding | 0.363 | 0.920 | 0.750 | -3.438 | Likely Benign | 0.189 | Likely Benign | Likely Benign | 0.286 | Likely Benign | -0.35 | Neutral | 0.233 | Benign | 0.013 | Benign | 4.10 | Benign | 0.00 | Affected | 0.2294 | 0.5530 | 0 | -2 | 2.9 | -59.07 | |||||||||||||||||||||||||||||||||||||||
| c.1594A>T | T532S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T532S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. Only FATHMM predicts a pathogenic outcome. When high‑accuracy methods are considered separately, AlphaMissense‑Optimized remains benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) also reports a benign effect. No prediction or stability result is missing or inconclusive. Based on the preponderance of evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.275179 | Structured | 0.021478 | Uncertain | 0.889 | 0.385 | 0.000 | 0.616 | Likely Benign | 0.062 | Likely Benign | Likely Benign | 0.15 | Likely Benign | 0.1 | -0.23 | Likely Benign | -0.04 | Likely Benign | -0.10 | Likely Benign | 0.160 | Likely Benign | 0.53 | Neutral | 0.005 | Benign | 0.013 | Benign | -1.22 | Pathogenic | 1.00 | Tolerated | 0.2988 | 0.2866 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||
| c.1663G>T | V555F 2D  3DClick to see structure in 3D Viewer AISynGAP1 V555F is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SGM‑Consensus, PROVEAN, ESM1b, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) as benign. No prediction or stability result is missing or inconclusive. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation because none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.013265 | Structured | 0.008218 | Uncertain | 0.943 | 0.225 | 0.000 | -10.965 | Likely Pathogenic | 0.599 | Likely Pathogenic | Likely Benign | 0.17 | Likely Benign | 0.3 | -2.01 | Stabilizing | -0.92 | Ambiguous | 0.16 | Likely Benign | 0.440 | Likely Benign | -2.85 | Deleterious | 0.011 | Benign | 0.013 | Benign | -1.20 | Pathogenic | 0.16 | Tolerated | 0.0727 | 0.2366 | -1 | -1 | -1.4 | 48.04 | |||||||||||||||||||||||||||||
| c.1672C>T | H558Y 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 H558Y missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while FoldX, premPS, and ESM1b are uncertain. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign result; and Foldetta also predicts benign. No prediction or folding stability result is missing or inconclusive. Overall, the variant is most likely benign, and this conclusion does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.033407 | Structured | 0.011039 | Uncertain | 0.897 | 0.200 | 0.000 | -7.208 | In-Between | 0.187 | Likely Benign | Likely Benign | -0.58 | Ambiguous | 0.0 | -0.36 | Likely Benign | -0.47 | Likely Benign | -0.56 | Ambiguous | 0.359 | Likely Benign | 0.55 | Neutral | 0.019 | Benign | 0.013 | Benign | -1.24 | Pathogenic | 1.00 | Tolerated | 0.0938 | 0.2363 | 0 | 2 | 1.9 | 26.03 | ||||||||||||||||||||||||||||||
| c.2200C>A | P734T 2D  AIThe SynGAP1 missense variant P734T is not reported in ClinVar and is absent from gnomAD. All available in‑silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. The high‑accuracy consensus (SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports the variant as “Likely Benign,” and AlphaMissense‑Optimized also predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no result available for this variant. Based on the unanimous benign predictions and the lack of any pathogenic evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.411273 | Uncertain | 0.368 | 0.721 | 0.875 | -4.469 | Likely Benign | 0.066 | Likely Benign | Likely Benign | 0.042 | Likely Benign | -2.08 | Neutral | 0.040 | Benign | 0.013 | Benign | 2.78 | Benign | 0.09 | Tolerated | 0.1764 | 0.4076 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.2210A>C | Q737P 2D  AIThe SynGAP1 missense variant Q737P is listed in ClinVar (ID 2580571.0) with an uncertain significance designation and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign, while only SIFT indicates a pathogenic effect. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta stability analysis is unavailable. Taken together, the preponderance of evidence supports a benign classification for Q737P, which is consistent with its ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.889439 | Disordered | 0.425743 | Uncertain | 0.323 | 0.803 | 0.875 | Uncertain | 1 | -2.407 | Likely Benign | 0.054 | Likely Benign | Likely Benign | 0.154 | Likely Benign | -1.22 | Neutral | 0.005 | Benign | 0.013 | Benign | 2.78 | Benign | 0.04 | Affected | 4.07 | 3 | 0.2366 | 0.4981 | -1 | 0 | 1.9 | -31.01 | |||||||||||||||||||||||||||||||||||
| c.2296T>C | S766P 2D  AIThe SynGAP1 missense variant S766P is reported in gnomAD (ID 6‑33442454‑T‑C) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized all classify the change as benign, while SIFT uniquely predicts it as pathogenic. The remaining tools, ESM1b and AlphaMissense‑Default, are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, and Foldetta stability analysis is unavailable. Overall, the preponderance of evidence points to a benign impact, and this conclusion does not contradict any ClinVar status, as none is available. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.405110 | Structured | 0.923125 | Binding | 0.338 | 0.874 | 0.250 | 6-33442454-T-C | 1 | 1.28e-6 | -7.343 | In-Between | 0.374 | Ambiguous | Likely Benign | 0.193 | Likely Benign | -0.91 | Neutral | 0.006 | Benign | 0.013 | Benign | 4.07 | Benign | 0.01 | Affected | 3.64 | 6 | 0.2216 | 0.5862 | -1 | 1 | -0.8 | 10.04 | |||||||||||||||||||||||||||||||||||
| c.3160G>A | G1054S 2D  AIThe SynGAP1 missense variant G1054S is listed in ClinVar (ID 699126.0) as Benign and is present in gnomAD (variant ID 6‑33443712‑G‑A). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic or likely pathogenic outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts Benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence strongly supports a benign effect, consistent with the ClinVar designation and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.983019 | Disordered | 0.878015 | Binding | 0.389 | 0.936 | 0.875 | Benign | 1 | 6-33443712-G-A | 32 | 1.99e-5 | -5.294 | Likely Benign | 0.075 | Likely Benign | Likely Benign | 0.160 | Likely Benign | 0.21 | Neutral | 0.121 | Benign | 0.013 | Benign | 4.04 | Benign | 0.63 | Tolerated | 3.77 | 5 | 0.2506 | 0.5311 | 1 | 0 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||
| c.3289C>A | P1097T 2D  AIThe SynGAP1 missense variant P1097T is not reported in ClinVar and is absent from gnomAD, indicating no documented allele frequency data. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, is not available for this variant. Overall, the computational evidence strongly supports a benign impact, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.974957 | Binding | 0.384 | 0.858 | 1.000 | -5.000 | Likely Benign | 0.097 | Likely Benign | Likely Benign | 0.110 | Likely Benign | -1.38 | Neutral | 0.009 | Benign | 0.013 | Benign | 2.61 | Benign | 0.21 | Tolerated | 0.1751 | 0.6567 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.3356G>A | G1119E 2D  AIThe SynGAP1 missense variant G1119E is reported in gnomAD (variant ID 6-33443908‑G‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only ESM1b predicts a pathogenic outcome, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the preponderance of evidence indicates that G1119E is most likely benign, and this conclusion is not contradicted by any ClinVar classification (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.818538 | Binding | 0.339 | 0.928 | 0.875 | 6-33443908-G-A | 1 | 6.61e-7 | -9.151 | Likely Pathogenic | 0.338 | Likely Benign | Likely Benign | 0.284 | Likely Benign | -0.41 | Neutral | 0.005 | Benign | 0.013 | Benign | 3.93 | Benign | 0.12 | Tolerated | 4.32 | 2 | 0.1654 | 0.4259 | -2 | 0 | -3.1 | 72.06 | ||||||||||||||||||||||||||||||||||
| c.3394T>G | S1132A 2D  AIThe SynGAP1 missense variant S1132A is not reported in ClinVar and is absent from gnomAD, indicating no documented population frequency. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign status. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.845506 | Binding | 0.289 | 0.894 | 0.750 | -3.401 | Likely Benign | 0.078 | Likely Benign | Likely Benign | 0.213 | Likely Benign | -0.60 | Neutral | 0.061 | Benign | 0.013 | Benign | 5.47 | Benign | 0.55 | Tolerated | 0.4416 | 0.5683 | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||||||||||||||||
| c.347A>C | Y116S 2D  AIThe SynGAP1 missense variant Y116S is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions strongly suggests that Y116S is most likely benign, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.670235 | Binding | 0.381 | 0.878 | 0.625 | -0.249 | Likely Benign | 0.269 | Likely Benign | Likely Benign | 0.178 | Likely Benign | -0.05 | Neutral | 0.033 | Benign | 0.013 | Benign | 4.31 | Benign | 0.58 | Tolerated | 0.5111 | 0.1694 | Weaken | -3 | -2 | 0.5 | -76.10 | ||||||||||||||||||||||||||||||||||||||
| c.3781A>G | S1261G 2D  AIThe SynGAP1 missense variant S1261G is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates benign; Foldetta results are not available. Overall, the majority of evidence supports a benign impact for S1261G, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.501700 | Disordered | 0.671500 | Binding | 0.889 | 0.574 | 0.250 | -5.042 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.063 | Likely Benign | -0.46 | Neutral | 0.005 | Benign | 0.013 | Benign | 2.32 | Pathogenic | 0.65 | Tolerated | 0.2063 | 0.3477 | 1 | 0 | 0.4 | -30.03 | ||||||||||||||||||||||||||||||||||||||
| c.421A>C | I141L 2D  AIThe SynGAP1 missense variant I141L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM. Only AlphaMissense‑Default predicts a pathogenic outcome, while AlphaMissense‑Optimized is uncertain. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, SGM‑Consensus as Likely Benign, and Foldetta (a protein‑folding stability method combining FoldX‑MD and Rosetta) is not available for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar status, which has no entry for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.465241 | Structured | 0.577021 | Binding | 0.367 | 0.877 | 0.500 | -6.563 | Likely Benign | 0.787 | Likely Pathogenic | Ambiguous | 0.156 | Likely Benign | -1.00 | Neutral | 0.016 | Benign | 0.013 | Benign | 3.84 | Benign | 0.10 | Tolerated | 0.0742 | 0.3180 | 2 | 2 | -0.7 | 0.00 | |||||||||||||||||||||||||||||||||||||||
| c.9G>C | R3S 2D  AIThe SynGAP1 missense variant R3S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta results are not available for this variant. Overall, the majority of evidence points to a benign effect. The prediction is consistent with the lack of ClinVar annotation, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.538167 | Disordered | 0.550331 | Binding | 0.358 | 0.920 | 0.875 | -2.296 | Likely Benign | 0.310 | Likely Benign | Likely Benign | 0.111 | Likely Benign | -0.53 | Neutral | 0.115 | Benign | 0.013 | Benign | 4.02 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3148 | 0.4732 | -1 | 0 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||
| c.9G>T | R3S 2D  AIThe SynGAP1 missense variant R3S is reported in gnomAD (ID 6‑33420273‑G‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus itself is benign; Foldetta results are not available. Overall, the consensus of the majority of prediction tools and the high‑accuracy methods points to a benign effect. This conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.538167 | Disordered | 0.550331 | Binding | 0.358 | 0.920 | 0.875 | 6-33420273-G-T | 1 | 6.50e-7 | -2.296 | Likely Benign | 0.310 | Likely Benign | Likely Benign | 0.111 | Likely Benign | -0.53 | Neutral | 0.115 | Benign | 0.013 | Benign | 4.02 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3148 | 0.4732 | -1 | 0 | 3.7 | -69.11 | ||||||||||||||||||||||||||||||||||
| c.1187G>T | G396V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G396V is catalogued in gnomAD (6‑33438092‑G‑T) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic outcome are FoldX, Rosetta, Foldetta, and PROVEAN. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, while Foldetta (combining FoldX‑MD and Rosetta outputs) indicates a pathogenic effect. Overall, the majority of predictions lean toward a benign impact, and this consensus does not contradict any ClinVar status (none reported). Thus, based on the available computational evidence, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.414856 | Structured | 0.394626 | Uncertain | 0.640 | 0.584 | 0.500 | 6-33438092-G-T | 6 | 3.72e-6 | -5.663 | Likely Benign | 0.120 | Likely Benign | Likely Benign | 3.49 | Destabilizing | 1.7 | 5.28 | Destabilizing | 4.39 | Destabilizing | 0.34 | Likely Benign | 0.332 | Likely Benign | -2.56 | Deleterious | 0.062 | Benign | 0.014 | Benign | 3.90 | Benign | 0.24 | Tolerated | 3.41 | 15 | 0.1287 | 0.4337 | -3 | -1 | 4.6 | 42.08 | ||||||||||||||||||||||||
| c.1298C>G | A433G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A433G is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that reach a consensus all indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the substitution as tolerated. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized classifies the variant as benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports it as Likely Benign. The protein‑folding stability method Foldetta, which integrates FoldX‑MD and Rosetta outputs, yields an uncertain result and is therefore treated as unavailable evidence. Overall, the collective predictions strongly suggest that the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.098513 | Structured | 0.352258 | Uncertain | 0.938 | 0.302 | 0.000 | -5.044 | Likely Benign | 0.107 | Likely Benign | Likely Benign | 0.59 | Ambiguous | 0.0 | 1.21 | Ambiguous | 0.90 | Ambiguous | 0.70 | Ambiguous | 0.038 | Likely Benign | -1.54 | Neutral | 0.035 | Benign | 0.014 | Benign | 3.38 | Benign | 0.15 | Tolerated | 0.1629 | 0.2919 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||
| c.1585A>G | I529V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I529V is not reported in ClinVar (ClinVar status: not listed) but is present in gnomAD (gnomAD ID 6‑33438828‑A‑G). Prediction tools that uniformly indicate a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, the SGM Consensus is Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts Benign. Based on the collective predictions, the variant is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.318242 | Structured | 0.019545 | Uncertain | 0.901 | 0.403 | 0.000 | 6-33438828-A-G | 1 | 6.20e-7 | -2.342 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.18 | Likely Benign | 0.0 | 0.03 | Likely Benign | 0.11 | Likely Benign | 0.36 | Likely Benign | 0.230 | Likely Benign | -0.21 | Neutral | 0.019 | Benign | 0.014 | Benign | -1.24 | Pathogenic | 1.00 | Tolerated | 3.37 | 35 | 0.1043 | 0.3526 | 3 | 4 | -0.3 | -14.03 | ||||||||||||||||||||||||
| c.1922C>T | S641L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S641L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive because it yields an equal split of benign and pathogenic calls. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict any ClinVar annotation, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.125101 | Structured | 0.157322 | Uncertain | 0.786 | 0.270 | 0.000 | -9.501 | Likely Pathogenic | 0.237 | Likely Benign | Likely Benign | 0.33 | Likely Benign | 0.3 | -0.14 | Likely Benign | 0.10 | Likely Benign | 0.32 | Likely Benign | 0.103 | Likely Benign | -4.33 | Deleterious | 0.115 | Benign | 0.014 | Benign | 3.39 | Benign | 0.07 | Tolerated | 0.1248 | 0.5376 | -3 | -2 | 4.6 | 26.08 | ||||||||||||||||||||||||||||||
| c.1976C>A | S659Y 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S659Y is not reported in ClinVar (ClinVar status: none) and is absent from gnomAD (gnomAD ID: none). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default; FoldX is uncertain and therefore not considered evidence. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic. Overall, the majority of individual predictors lean toward a benign impact, and there is no ClinVar entry to contradict this assessment. Thus, based on the available predictions, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.067594 | Structured | 0.154597 | Uncertain | 0.954 | 0.283 | 0.000 | -10.832 | Likely Pathogenic | 0.600 | Likely Pathogenic | Likely Benign | -0.64 | Ambiguous | 0.1 | 0.00 | Likely Benign | -0.32 | Likely Benign | 0.29 | Likely Benign | 0.173 | Likely Benign | -4.24 | Deleterious | 0.084 | Benign | 0.014 | Benign | 3.38 | Benign | 0.04 | Affected | 0.0993 | 0.5962 | -3 | -2 | -0.5 | 76.10 | |||||||||||||||||||||||||||||
| c.2265G>A | M755I 2D  AIThe SynGAP1 missense variant M755I is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification, so there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.783855 | Binding | 0.336 | 0.873 | 0.375 | -3.856 | Likely Benign | 0.240 | Likely Benign | Likely Benign | 0.030 | Likely Benign | -0.55 | Neutral | 0.039 | Benign | 0.014 | Benign | 2.81 | Benign | 0.14 | Tolerated | 0.1059 | 0.2403 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2265G>C | M755I 2D  AIThe SynGAP1 missense variant M755I is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification, so there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.783855 | Binding | 0.336 | 0.873 | 0.375 | -3.856 | Likely Benign | 0.240 | Likely Benign | Likely Benign | 0.031 | Likely Benign | -0.55 | Neutral | 0.039 | Benign | 0.014 | Benign | 2.81 | Benign | 0.14 | Tolerated | 0.1059 | 0.2403 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2265G>T | M755I 2D  AIThe SynGAP1 missense variant M755I is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification, so there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.783855 | Binding | 0.336 | 0.873 | 0.375 | -3.856 | Likely Benign | 0.240 | Likely Benign | Likely Benign | 0.030 | Likely Benign | -0.55 | Neutral | 0.039 | Benign | 0.014 | Benign | 2.81 | Benign | 0.14 | Tolerated | 0.1059 | 0.2403 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2311T>G | S771A 2D  AIThe SynGAP1 missense variant S771A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification—there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.436924 | Structured | 0.922503 | Binding | 0.306 | 0.883 | 0.250 | -4.337 | Likely Benign | 0.107 | Likely Benign | Likely Benign | 0.067 | Likely Benign | -1.09 | Neutral | 0.025 | Benign | 0.014 | Benign | 4.09 | Benign | 0.62 | Tolerated | 0.5201 | 0.4745 | Weaken | 1 | 1 | 2.6 | -16.00 | ||||||||||||||||||||||||||||||||||||||
| c.241C>G | L81V 2D  AIThe SynGAP1 missense variant L81V is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign status. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the preponderance of evidence supports a benign classification for L81V, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.632174 | Disordered | 0.502033 | Binding | 0.291 | 0.878 | 0.250 | -5.207 | Likely Benign | 0.289 | Likely Benign | Likely Benign | 0.038 | Likely Benign | -0.51 | Neutral | 0.178 | Benign | 0.014 | Benign | 4.04 | Benign | 0.00 | Affected | 0.1423 | 0.2653 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.283C>T | H95Y 2D  AIThe SynGAP1 missense variant H95Y is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is Likely Benign. Foldetta results are unavailable. Overall, the majority of evidence indicates that H95Y is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.590542 | Binding | 0.335 | 0.875 | 0.625 | -3.610 | Likely Benign | 0.112 | Likely Benign | Likely Benign | 0.090 | Likely Benign | -1.45 | Neutral | 0.219 | Benign | 0.014 | Benign | 4.14 | Benign | 0.00 | Affected | 0.0880 | 0.4122 | 0 | 2 | 1.9 | 26.03 | |||||||||||||||||||||||||||||||||||||||
| c.3350G>T | G1117V 2D  AIThe SynGAP1 missense variant G1117V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while ESM1b is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.882776 | Disordered | 0.853192 | Binding | 0.323 | 0.914 | 0.750 | -7.251 | In-Between | 0.083 | Likely Benign | Likely Benign | 0.284 | Likely Benign | -1.32 | Neutral | 0.011 | Benign | 0.014 | Benign | 4.57 | Benign | 0.04 | Affected | 0.1312 | 0.3680 | -1 | -3 | 4.6 | 42.08 | |||||||||||||||||||||||||||||||||||||||
| c.373C>A | P125T 2D  AIThe SynGAP1 missense variant P125T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.704227 | Binding | 0.373 | 0.878 | 0.625 | -4.780 | Likely Benign | 0.273 | Likely Benign | Likely Benign | 0.215 | Likely Benign | -3.78 | Deleterious | 0.036 | Benign | 0.014 | Benign | 2.85 | Benign | 0.01 | Affected | 0.1502 | 0.4995 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.40C>A | P14T 2D  AIThe SynGAP1 missense variant P14T is reported in gnomAD (ID 6‑33420304‑C‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus also reports likely benign; Foldetta results are not available. Overall, the preponderance of evidence points to a benign impact for P14T, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.471596 | Uncertain | 0.399 | 0.909 | 0.375 | 6-33420304-C-A | -3.261 | Likely Benign | 0.150 | Likely Benign | Likely Benign | 0.078 | Likely Benign | -0.27 | Neutral | 0.212 | Benign | 0.014 | Benign | 4.20 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1912 | 0.6658 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||||
| c.40C>T | P14S 2D  AIThe SynGAP1 missense variant P14S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.471596 | Uncertain | 0.399 | 0.909 | 0.375 | -2.352 | Likely Benign | 0.123 | Likely Benign | Likely Benign | 0.068 | Likely Benign | -0.02 | Neutral | 0.212 | Benign | 0.014 | Benign | 4.23 | Benign | 0.00 | Affected | 0.3935 | 0.5938 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.97C>G | Q33E 2D  AIThe SynGAP1 missense variant Q33E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign status: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.436712 | Uncertain | 0.342 | 0.860 | 0.375 | 0.191 | Likely Benign | 0.110 | Likely Benign | Likely Benign | 0.024 | Likely Benign | -0.13 | Neutral | 0.017 | Benign | 0.014 | Benign | 4.25 | Benign | 0.00 | Affected | 0.1773 | 0.3326 | 2 | 2 | 0.0 | 0.98 | |||||||||||||||||||||||||||||||||||||||
| c.1511A>G | K504R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant K504R is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33438543‑A‑G). Consensus from most in‑silico predictors is benign: REVEL, FoldX, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all report a benign effect, while only FATHMM predicts pathogenicity. Uncertain calls come from Rosetta and premPS. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, also predicts benign. Overall, the preponderance of evidence indicates the variant is most likely benign, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.028107 | Structured | 0.304984 | Uncertain | 0.850 | 0.189 | 0.000 | Uncertain | 1 | 6-33438543-A-G | 2 | 1.24e-6 | -4.365 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.13 | Likely Benign | 0.1 | 0.51 | Ambiguous | 0.32 | Likely Benign | 0.94 | Ambiguous | 0.238 | Likely Benign | -2.16 | Neutral | 0.002 | Benign | 0.015 | Benign | -1.41 | Pathogenic | 0.11 | Tolerated | 3.37 | 35 | 0.3363 | 0.0647 | 2 | 3 | -0.6 | 28.01 | ||||||||||||||||||||||
| c.1663G>C | V555L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V555L is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The remaining tools—FoldX, Foldetta, ESM1b, and AlphaMissense‑Default—return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is unavailable due to a tie between pathogenic and benign calls, and Foldetta is uncertain. Overall, the preponderance of evidence points to a benign effect. This conclusion is consistent with the lack of ClinVar annotation and does not contradict any existing clinical classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.013265 | Structured | 0.008218 | Uncertain | 0.943 | 0.225 | 0.000 | -7.816 | In-Between | 0.521 | Ambiguous | Likely Benign | -1.43 | Ambiguous | 0.1 | -0.14 | Likely Benign | -0.79 | Ambiguous | 0.16 | Likely Benign | 0.260 | Likely Benign | -0.97 | Neutral | 0.025 | Benign | 0.015 | Benign | -1.22 | Pathogenic | 0.14 | Tolerated | 0.1036 | 0.2958 | 2 | 1 | -0.4 | 14.03 | ||||||||||||||||||||||||||||||
| c.2227C>T | P743S 2D  AIThe SynGAP1 missense variant P743S is listed in gnomAD (ID 6‑33441692‑C‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all classify the change as benign. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support benignity: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is “Likely Benign.” No Foldetta stability data are available, so it does not influence the assessment. Overall, the preponderance of evidence indicates that P743S is most likely benign, and this conclusion is not contradicted by any ClinVar status (none is reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.852992 | Disordered | 0.526809 | Binding | 0.317 | 0.862 | 0.875 | 6-33441692-C-T | 1 | 6.19e-7 | -4.286 | Likely Benign | 0.063 | Likely Benign | Likely Benign | 0.085 | Likely Benign | -0.33 | Neutral | 0.021 | Benign | 0.015 | Benign | 2.93 | Benign | 0.00 | Affected | 4.32 | 2 | 0.3087 | 0.3948 | -1 | 1 | 0.8 | -10.04 | 10.1016/j.ajhg.2020.11.011 | |||||||||||||||||||||||||||||||||
| c.2257G>T | A753S 2D  AIThe SynGAP1 missense variant A753S is reported as “Likely Benign” in ClinVar and is not present in gnomAD. Prediction tools that assess functional impact all converge on a benign outcome: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool in the dataset predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates a likely benign effect. Foldetta results are unavailable. Overall, the variant is most likely benign, and this assessment does not contradict the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.521092 | Disordered | 0.722781 | Binding | 0.381 | 0.873 | 0.625 | -3.656 | Likely Benign | 0.069 | Likely Benign | Likely Benign | 0.105 | Likely Benign | 0.25 | Neutral | 0.062 | Benign | 0.015 | Benign | 3.03 | Benign | 0.59 | Tolerated | 0.2905 | 0.5899 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.229A>C | S77R 2D  AIThe SynGAP1 missense variant S77R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, and the SGM‑Consensus (majority of the high‑accuracy tools) also indicates Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the collective evidence points to a benign effect for S77R, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.446124 | Uncertain | 0.310 | 0.855 | 0.375 | -2.823 | Likely Benign | 0.482 | Ambiguous | Likely Benign | 0.011 | Likely Benign | -1.22 | Neutral | 0.198 | Benign | 0.015 | Benign | 4.09 | Benign | 0.00 | Affected | 0.0752 | 0.3247 | 0 | -1 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||||
| c.231T>A | S77R 2D  AIThe SynGAP1 missense variant S77R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority of the high‑accuracy tools) is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.446124 | Uncertain | 0.310 | 0.855 | 0.375 | -2.823 | Likely Benign | 0.482 | Ambiguous | Likely Benign | 0.026 | Likely Benign | -1.22 | Neutral | 0.198 | Benign | 0.015 | Benign | 4.09 | Benign | 0.00 | Affected | 0.0752 | 0.3247 | 0 | -1 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||||
| c.231T>G | S77R 2D  AIThe SynGAP1 missense variant S77R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority of the high‑accuracy tools) is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.446124 | Uncertain | 0.310 | 0.855 | 0.375 | -2.823 | Likely Benign | 0.482 | Ambiguous | Likely Benign | 0.026 | Likely Benign | -1.22 | Neutral | 0.198 | Benign | 0.015 | Benign | 4.09 | Benign | 0.00 | Affected | 0.0752 | 0.3247 | 0 | -1 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||||
| c.232C>A | R78S 2D  AIThe SynGAP1 missense variant R78S is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. AlphaMissense‑Optimized returns an uncertain result. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments are: AlphaMissense‑Optimized (Uncertain), SGM‑Consensus (Likely Benign), and Foldetta (no data available). Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict the ClinVar status, which contains no pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.448183 | Uncertain | 0.304 | 0.866 | 0.500 | -3.680 | Likely Benign | 0.792 | Likely Pathogenic | Ambiguous | 0.063 | Likely Benign | -1.11 | Neutral | 0.385 | Benign | 0.015 | Benign | 3.86 | Benign | 0.00 | Affected | 0.3081 | 0.3274 | 0 | -1 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||||
| c.2564T>G | L855R 2D  AIThe SynGAP1 missense variant L855R is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.724957 | Disordered | 0.485558 | Uncertain | 0.285 | 0.823 | 0.625 | -3.703 | Likely Benign | 0.229 | Likely Benign | Likely Benign | 0.086 | Likely Benign | -1.62 | Neutral | 0.026 | Benign | 0.015 | Benign | 4.00 | Benign | 0.03 | Affected | 0.1303 | 0.1187 | -3 | -2 | -8.3 | 43.03 | |||||||||||||||||||||||||||||||||||||||
| c.268G>C | V90L 2D  AIThe SynGAP1 missense variant V90L is not reported in ClinVar and has no entries in gnomAD, indicating it is not catalogued in these databases. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign, while SIFT uniquely predicts it as pathogenic. The consensus from the SGM framework, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a Likely Benign verdict. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized reports a benign outcome, the SGM Consensus also indicates Likely Benign, and Foldetta data are unavailable. With the majority of evidence pointing to a benign effect and no conflicting ClinVar annotation, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.570702 | Disordered | 0.542047 | Binding | 0.343 | 0.873 | 0.500 | -3.676 | Likely Benign | 0.522 | Ambiguous | Likely Benign | 0.056 | Likely Benign | -0.24 | Neutral | 0.103 | Benign | 0.015 | Benign | 4.02 | Benign | 0.00 | Affected | 0.1334 | 0.4487 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.268G>T | V90L 2D  AIThe SynGAP1 missense variant V90L is not reported in ClinVar and has no entries in gnomAD, indicating it is not catalogued in these databases. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, SGM‑Consensus indicates Likely Benign, and Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign effect for V90L, and this conclusion does not contradict any ClinVar status, as none is assigned. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.570702 | Disordered | 0.542047 | Binding | 0.343 | 0.873 | 0.500 | -3.676 | Likely Benign | 0.522 | Ambiguous | Likely Benign | 0.056 | Likely Benign | -0.24 | Neutral | 0.103 | Benign | 0.015 | Benign | 4.02 | Benign | 0.00 | Affected | 0.1334 | 0.4487 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2701G>T | A901S 2D  AIThe SynGAP1 missense variant A901S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools that assess pathogenicity uniformly predict a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate benign. No tool in the dataset predicts pathogenicity, so the pathogenic group is empty. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Overall, the collective predictions strongly suggest that the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.489838 | Uncertain | 0.306 | 0.917 | 0.375 | -3.284 | Likely Benign | 0.097 | Likely Benign | Likely Benign | 0.043 | Likely Benign | -0.62 | Neutral | 0.009 | Benign | 0.015 | Benign | 2.73 | Benign | 0.35 | Tolerated | 0.2695 | 0.6582 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.2746G>A | V916I 2D  AIThe SynGAP1 missense variant V916I is reported in gnomAD (variant ID 6-33443298‑G‑A) but has no ClinVar entry. Functional prediction tools uniformly classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate a benign effect. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability predictor, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and this assessment is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.642678 | Disordered | 0.835395 | Binding | 0.308 | 0.879 | 0.250 | 6-33443298-G-A | 25 | 1.55e-5 | -4.336 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.062 | Likely Benign | -0.05 | Neutral | 0.010 | Benign | 0.015 | Benign | 2.71 | Benign | 0.09 | Tolerated | 3.77 | 5 | 0.0905 | 0.4463 | 3 | 4 | 0.3 | 14.03 | ||||||||||||||||||||||||||||||||||
| c.280C>A | P94T 2D  AIThe SynGAP1 missense variant P94T is reported in gnomAD (variant ID 6‑33425888‑C‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign status. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Consequently, the collective evidence indicates that P94T is most likely benign, and this assessment does not contradict any ClinVar classification because none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.570978 | Binding | 0.350 | 0.869 | 0.625 | 6-33425888-C-A | 1 | 6.20e-7 | -4.254 | Likely Benign | 0.085 | Likely Benign | Likely Benign | 0.088 | Likely Benign | -2.35 | Neutral | 0.198 | Benign | 0.015 | Benign | 4.12 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1326 | 0.4789 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||
| c.2824C>T | P942S 2D  AIThe SynGAP1 missense variant P942S is catalogued in gnomAD (ID 6‑33443376‑C‑T) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.878102 | Binding | 0.365 | 0.915 | 0.625 | 6-33443376-C-T | 1 | 6.20e-7 | -3.919 | Likely Benign | 0.062 | Likely Benign | Likely Benign | 0.036 | Likely Benign | -0.80 | Neutral | 0.011 | Benign | 0.015 | Benign | 2.43 | Pathogenic | 0.00 | Affected | 4.32 | 4 | 0.3226 | 0.4806 | -1 | 1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||||
| c.2825C>A | P942Q 2D  AIThe SynGAP1 missense variant P942Q is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact for P942Q, and this conclusion does not contradict any ClinVar annotation (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.878102 | Binding | 0.365 | 0.915 | 0.625 | -6.094 | Likely Benign | 0.080 | Likely Benign | Likely Benign | 0.028 | Likely Benign | -1.12 | Neutral | 0.026 | Benign | 0.015 | Benign | 2.36 | Pathogenic | 0.00 | Affected | 0.1508 | 0.4703 | 0 | -1 | -1.9 | 31.01 | |||||||||||||||||||||||||||||||||||||||
| c.2855G>A | G952D 2D  AIThe SynGAP1 missense variant G952D is not reported in ClinVar and is absent from gnomAD. Consensus predictions from multiple in‑silico tools overwhelmingly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score benign, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is labeled Likely Benign. Only SIFT predicts a pathogenic outcome, and ESM1b remains uncertain. High‑accuracy assessments corroborate the benign trend: AlphaMissense‑Optimized is benign and the SGM‑Consensus is Likely Benign; Foldetta results are not available. Overall, the computational evidence strongly supports a benign classification, and this is consistent with the lack of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.985964 | Disordered | 0.910621 | Binding | 0.341 | 0.926 | 0.750 | -7.299 | In-Between | 0.274 | Likely Benign | Likely Benign | 0.241 | Likely Benign | -0.75 | Neutral | 0.033 | Benign | 0.015 | Benign | 3.20 | Benign | 0.05 | Affected | 0.1928 | 0.2826 | 1 | -1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||||||||||||
| c.2857C>A | P953T 2D  AIThe SynGAP1 missense variant P953T is predicted to be benign by all evaluated in silico tools. Consensus predictions from SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) classify the variant as Likely Benign. High‑accuracy predictors AlphaMissense‑Optimized also report a benign effect. Other pathogenicity predictors—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM)—uniformly predict benign. No tools predict pathogenicity. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its stability impact remains unknown. The variant is not listed in ClinVar and has no entry in gnomAD, so no population frequency or clinical classification is available. Based on the unanimous benign predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status because none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.983019 | Disordered | 0.920633 | Binding | 0.403 | 0.926 | 0.750 | -6.646 | Likely Benign | 0.072 | Likely Benign | Likely Benign | 0.075 | Likely Benign | -0.80 | Neutral | 0.009 | Benign | 0.015 | Benign | 2.88 | Benign | 0.24 | Tolerated | 0.2261 | 0.6004 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.2858C>A | P953Q 2D  AIThe SynGAP1 missense variant P953Q is listed in ClinVar with an uncertain significance (ClinVar ID 1176820.0) and is not found in gnomAD. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign effects, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also reports likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions points to a benign impact, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.983019 | Disordered | 0.920633 | Binding | 0.403 | 0.926 | 0.750 | Uncertain | 1 | -6.038 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.086 | Likely Benign | -0.78 | Neutral | 0.058 | Benign | 0.015 | Benign | 2.78 | Benign | 0.29 | Tolerated | 3.77 | 5 | 0.2105 | 0.5108 | 0 | -1 | -1.9 | 31.01 | |||||||||||||||||||||||||||||||||||
| c.2965T>C | S989P 2D  AIThe SynGAP1 missense variant S989P is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign; Foldetta results are unavailable. Overall, the consensus of available predictions indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.795062 | Disordered | 0.908835 | Binding | 0.296 | 0.911 | 0.750 | -2.688 | Likely Benign | 0.209 | Likely Benign | Likely Benign | 0.147 | Likely Benign | -2.48 | Neutral | 0.010 | Benign | 0.015 | Benign | 2.67 | Benign | 0.00 | Affected | 0.1690 | 0.4941 | 1 | -1 | -0.8 | 10.04 | |||||||||||||||||||||||||||||||||||||||
| c.2983C>T | P995S 2D  AIThe SynGAP1 missense variant P995S is listed in ClinVar (ID 436929.0) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). The only tool that predicts a pathogenic outcome is SIFT. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates “Likely Benign.” No Foldetta (FoldX‑MD/Rosetta) stability result is available, so it does not influence the assessment. Overall, the majority of predictions, including the high‑accuracy tools, point to a benign effect, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.862302 | Disordered | 0.935305 | Binding | 0.338 | 0.902 | 0.750 | Uncertain | 1 | -4.457 | Likely Benign | 0.071 | Likely Benign | Likely Benign | 0.042 | Likely Benign | -1.03 | Neutral | 0.011 | Benign | 0.015 | Benign | 4.24 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3204 | 0.5236 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||
| c.3086A>C | Q1029P 2D  AIThe SynGAP1 missense variant Q1029P is not reported in ClinVar and is absent from gnomAD, indicating no known population frequency data. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta results are not available for this variant. Overall, the consensus of all available predictions strongly supports a benign impact, and this conclusion is consistent with the lack of ClinVar evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.852992 | Disordered | 0.995643 | Binding | 0.375 | 0.734 | 0.500 | -2.940 | Likely Benign | 0.124 | Likely Benign | Likely Benign | 0.105 | Likely Benign | -1.23 | Neutral | 0.005 | Benign | 0.015 | Benign | 2.68 | Benign | 0.15 | Tolerated | 0.1966 | 0.4986 | 0 | -1 | 1.9 | -31.01 | |||||||||||||||||||||||||||||||||||||||
| c.3100C>T | P1034S 2D  AIThe SynGAP1 missense variant P1034S is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact for P1034S, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.926919 | Disordered | 0.991713 | Binding | 0.343 | 0.752 | 0.625 | -3.730 | Likely Benign | 0.262 | Likely Benign | Likely Benign | 0.054 | Likely Benign | -2.28 | Neutral | 0.011 | Benign | 0.015 | Benign | 2.44 | Pathogenic | 0.05 | Affected | 0.3044 | 0.5853 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.3130C>T | P1044S 2D  AIThe SynGAP1 missense variant P1044S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.979741 | Disordered | 0.952126 | Binding | 0.331 | 0.855 | 0.750 | -4.114 | Likely Benign | 0.078 | Likely Benign | Likely Benign | 0.311 | Likely Benign | -0.79 | Neutral | 0.011 | Benign | 0.015 | Benign | 5.51 | Benign | 0.14 | Tolerated | 0.3194 | 0.6081 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.3131C>A | P1044Q 2D  AIThe SynGAP1 missense variant P1044Q is not reported in ClinVar and is absent from gnomAD. All evaluated in‑silico predictors—REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods likewise support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.979741 | Disordered | 0.952126 | Binding | 0.331 | 0.855 | 0.750 | -4.439 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.338 | Likely Benign | 0.51 | Neutral | 0.004 | Benign | 0.015 | Benign | 5.43 | Benign | 0.45 | Tolerated | 0.1544 | 0.5591 | 0 | -1 | -1.9 | 31.01 | |||||||||||||||||||||||||||||||||||||||
| c.3335A>G | E1112G 2D  AIThe SynGAP1 missense variant E1112G is reported in gnomAD (ID 6‑33443887‑A‑G) but has no ClinVar entry. Functional prediction tools cluster into two groups: benign predictions include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; pathogenic predictions come from PROVEAN and SIFT. The high‑accuracy consensus, SGM‑Consensus, is derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yielding a “Likely Benign” classification. AlphaMissense‑Optimized independently predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, is not available for this variant. Overall, the preponderance of evidence from both general and high‑accuracy tools indicates the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is assigned). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.894241 | Disordered | 0.909381 | Binding | 0.335 | 0.902 | 0.875 | 6-33443887-A-G | 1 | 6.73e-7 | -3.459 | Likely Benign | 0.277 | Likely Benign | Likely Benign | 0.124 | Likely Benign | -2.58 | Deleterious | 0.058 | Benign | 0.015 | Benign | 2.70 | Benign | 0.01 | Affected | 4.32 | 2 | 0.2795 | 0.6220 | -2 | 0 | 3.1 | -72.06 | ||||||||||||||||||||||||||||||||||
| c.3361A>C | S1121R 2D  AIThe SynGAP1 missense variant S1121R is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign, reflecting the majority of benign predictions. High‑accuracy assessments show AlphaMissense‑Optimized as benign, and the SGM‑Consensus (majority vote) also indicates benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact for S1121R, and this conclusion is consistent with the lack of ClinVar reporting. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.894241 | Disordered | 0.810024 | Binding | 0.365 | 0.935 | 0.875 | -6.945 | Likely Benign | 0.597 | Likely Pathogenic | Likely Benign | 0.142 | Likely Benign | -0.34 | Neutral | 0.016 | Benign | 0.015 | Benign | 5.45 | Benign | 0.00 | Affected | 3.77 | 5 | 0.1346 | 0.3431 | -1 | 0 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||
| c.3363C>A | S1121R 2D  AIThe SynGAP1 missense variant S1121R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.894241 | Disordered | 0.810024 | Binding | 0.365 | 0.935 | 0.875 | -6.945 | Likely Benign | 0.597 | Likely Pathogenic | Likely Benign | 0.100 | Likely Benign | -0.34 | Neutral | 0.016 | Benign | 0.015 | Benign | 5.45 | Benign | 0.00 | Affected | 3.77 | 5 | 0.1346 | 0.3431 | -1 | 0 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||
| c.3363C>G | S1121R 2D  AIThe SynGAP1 missense variant S1121R is catalogued in gnomAD (ID 6‑33443915‑C‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized all report benign. Only SIFT and AlphaMissense‑Default predict pathogenicity, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.894241 | Disordered | 0.810024 | Binding | 0.365 | 0.935 | 0.875 | 6-33443915-C-G | -6.945 | Likely Benign | 0.597 | Likely Pathogenic | Likely Benign | 0.101 | Likely Benign | -0.34 | Neutral | 0.016 | Benign | 0.015 | Benign | 5.45 | Benign | 0.00 | Affected | 3.77 | 5 | 0.1346 | 0.3431 | -1 | 0 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||||||
| c.3391C>T | P1131S 2D  AIThe SynGAP1 missense variant P1131S is reported in gnomAD (variant ID 6‑33443943‑C‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. In contrast, PROVEAN and SIFT predict a pathogenic impact. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a “Likely Benign” verdict. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Thus, the variant is most likely benign, and this is not contradictory to ClinVar. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.855155 | Binding | 0.360 | 0.899 | 0.750 | 6-33443943-C-T | 1 | 6.72e-7 | -4.089 | Likely Benign | 0.246 | Likely Benign | Likely Benign | 0.209 | Likely Benign | -2.62 | Deleterious | 0.025 | Benign | 0.015 | Benign | 5.54 | Benign | 0.00 | Affected | 4.32 | 4 | 0.2936 | 0.5936 | -1 | 1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||||
| c.3424T>G | S1142A 2D  AIThe SynGAP1 missense variant S1142A is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it to be pathogenic. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports the variant as “Likely Benign.” High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) confirms a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that S1142A is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.719935 | Binding | 0.276 | 0.844 | 1.000 | -3.694 | Likely Benign | 0.085 | Likely Benign | Likely Benign | 0.074 | Likely Benign | -1.31 | Neutral | 0.002 | Benign | 0.015 | Benign | 2.73 | Benign | 0.00 | Affected | 0.4689 | 0.5319 | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||||||||||||||||
| c.34A>G | S12G 2D  AIThe SynGAP1 missense variant S12G is reported in gnomAD (ID 6‑33420298‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign status. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence points to a benign effect for S12G, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.480142 | Structured | 0.490599 | Uncertain | 0.355 | 0.916 | 0.500 | 6-33420298-A-G | -4.229 | Likely Benign | 0.093 | Likely Benign | Likely Benign | 0.079 | Likely Benign | 0.22 | Neutral | 0.103 | Benign | 0.015 | Benign | 4.11 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2876 | 0.5080 | 0 | 1 | 0.4 | -30.03 | ||||||||||||||||||||||||||||||||||||
| c.4022C>A | A1341E 2D  AIThe SynGAP1 missense variant A1341E is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic call comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign consensus. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign. Foldetta stability analysis is not available for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.980111 | Binding | 0.383 | 0.696 | 1.000 | -3.217 | Likely Benign | 0.307 | Likely Benign | Likely Benign | 0.056 | Likely Benign | -1.18 | Neutral | 0.012 | Benign | 0.015 | Benign | 4.05 | Benign | 0.01 | Affected | 0.1633 | 0.2624 | 0 | -1 | -5.3 | 58.04 | |||||||||||||||||||||||||||||||||||||||
| c.44C>T | A15V 2D  AIThe SynGAP1 missense variant A15V is listed in ClinVar (ID 1801174.0) with an “Uncertain” status and is present in gnomAD (6‑33420308‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized, while polyPhen‑2 HumDiv and SIFT predict a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this does not contradict the ClinVar designation, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.436924 | Structured | 0.466055 | Uncertain | 0.330 | 0.912 | 0.375 | Uncertain | 1 | 6-33420308-C-T | 1 | 6.49e-7 | -3.560 | Likely Benign | 0.161 | Likely Benign | Likely Benign | 0.105 | Likely Benign | 0.20 | Neutral | 0.602 | Possibly Damaging | 0.015 | Benign | 4.19 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1809 | 0.7669 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||
| c.502C>A | H168N 2D  AIThe SynGAP1 missense variant H168N is not reported in ClinVar and is absent from gnomAD. All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. No tools predict pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.502450 | Binding | 0.402 | 0.678 | 0.125 | -4.764 | Likely Benign | 0.089 | Likely Benign | Likely Benign | 0.081 | Likely Benign | -0.14 | Neutral | 0.016 | Benign | 0.015 | Benign | 4.29 | Benign | 0.60 | Tolerated | 0.1516 | 0.2589 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.658T>G | F220V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F220V is not reported in ClinVar and is absent from gnomAD. Prediction tools that classify the variant as benign include polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. The remaining tools—REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized—predict a pathogenic effect. High‑accuracy assessments further support a deleterious impact: AlphaMissense‑Optimized is pathogenic; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is likely pathogenic; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is pathogenic. Based on the preponderance of pathogenic predictions, the variant is most likely pathogenic, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.219301 | Structured | 0.429422 | Uncertain | 0.898 | 0.295 | 0.000 | -11.599 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 3.83 | Destabilizing | 0.1 | 4.11 | Destabilizing | 3.97 | Destabilizing | 1.56 | Destabilizing | 0.911 | Likely Pathogenic | -5.81 | Deleterious | 0.075 | Benign | 0.015 | Benign | 4.04 | Benign | 0.01 | Affected | 0.2351 | 0.3315 | -1 | -1 | 1.4 | -48.04 | |||||||||||||||||||||||||||||
| c.661G>C | E221Q 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 E221Q missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Two tools predict a pathogenic outcome: ESM1b and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (two pathogenic vs two benign), and Foldetta predicts a benign effect. Overall, the majority of evidence (10 benign vs 2 pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.127496 | Structured | 0.413334 | Uncertain | 0.891 | 0.283 | 0.000 | -8.243 | Likely Pathogenic | 0.825 | Likely Pathogenic | Ambiguous | -0.26 | Likely Benign | 0.1 | -0.33 | Likely Benign | -0.30 | Likely Benign | 0.12 | Likely Benign | 0.455 | Likely Benign | -1.64 | Neutral | 0.028 | Benign | 0.015 | Benign | 6.13 | Benign | 0.23 | Tolerated | 0.1207 | 0.7690 | 2 | 2 | 0.0 | -0.98 | ||||||||||||||||||||||||||||||
| c.1039A>G | T347A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T347A is catalogued in gnomAD (ID 6‑33437944‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign or tolerated. Only FATHMM predicts a pathogenic outcome, while Foldetta, premPS, and Rosetta are inconclusive. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized is benign, SGM‑Consensus is Likely Benign, and Foldetta remains uncertain. Overall, the consensus of the majority of tools indicates the variant is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.268042 | Structured | 0.349915 | Uncertain | 0.951 | 0.434 | 0.000 | 6-33437944-A-G | 9 | 5.58e-6 | -5.858 | Likely Benign | 0.086 | Likely Benign | Likely Benign | 0.34 | Likely Benign | 0.1 | 0.70 | Ambiguous | 0.52 | Ambiguous | 0.70 | Ambiguous | 0.093 | Likely Benign | -1.52 | Neutral | 0.031 | Benign | 0.016 | Benign | 1.68 | Pathogenic | 0.42 | Tolerated | 3.37 | 25 | 0.4032 | 0.4615 | 0 | 1 | 2.5 | -30.03 | ||||||||||||||||||||||||
| c.1096A>G | T366A 2D  AIThe SynGAP1 missense variant T366A is not reported in ClinVar and is absent from gnomAD. Consensus among most in silico predictors indicates a benign effect: REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the substitution as tolerated. Only FATHMM predicts a pathogenic outcome, while Foldetta, premPS, and Rosetta are inconclusive and are treated as unavailable evidence. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is Likely Benign, and Foldetta provides no definitive stability change. Overall, the computational evidence overwhelmingly favors a benign classification, and this is consistent with the absence of any ClinVar assertion. Therefore, the variant is most likely benign, with no conflict with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.414856 | Structured | 0.441902 | Uncertain | 0.897 | 0.642 | 0.250 | -3.983 | Likely Benign | 0.082 | Likely Benign | Likely Benign | 0.21 | Likely Benign | 0.5 | 0.86 | Ambiguous | 0.54 | Ambiguous | 0.51 | Ambiguous | 0.046 | Likely Benign | -0.94 | Neutral | 0.031 | Benign | 0.016 | Benign | 1.73 | Pathogenic | 0.45 | Tolerated | 0.4141 | 0.4785 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||
| c.1105A>G | T369A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T369A is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while Rosetta and Foldetta are inconclusive. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, SGM‑Consensus is Likely Benign, and Foldetta remains uncertain. Overall, the preponderance of evidence indicates that T369A is most likely benign, and this conclusion does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.468512 | Structured | 0.437011 | Uncertain | 0.417 | 0.707 | 0.500 | -1.957 | Likely Benign | 0.056 | Likely Benign | Likely Benign | 0.09 | Likely Benign | 0.1 | 1.18 | Ambiguous | 0.64 | Ambiguous | 0.26 | Likely Benign | 0.090 | Likely Benign | -1.93 | Neutral | 0.012 | Benign | 0.016 | Benign | 1.72 | Pathogenic | 0.30 | Tolerated | 0.4538 | 0.5053 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||
| c.1112G>T | S371I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S371I is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from FoldX and Rosetta, which are treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) reports a benign effect. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.444081 | Structured | 0.432086 | Uncertain | 0.294 | 0.746 | 0.375 | -6.888 | Likely Benign | 0.207 | Likely Benign | Likely Benign | 0.50 | Ambiguous | 0.4 | -0.50 | Ambiguous | 0.00 | Likely Benign | -0.11 | Likely Benign | 0.433 | Likely Benign | -1.06 | Neutral | 0.028 | Benign | 0.016 | Benign | 4.62 | Benign | 0.07 | Tolerated | 0.1423 | 0.5924 | -1 | -2 | 5.3 | 26.08 | |||||||||||||||||||||||||||||
| c.142T>C | F48L 2D  AIThe SynGAP1 missense variant F48L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus remains benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of predictions lean toward a benign impact, but the high‑accuracy tools provide conflicting evidence and no Foldetta data is available. Thus, the variant is most likely benign based on the consensus of available predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.298791 | Structured | 0.440452 | Uncertain | 0.558 | 0.707 | 0.125 | -4.955 | Likely Benign | 0.956 | Likely Pathogenic | Likely Pathogenic | 0.108 | Likely Benign | -1.56 | Neutral | 0.022 | Benign | 0.016 | Benign | 4.16 | Benign | 0.00 | Affected | 0.2641 | 0.3082 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.144C>A | F48L 2D  AIThe SynGAP1 missense variant F48L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus remains benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of predictions lean toward a benign impact, but the high‑accuracy tools provide conflicting evidence and no Foldetta data is available. Thus, the variant is most likely benign based on the consensus of available predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.298791 | Structured | 0.440452 | Uncertain | 0.558 | 0.707 | 0.125 | -4.955 | Likely Benign | 0.956 | Likely Pathogenic | Likely Pathogenic | 0.052 | Likely Benign | -1.56 | Neutral | 0.022 | Benign | 0.016 | Benign | 4.16 | Benign | 0.00 | Affected | 0.2641 | 0.3082 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.144C>G | F48L 2D  AIThe SynGAP1 missense variant F48L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus remains benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of predictions lean toward a benign impact, but the high‑accuracy tools provide conflicting evidence and no Foldetta data is available. Thus, the variant is most likely benign based on the consensus of available predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.298791 | Structured | 0.440452 | Uncertain | 0.558 | 0.707 | 0.125 | -4.955 | Likely Benign | 0.956 | Likely Pathogenic | Likely Pathogenic | 0.053 | Likely Benign | -1.56 | Neutral | 0.022 | Benign | 0.016 | Benign | 4.16 | Benign | 0.00 | Affected | 0.2641 | 0.3082 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.2125C>G | L709V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L709V is not reported in ClinVar and is absent from gnomAD. Across the available in‑silico predictors, every tool that produced a definitive call classifies the variant as benign: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. The only inconclusive results are FoldX (uncertain) and Foldetta (uncertain), which are treated as unavailable evidence. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized is benign; the SGM‑Consensus, derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is also benign; Foldetta remains uncertain. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.243554 | Structured | 0.365830 | Uncertain | 0.934 | 0.379 | 0.000 | -4.406 | Likely Benign | 0.101 | Likely Benign | Likely Benign | 0.75 | Ambiguous | 0.0 | 0.42 | Likely Benign | 0.59 | Ambiguous | 0.03 | Likely Benign | 0.063 | Likely Benign | -0.36 | Neutral | 0.062 | Benign | 0.016 | Benign | 3.42 | Benign | 0.45 | Tolerated | 0.1162 | 0.2460 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||
| c.2699C>T | T900M 2D  AIThe SynGAP1 missense variant T900M is listed in ClinVar (ID 1063691.0) with an “Uncertain” clinical significance and is present in the gnomAD database (gnomAD ID 6‑33443251‑C‑T). Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign effects. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, which does not contradict the ClinVar “Uncertain” status. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.465347 | Uncertain | 0.289 | 0.924 | 0.375 | Conflicting | 2 | 6-33443251-C-T | 14 | 8.68e-6 | -3.852 | Likely Benign | 0.176 | Likely Benign | Likely Benign | 0.015 | Likely Benign | -0.81 | Neutral | 0.060 | Benign | 0.016 | Benign | 2.79 | Benign | 0.08 | Tolerated | 4.32 | 4 | 0.1356 | 0.6533 | -1 | -1 | 2.6 | 30.09 | ||||||||||||||||||||||||||||||||
| c.2995T>G | S999A 2D  AIThe SynGAP1 missense variant S999A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion is consistent with the lack of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.834292 | Disordered | 0.950682 | Binding | 0.262 | 0.897 | 0.625 | -3.719 | Likely Benign | 0.075 | Likely Benign | Likely Benign | 0.051 | Likely Benign | -0.83 | Neutral | 0.005 | Benign | 0.016 | Benign | 2.71 | Benign | 0.81 | Tolerated | 0.4516 | 0.5434 | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||||||||||||||||
| c.3398T>G | I1133S 2D  AIThe SynGAP1 missense variant I1133S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign classification. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign. Foldetta results are unavailable, so they do not influence the assessment. Overall, the collective evidence strongly supports a benign impact for I1133S, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.832785 | Binding | 0.316 | 0.892 | 0.750 | -4.034 | Likely Benign | 0.492 | Ambiguous | Likely Benign | 0.186 | Likely Benign | -0.60 | Neutral | 0.007 | Benign | 0.016 | Benign | 5.70 | Benign | 0.08 | Tolerated | 0.2762 | 0.1110 | -1 | -2 | -5.3 | -26.08 | |||||||||||||||||||||||||||||||||||||||
| c.3442A>C | M1148L 2D  AIThe SynGAP1 missense variant M1148L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and there is no conflict with ClinVar status because no ClinVar classification exists. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.774279 | Binding | 0.343 | 0.835 | 0.875 | -1.777 | Likely Benign | 0.093 | Likely Benign | Likely Benign | 0.068 | Likely Benign | -1.13 | Neutral | 0.016 | Benign | 0.016 | Benign | 2.62 | Benign | 0.00 | Affected | 4.32 | 2 | 0.1641 | 0.4135 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||
| c.3442A>T | M1148L 2D  AIThe SynGAP1 missense variant M1148L is listed in ClinVar (ID 1010061.0) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this is consistent with the ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.774279 | Binding | 0.343 | 0.835 | 0.875 | Uncertain | 1 | -1.777 | Likely Benign | 0.093 | Likely Benign | Likely Benign | 0.068 | Likely Benign | -1.13 | Neutral | 0.016 | Benign | 0.016 | Benign | 2.62 | Benign | 0.00 | Affected | 4.32 | 2 | 0.1641 | 0.4135 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||
| c.430A>T | T144S 2D  AIThe SynGAP1 missense variant T144S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default, while ESM1b remains uncertain. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact for T144S. This conclusion does not contradict ClinVar, which has no entry for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.666105 | Disordered | 0.524000 | Binding | 0.335 | 0.838 | 0.625 | -7.730 | In-Between | 0.672 | Likely Pathogenic | Likely Benign | 0.081 | Likely Benign | -1.90 | Neutral | 0.018 | Benign | 0.016 | Benign | 3.84 | Benign | 0.00 | Affected | 0.4039 | 0.4183 | 1 | 1 | -0.1 | -14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.590A>C | E197A 2D  AIThe SynGAP1 E197A missense variant is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Those that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. Two tools give uncertain results: ESM1b and AlphaMissense‑Optimized. High‑accuracy assessment shows that the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) predicts pathogenicity, while AlphaMissense‑Optimized remains uncertain and Foldetta (which would combine FoldX‑MD and Rosetta outputs) has no available result. Overall, the balance of evidence—including the pathogenic majority in the high‑accuracy consensus—suggests that the variant is most likely pathogenic. This conclusion does not contradict ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.349426 | Structured | 0.431896 | Uncertain | 0.452 | 0.492 | 0.125 | -7.956 | In-Between | 0.787 | Likely Pathogenic | Ambiguous | 0.155 | Likely Benign | -3.56 | Deleterious | 0.055 | Benign | 0.016 | Benign | 4.06 | Benign | 0.02 | Affected | 0.3134 | 0.5290 | 0 | -1 | 5.3 | -58.04 | ||||||||||||||||||||||||||||||||||||||||
| c.94A>G | T32A 2D  AIThe SynGAP1 missense variant T32A is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.437154 | Uncertain | 0.349 | 0.879 | 0.375 | -3.330 | Likely Benign | 0.072 | Likely Benign | Likely Benign | 0.047 | Likely Benign | -0.63 | Neutral | 0.043 | Benign | 0.016 | Benign | 4.21 | Benign | 0.00 | Affected | 0.4174 | 0.4742 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||||||||||||
| c.1831A>C | M611L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M611L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote). Only FATHMM predicts a pathogenic outcome, while Rosetta, Foldetta, and premPS are inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as Likely Benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.236433 | Structured | 0.210791 | Uncertain | 0.870 | 0.253 | 0.000 | -6.721 | Likely Benign | 0.111 | Likely Benign | Likely Benign | 0.39 | Likely Benign | 0.1 | 0.65 | Ambiguous | 0.52 | Ambiguous | 0.56 | Ambiguous | 0.229 | Likely Benign | -1.29 | Neutral | 0.059 | Benign | 0.017 | Benign | -1.07 | Pathogenic | 0.76 | Tolerated | 0.1130 | 0.3547 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||
| c.1831A>T | M611L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M611L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: SGM‑Consensus (Likely Benign), REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only FATHMM predicts pathogenic, while Rosetta, Foldetta, and premPS are uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates Likely Benign; Foldetta, which integrates FoldX‑MD and Rosetta outputs, remains uncertain. Overall, the preponderance of evidence points to a benign impact for M611L, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.236433 | Structured | 0.210791 | Uncertain | 0.870 | 0.253 | 0.000 | -6.721 | Likely Benign | 0.111 | Likely Benign | Likely Benign | 0.39 | Likely Benign | 0.1 | 0.65 | Ambiguous | 0.52 | Ambiguous | 0.56 | Ambiguous | 0.229 | Likely Benign | -1.29 | Neutral | 0.059 | Benign | 0.017 | Benign | -1.07 | Pathogenic | 0.76 | Tolerated | 0.1130 | 0.3547 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||
| c.2027G>C | S676T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S676T is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and Rosetta. No tool predicts a pathogenic outcome; the only inconclusive results are from FoldX (Uncertain) and Foldetta (Uncertain). High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign, and Foldetta remains Uncertain. Overall, the consensus of available predictions indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar status because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.209395 | Structured | 0.113632 | Uncertain | 0.551 | 0.338 | 0.125 | -5.621 | Likely Benign | 0.092 | Likely Benign | Likely Benign | 0.79 | Ambiguous | 0.1 | 0.34 | Likely Benign | 0.57 | Ambiguous | -0.36 | Likely Benign | 0.098 | Likely Benign | 0.91 | Neutral | 0.050 | Benign | 0.017 | Benign | 3.52 | Benign | 1.00 | Tolerated | 0.1575 | 0.6286 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.223G>A | E75K 2D  AIThe SynGAP1 missense variant E75K is listed in ClinVar as Benign (ClinVar ID 3360083.0) and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, aligning with the ClinVar classification and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.443881 | Uncertain | 0.303 | 0.822 | 0.500 | Benign/Likely benign | 2 | -4.020 | Likely Benign | 0.358 | Ambiguous | Likely Benign | 0.134 | Likely Benign | -1.12 | Neutral | 0.748 | Possibly Damaging | 0.017 | Benign | 4.07 | Benign | 0.00 | Affected | 0.2565 | 0.6908 | 0 | 1 | -0.4 | -0.94 | |||||||||||||||||||||||||||||||||||||
| c.2372A>C | K791T 2D  AIThe SynGAP1 missense variant K791T is listed in gnomAD (ID 6‑33442924‑A‑C) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). No tool predicts pathogenicity. The high‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign outcome, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also favors benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence overwhelmingly supports a benign classification, and this is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.966441 | Disordered | 0.478670 | Uncertain | 0.356 | 0.896 | 0.875 | 6-33442924-A-C | 2 | 1.24e-6 | -1.578 | Likely Benign | 0.415 | Ambiguous | Likely Benign | 0.033 | Likely Benign | -0.92 | Neutral | 0.032 | Benign | 0.017 | Benign | 4.17 | Benign | 0.16 | Tolerated | 3.64 | 6 | 0.2734 | 0.3340 | -1 | 0 | 3.2 | -27.07 | |||||||||||||||||||||||||||||||||
| c.2374G>A | E792K 2D  AIThe SynGAP1 missense variant E792K is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus also as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.974374 | Disordered | 0.452261 | Uncertain | 0.352 | 0.896 | 0.875 | -4.942 | Likely Benign | 0.753 | Likely Pathogenic | Likely Benign | 0.059 | Likely Benign | -2.47 | Neutral | 0.033 | Benign | 0.017 | Benign | 3.90 | Benign | 0.01 | Affected | 0.2646 | 0.7584 | 0 | 1 | -0.4 | -0.94 | ||||||||||||||||||||||||||||||||||||||
| c.2659C>T | P887S 2D  AIThe SynGAP1 missense variant P887S is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the predictions strongly suggest that P887S is most likely benign, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.716283 | Disordered | 0.602269 | Binding | 0.348 | 0.925 | 0.500 | -3.462 | Likely Benign | 0.065 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -1.26 | Neutral | 0.032 | Benign | 0.017 | Benign | 2.85 | Benign | 0.25 | Tolerated | 0.2770 | 0.3946 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.281C>T | P94L 2D  AIThe SynGAP1 missense variant P94L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.570978 | Binding | 0.350 | 0.869 | 0.625 | -2.721 | Likely Benign | 0.111 | Likely Benign | Likely Benign | 0.074 | Likely Benign | -2.27 | Neutral | 0.198 | Benign | 0.017 | Benign | 4.13 | Benign | 0.00 | Affected | 0.2125 | 0.5862 | -3 | -3 | 5.4 | 16.04 | |||||||||||||||||||||||||||||||||||||||
| c.285C>A | H95Q 2D  AIThe SynGAP1 missense variant H95Q is reported in gnomAD (ID 6‑33425893‑C‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. In contrast, polyPhen‑2 HumDiv and SIFT predict a pathogenic impact. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates benign. No Foldetta stability analysis is available, so it does not influence the overall assessment. Overall, the preponderance of predictions indicates that H95Q is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.590542 | Binding | 0.335 | 0.875 | 0.625 | 6-33425893-C-A | 1 | 6.20e-7 | -3.355 | Likely Benign | 0.084 | Likely Benign | Likely Benign | 0.070 | Likely Benign | -0.97 | Neutral | 0.633 | Possibly Damaging | 0.017 | Benign | 4.21 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1551 | 0.3375 | 0 | 3 | -0.3 | -9.01 | ||||||||||||||||||||||||||||||||||
| c.285C>G | H95Q 2D  AIThe SynGAP1 missense variant H95Q is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also indicates a likely benign outcome. In contrast, polyPhen‑2 HumDiv and SIFT predict pathogenicity, but these two tools are in minority. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.590542 | Binding | 0.335 | 0.875 | 0.625 | -3.355 | Likely Benign | 0.084 | Likely Benign | Likely Benign | 0.070 | Likely Benign | -0.97 | Neutral | 0.633 | Possibly Damaging | 0.017 | Benign | 4.21 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1551 | 0.3375 | 0 | 3 | -0.3 | -9.01 | |||||||||||||||||||||||||||||||||||||
| c.3027G>C | E1009D 2D  AIThe SynGAP1 missense variant E1009D is reported in gnomAD (ID 6‑33443579‑G‑C) but has no ClinVar entry. All in‑silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized indicates benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly supports a benign effect, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.728858 | Disordered | 0.914552 | Binding | 0.325 | 0.885 | 0.500 | 6-33443579-G-C | 1 | 6.20e-7 | -2.958 | Likely Benign | 0.104 | Likely Benign | Likely Benign | 0.054 | Likely Benign | -0.37 | Neutral | 0.011 | Benign | 0.017 | Benign | 2.50 | Benign | 0.30 | Tolerated | 3.77 | 5 | 0.2077 | 0.4817 | 2 | 3 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||||
| c.3027G>T | E1009D 2D  AIThe SynGAP1 missense variant E1009D is not reported in ClinVar and is absent from gnomAD. All available in‑silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta results are not available, so they do not influence the assessment. Overall, the variant is most likely benign, and this conclusion is consistent with the lack of ClinVar evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.728858 | Disordered | 0.914552 | Binding | 0.325 | 0.885 | 0.500 | -2.958 | Likely Benign | 0.104 | Likely Benign | Likely Benign | 0.054 | Likely Benign | -0.37 | Neutral | 0.011 | Benign | 0.017 | Benign | 2.50 | Benign | 0.30 | Tolerated | 3.77 | 5 | 0.2077 | 0.4817 | 2 | 3 | 0.0 | -14.03 | |||||||||||||||||||||||||||||||||||||
| c.3095T>C | L1032P 2D  AIThe SynGAP1 missense variant L1032P has no ClinVar entry and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and the SGM‑Consensus score all classify the change as benign or likely benign. AlphaMissense‑Optimized also predicts a benign outcome, whereas SIFT uniquely flags the variant as pathogenic. The AlphaMissense‑Default assessment is uncertain, and no Foldetta stability data are available. High‑accuracy analyses reinforce the benign consensus: AlphaMissense‑Optimized reports a benign effect, and the SGM‑Consensus (derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates benign. Foldetta results are missing, so they do not influence the interpretation. Overall, the majority of computational evidence supports a benign classification, which is consistent with the absence of a ClinVar pathogenic annotation. Thus, the variant is most likely benign, and this conclusion does not contradict ClinVar status, as no ClinVar pathogenic claim exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.865454 | Disordered | 0.995318 | Binding | 0.365 | 0.735 | 0.500 | -2.560 | Likely Benign | 0.449 | Ambiguous | Likely Benign | 0.142 | Likely Benign | -0.71 | Neutral | 0.012 | Benign | 0.017 | Benign | 2.64 | Benign | 0.05 | Affected | 0.3024 | 0.2330 | -3 | -3 | -5.4 | -16.04 | |||||||||||||||||||||||||||||||||||||||
| c.3110T>G | I1037S 2D  AIThe SynGAP1 missense variant I1037S is not reported in ClinVar and has no entries in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), which classifies the variant as Likely Benign. Only AlphaMissense‑Default predicts a pathogenic outcome, while AlphaMissense‑Optimized is uncertain and Foldetta (a protein‑folding stability method combining FoldX‑MD and Rosetta) has no available result for this variant. Based on the overall consensus of the majority of tools, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.939629 | Disordered | 0.986140 | Binding | 0.309 | 0.774 | 0.625 | -2.247 | Likely Benign | 0.935 | Likely Pathogenic | Ambiguous | 0.120 | Likely Benign | 0.43 | Neutral | 0.032 | Benign | 0.017 | Benign | 2.83 | Benign | 0.27 | Tolerated | 0.2634 | 0.1110 | -1 | -2 | -5.3 | -26.08 | |||||||||||||||||||||||||||||||||||||||
| c.3134C>T | A1045V 2D  AIThe SynGAP1 missense variant A1045V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion is consistent with the lack of ClinVar evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.975609 | Disordered | 0.948874 | Binding | 0.352 | 0.882 | 0.750 | -4.229 | Likely Benign | 0.076 | Likely Benign | Likely Benign | 0.035 | Likely Benign | -0.35 | Neutral | 0.011 | Benign | 0.017 | Benign | 3.06 | Benign | 1.00 | Tolerated | 0.1487 | 0.5474 | 0 | 0 | 2.4 | 28.05 | |||||||||||||||||||||||||||||||||||||||
| c.3203T>C | L1068S 2D  AIThe SynGAP1 missense variant L1068S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign, while only SIFT predicts it as pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.947281 | Disordered | 0.981041 | Binding | 0.362 | 0.907 | 0.875 | -6.297 | Likely Benign | 0.211 | Likely Benign | Likely Benign | 0.175 | Likely Benign | -0.57 | Neutral | 0.032 | Benign | 0.017 | Benign | 2.59 | Benign | 0.00 | Affected | 0.2810 | 0.1853 | -3 | -2 | -4.6 | -26.08 | |||||||||||||||||||||||||||||||||||||||
| c.3287A>T | E1096V 2D  AIThe SynGAP1 missense variant E1096V is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include SGM‑Consensus, REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is Likely Benign. No Foldetta stability result is available. Overall, the majority of evidence indicates the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.926919 | Disordered | 0.976475 | Binding | 0.308 | 0.858 | 1.000 | -3.588 | Likely Benign | 0.650 | Likely Pathogenic | Likely Benign | 0.138 | Likely Benign | -1.06 | Neutral | 0.043 | Benign | 0.017 | Benign | 2.81 | Benign | 0.03 | Affected | 0.1082 | 0.7596 | -2 | -2 | 7.7 | -29.98 | |||||||||||||||||||||||||||||||||||||||
| c.3338G>A | G1113D 2D  AIThe SynGAP1 missense variant G1113D is listed in ClinVar with an uncertain significance (ClinVar ID 2766136.0) and is present in gnomAD (variant ID 6‑33443890‑G‑A). Functional prediction tools that agree on a benign outcome include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM—all of which classify the substitution as benign. AlphaMissense‑Optimized also predicts a benign effect, whereas AlphaMissense‑Default remains uncertain. No tool predicts a pathogenic effect. The high‑accuracy consensus methods reinforce this benign assessment: the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and AlphaMissense‑Optimized also indicates benign. Foldetta, a protein‑folding stability predictor combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the collective evidence strongly supports a benign impact, and this conclusion does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.905695 | Disordered | 0.900456 | Binding | 0.327 | 0.910 | 0.875 | Uncertain | 1 | 6-33443890-G-A | -4.638 | Likely Benign | 0.354 | Ambiguous | Likely Benign | 0.061 | Likely Benign | -0.72 | Neutral | 0.029 | Benign | 0.017 | Benign | 2.58 | Benign | 0.34 | Tolerated | 4.32 | 2 | 0.1872 | 0.2452 | -1 | 1 | -3.1 | 58.04 | ||||||||||||||||||||||||||||||||||
| c.3394T>A | S1132T 2D  AIThe SynGAP1 missense variant S1132T is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification—there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.845506 | Binding | 0.289 | 0.894 | 0.750 | -3.700 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.190 | Likely Benign | -0.71 | Neutral | 0.011 | Benign | 0.017 | Benign | 5.45 | Benign | 0.50 | Tolerated | 0.1552 | 0.6309 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3533A>T | Y1178F 2D  AIThe SynGAP1 missense variant Y1178F is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population data. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates a likely benign outcome. Foldetta results are unavailable. Based on the consensus of all available predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.541878 | Disordered | 0.568433 | Binding | 0.554 | 0.688 | 0.250 | -3.081 | Likely Benign | 0.131 | Likely Benign | Likely Benign | 0.162 | Likely Benign | -0.64 | Neutral | 0.012 | Benign | 0.017 | Benign | 5.44 | Benign | 0.24 | Tolerated | 0.1893 | 0.2758 | 7 | 3 | 4.1 | -16.00 | ||||||||||||||||||||||||||||||||||||||
| c.3547T>C | Y1183H 2D  AIThe SynGAP1 missense variant Y1183H is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM, all of which classify the substitution as benign. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. In contrast, AlphaMissense‑Default predicts a pathogenic effect, while AlphaMissense‑Optimized is uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, SGM‑Consensus as likely benign, and no Foldetta stability data are available. Overall, the preponderance of evidence points to a benign impact for Y1183H, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.566480 | Disordered | 0.527818 | Binding | 0.523 | 0.652 | 0.500 | -3.289 | Likely Benign | 0.925 | Likely Pathogenic | Ambiguous | 0.026 | Likely Benign | -0.70 | Neutral | 0.029 | Benign | 0.017 | Benign | 2.75 | Benign | 0.18 | Tolerated | 0.2227 | 0.0243 | 0 | 2 | -1.9 | -26.03 | ||||||||||||||||||||||||||||||||||||||
| c.3737A>G | K1246R 2D  AIThe SynGAP1 missense variant K1246R is reported in gnomAD (ID 6‑33446729‑A‑G) and has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as “Likely Benign.” High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, and the Foldetta stability analysis is unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.750527 | Disordered | 0.375382 | Uncertain | 0.887 | 0.564 | 0.625 | 6-33446729-A-G | 1 | 6.20e-7 | -2.444 | Likely Benign | 0.097 | Likely Benign | Likely Benign | 0.026 | Likely Benign | -0.86 | Neutral | 0.031 | Benign | 0.017 | Benign | 2.66 | Benign | 0.04 | Affected | 3.77 | 5 | 0.3790 | 0.0515 | 2 | 3 | -0.6 | 28.01 | |||||||||||||||||||||||||||||||||
| c.3836C>G | A1279G 2D  AIThe SynGAP1 missense variant A1279G is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate a benign effect, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also reports “Likely Benign.” No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, SGM‑Consensus reports likely benign, and Foldetta (a protein‑folding stability method combining FoldX‑MD and Rosetta outputs) has no available result for this variant. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.842060 | Disordered | 0.814139 | Binding | 0.485 | 0.724 | 0.750 | -3.469 | Likely Benign | 0.078 | Likely Benign | Likely Benign | 0.064 | Likely Benign | -0.97 | Neutral | 0.033 | Benign | 0.017 | Benign | 2.69 | Benign | 0.19 | Tolerated | 0.1884 | 0.3473 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3836C>T | A1279V 2D  AIThe SynGAP1 missense variant A1279V is reported in gnomAD (ID 6‑33447884‑C‑T) but has no ClinVar entry. Across a broad panel of in silico predictors, every tool examined—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—classifies the substitution as benign. No pathogenic predictions appear in the dataset, so the pathogenic group is empty. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized returns a benign score, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, has no available result for this variant. Taken together, the consensus of all available predictions is that A1279V is most likely benign, and this assessment does not contradict any ClinVar status, as none is assigned. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.842060 | Disordered | 0.814139 | Binding | 0.485 | 0.724 | 0.750 | 6-33447884-C-T | -4.892 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.049 | Likely Benign | -1.23 | Neutral | 0.017 | Benign | 0.017 | Benign | 2.68 | Benign | 0.07 | Tolerated | 3.77 | 5 | 0.0777 | 0.5005 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||||
| c.3838A>C | M1280L 2D  AIThe SynGAP1 missense variant M1280L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact for M1280L, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.882776 | Disordered | 0.822030 | Binding | 0.510 | 0.726 | 0.875 | -1.391 | Likely Benign | 0.092 | Likely Benign | Likely Benign | 0.204 | Likely Benign | -2.23 | Neutral | 0.052 | Benign | 0.017 | Benign | 2.40 | Pathogenic | 0.00 | Affected | 0.1113 | 0.3186 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3838A>T | M1280L 2D  AIThe SynGAP1 missense variant M1280L is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, AlphaMissense‑Default, AlphaMissense‑Optimized, and ESM1b. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable for this variant. Overall, the majority of evidence points to a benign impact. The predictions do not contradict any ClinVar status, as no ClinVar claim exists for this variant. Thus, based on current computational predictions, the M1280L variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.882776 | Disordered | 0.822030 | Binding | 0.510 | 0.726 | 0.875 | -1.391 | Likely Benign | 0.092 | Likely Benign | Likely Benign | 0.204 | Likely Benign | -2.23 | Neutral | 0.052 | Benign | 0.017 | Benign | 2.40 | Pathogenic | 0.00 | Affected | 0.1113 | 0.3186 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3860C>T | P1287L 2D  AIThe SynGAP1 missense variant P1287L is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33447908‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.813701 | Binding | 0.538 | 0.777 | 0.750 | Conflicting | 2 | 6-33447908-C-T | -2.800 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.061 | Likely Benign | -1.66 | Neutral | 0.021 | Benign | 0.017 | Benign | 2.76 | Benign | 0.02 | Affected | 3.77 | 5 | 0.1861 | 0.4727 | -3 | -3 | 5.4 | 16.04 | ||||||||||||||||||||||||||||||||||
| c.3967C>G | P1323A 2D  AIThe SynGAP1 missense variant P1323A is catalogued in gnomAD (ID 6‑33451841‑C‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign or tolerated outcomes, while the single pathogenic signal comes from SIFT. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign classification. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign; Foldetta results are not available. Overall, the consensus of available predictions points to a benign impact for P1323A, and this conclusion is not contradicted by ClinVar status, which is currently absent. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.901269 | Disordered | 0.907659 | Binding | 0.489 | 0.814 | 0.875 | 6-33451841-C-G | -4.802 | Likely Benign | 0.053 | Likely Benign | Likely Benign | 0.040 | Likely Benign | -0.73 | Neutral | 0.011 | Benign | 0.017 | Benign | 3.92 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3743 | 0.3976 | -1 | 1 | 3.4 | -26.04 | ||||||||||||||||||||||||||||||||||||
| c.3970C>G | P1324A 2D  AIThe SynGAP1 missense variant P1324A is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.891961 | Disordered | 0.899181 | Binding | 0.432 | 0.793 | 0.875 | -4.416 | Likely Benign | 0.056 | Likely Benign | Likely Benign | 0.030 | Likely Benign | -0.57 | Neutral | 0.011 | Benign | 0.017 | Benign | 4.30 | Benign | 0.00 | Affected | 0.3489 | 0.3776 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||||||
| c.43G>A | A15T 2D  AIThe SynGAP1 missense variant A15T is listed in ClinVar (ID 1925632.0) with an “Uncertain” clinical significance and is present in gnomAD (6‑33420307‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) as “Likely Benign”; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.436924 | Structured | 0.466055 | Uncertain | 0.330 | 0.912 | 0.375 | Uncertain | 1 | 6-33420307-G-A | 4 | 2.60e-6 | -3.720 | Likely Benign | 0.125 | Likely Benign | Likely Benign | 0.086 | Likely Benign | -0.08 | Neutral | 0.602 | Possibly Damaging | 0.017 | Benign | 4.16 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2053 | 0.7423 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||||
| c.908G>T | G303V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G303V is not reported in ClinVar and is absent from gnomAD. Consensus predictions from multiple in silico tools cluster around a benign effect: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign) all indicate a tolerated change. Pathogenic signals arise only from SIFT and FoldX, while Rosetta and Foldetta are inconclusive. High‑accuracy assessments reinforce the benign trend: AlphaMissense‑Optimized predicts benign, SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields Likely Benign, and Foldetta remains uncertain. Overall, the preponderance of evidence points to a benign effect, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.450668 | Structured | 0.271087 | Uncertain | 0.630 | 0.254 | 0.250 | -3.046 | Likely Benign | 0.198 | Likely Benign | Likely Benign | 3.06 | Destabilizing | 0.6 | -0.72 | Ambiguous | 1.17 | Ambiguous | 0.14 | Likely Benign | 0.071 | Likely Benign | -1.57 | Neutral | 0.011 | Benign | 0.017 | Benign | 3.96 | Benign | 0.01 | Affected | 0.1002 | 0.4577 | -1 | -3 | 4.6 | 42.08 | |||||||||||||||||||||||||||||
| c.1211C>T | A404V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 A404V missense variant is not reported in ClinVar (ClinVar ID None) but is present in gnomAD (ID 6‑33438116‑C‑T). Prediction tools that agree on a benign effect include REVEL, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are PROVEAN and ESM1b. Four tools (FoldX, Rosetta, AlphaMissense‑Default, Foldetta) give uncertain or inconclusive results. High‑accuracy methods give mixed evidence: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) predicts pathogenic, and Foldetta remains uncertain. Overall, the majority of predictions lean toward a benign impact, though a key consensus method suggests pathogenicity, leaving the variant’s effect unresolved. **The variant is most likely benign based on the prevailing predictions, and this assessment does not contradict ClinVar status, as no ClinVar classification exists.** Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.232838 | Structured | 0.415505 | Uncertain | 0.965 | 0.355 | 0.000 | 6-33438116-C-T | 1 | 6.20e-7 | -8.219 | Likely Pathogenic | 0.343 | Ambiguous | Likely Benign | 0.56 | Ambiguous | 0.3 | -1.55 | Ambiguous | -0.50 | Ambiguous | -0.05 | Likely Benign | 0.118 | Likely Benign | -2.96 | Deleterious | 0.345 | Benign | 0.018 | Benign | 4.20 | Benign | 0.47 | Tolerated | 3.38 | 28 | 0.1288 | 0.6842 | 0 | 0 | 2.4 | 28.05 | |||||||||||||||||||||||||
| c.1280A>G | H427R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant H427R is not reported in ClinVar (ClinVar ID None) but is present in gnomAD (ID 6‑33438185‑A‑G). Functional prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and SIFT, while premPS and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also resolves to benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts a benign stability change. No predictions or folding results are missing or inconclusive. Overall, the majority of evidence indicates the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.081712 | Structured | 0.394261 | Uncertain | 0.962 | 0.287 | 0.000 | 6-33438185-A-G | -3.259 | Likely Benign | 0.439 | Ambiguous | Likely Benign | -0.04 | Likely Benign | 0.1 | 0.48 | Likely Benign | 0.22 | Likely Benign | 0.72 | Ambiguous | 0.168 | Likely Benign | -2.61 | Deleterious | 0.174 | Benign | 0.018 | Benign | 3.51 | Benign | 0.03 | Affected | 3.38 | 25 | 0.2108 | 0.2121 | 0 | 2 | -1.3 | 19.05 | |||||||||||||||||||||||||||
| c.1933T>G | F645V 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant F645V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect are premPS, PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. The high‑accuracy assessments give an uncertain result from AlphaMissense‑Optimized, a pathogenic outcome from the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and an uncertain outcome from Foldetta (combining FoldX‑MD and Rosetta). No definitive folding‑stability change is reported. Overall, the majority of evidence points to a pathogenic impact for F645V, and this conclusion does not contradict any ClinVar annotation because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.046336 | Structured | 0.276445 | Uncertain | 0.921 | 0.325 | 0.000 | -12.175 | Likely Pathogenic | 0.810 | Likely Pathogenic | Ambiguous | 1.98 | Ambiguous | 0.1 | 1.70 | Ambiguous | 1.84 | Ambiguous | 1.02 | Destabilizing | 0.316 | Likely Benign | -4.41 | Deleterious | 0.036 | Benign | 0.018 | Benign | 3.40 | Benign | 0.02 | Affected | 0.2163 | 0.2919 | -1 | -1 | 1.4 | -48.04 | |||||||||||||||||||||||||||||
| c.2257G>A | A753T 2D  AIThe SynGAP1 missense variant A753T is catalogued in gnomAD (ID 6‑33441722‑G‑A) but has no ClinVar entry. Across the spectrum of in‑silico predictors, every tool listed—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently classifies the change as benign. No pathogenic predictions are reported. Grouping by agreement, all available tools fall into the benign category, with no tools indicating pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status remains unavailable. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.521092 | Disordered | 0.722781 | Binding | 0.381 | 0.873 | 0.625 | 6-33441722-G-A | 2 | 1.24e-6 | -4.298 | Likely Benign | 0.069 | Likely Benign | Likely Benign | 0.071 | Likely Benign | -0.42 | Neutral | 0.022 | Benign | 0.018 | Benign | 2.70 | Benign | 0.28 | Tolerated | 3.99 | 5 | 0.1672 | 0.6997 | 0 | 1 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||||||
| c.235A>G | N79D 2D  AIThe SynGAP1 missense variant N79D is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also as benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.618285 | Disordered | 0.457064 | Uncertain | 0.290 | 0.876 | 0.375 | -3.746 | Likely Benign | 0.281 | Likely Benign | Likely Benign | 0.021 | Likely Benign | -0.92 | Neutral | 0.371 | Benign | 0.018 | Benign | 4.19 | Benign | 0.00 | Affected | 0.1893 | 0.2535 | 2 | 1 | 0.0 | 0.98 | |||||||||||||||||||||||||||||||||||||||
| c.236A>C | N79T 2D  AIThe SynGAP1 missense variant N79T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Based on the preponderance of evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.618285 | Disordered | 0.457064 | Uncertain | 0.290 | 0.876 | 0.375 | -3.752 | Likely Benign | 0.102 | Likely Benign | Likely Benign | 0.022 | Likely Benign | -0.61 | Neutral | 0.371 | Benign | 0.018 | Benign | 4.21 | Benign | 0.00 | Affected | 0.1133 | 0.4890 | 0 | 0 | 2.8 | -13.00 | |||||||||||||||||||||||||||||||||||||||
| c.2851C>A | H951N 2D  AIThe SynGAP1 missense variant H951N is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the evidence strongly supports a benign classification, and this conclusion is consistent with the lack of a ClinVar entry, so there is no contradiction with existing clinical annotations. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988291 | Disordered | 0.901477 | Binding | 0.415 | 0.925 | 0.750 | -5.833 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.140 | Likely Benign | -0.41 | Neutral | 0.011 | Benign | 0.018 | Benign | 5.43 | Benign | 0.16 | Tolerated | 0.2590 | 0.3197 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.2852A>T | H951L 2D  AIThe SynGAP1 missense variant H951L is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta results are not available, so they do not influence the assessment. Overall, the variant is most likely benign, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988291 | Disordered | 0.901477 | Binding | 0.415 | 0.925 | 0.750 | -4.822 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.192 | Likely Benign | -0.99 | Neutral | 0.022 | Benign | 0.018 | Benign | 5.47 | Benign | 0.43 | Tolerated | 0.2224 | 0.4526 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.2893C>G | H965D 2D  AIThe SynGAP1 missense variant H965D is reported in gnomAD (6‑33443445‑C‑G) and has no ClinVar entry. Consensus from most in silico predictors—REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—classifies the change as benign, while only the ESM1b model flags it as pathogenic. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized returns a benign score, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this residue, so its status is unavailable. Overall, the preponderance of evidence indicates that H965D is most likely benign, and this assessment does not contradict any ClinVar classification because none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988505 | Disordered | 0.978700 | Binding | 0.342 | 0.882 | 0.750 | 6-33443445-C-G | 1 | 6.20e-7 | -9.827 | Likely Pathogenic | 0.192 | Likely Benign | Likely Benign | 0.147 | Likely Benign | -0.94 | Neutral | 0.007 | Benign | 0.018 | Benign | 4.09 | Benign | 0.62 | Tolerated | 3.77 | 5 | 0.2697 | 0.3066 | -1 | 1 | -0.3 | -22.05 | ||||||||||||||||||||||||||||||||||
| c.2894A>T | H965L 2D  AIThe SynGAP1 missense variant H965L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect. Benign predictors include REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; no tool in the dataset returned a pathogenic prediction. Consensus predictors such as SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) classify the variant as Likely Benign. High‑accuracy methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus is Likely Benign; Foldetta results are not available. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988505 | Disordered | 0.978700 | Binding | 0.342 | 0.882 | 0.750 | -6.708 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.128 | Likely Benign | -1.66 | Neutral | 0.033 | Benign | 0.018 | Benign | 4.06 | Benign | 1.00 | Tolerated | 0.1606 | 0.5338 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.2909A>C | E970A 2D  AIThe SynGAP1 missense variant E970A is not reported in ClinVar and is absent from gnomAD. All available in‑silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Based on the consensus of all predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.951925 | Disordered | 0.953422 | Binding | 0.342 | 0.902 | 0.750 | -1.704 | Likely Benign | 0.156 | Likely Benign | Likely Benign | 0.068 | Likely Benign | -0.89 | Neutral | 0.069 | Benign | 0.018 | Benign | 4.15 | Benign | 0.19 | Tolerated | 0.3679 | 0.6798 | 0 | -1 | 5.3 | -58.04 | |||||||||||||||||||||||||||||||||||||||
| c.3001C>A | L1001I 2D  AIThe SynGAP1 missense variant L1001I is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy consensus methods reinforce the benign assessment: AlphaMissense‑Optimized scores the variant as benign, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” No Foldetta stability prediction is available for this variant. Overall, the preponderance of evidence from both general and high‑accuracy tools points to a benign effect, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.958507 | Binding | 0.269 | 0.902 | 0.375 | -4.486 | Likely Benign | 0.076 | Likely Benign | Likely Benign | 0.034 | Likely Benign | -0.17 | Neutral | 0.022 | Benign | 0.018 | Benign | 2.69 | Benign | 0.00 | Affected | 0.0994 | 0.3264 | 2 | 2 | 0.7 | 0.00 | |||||||||||||||||||||||||||||||||||||||
| c.3091A>C | M1031L 2D  AIThe SynGAP1 missense variant M1031L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Overall, the variant is most likely benign, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.865454 | Disordered | 0.995959 | Binding | 0.340 | 0.736 | 0.500 | -2.213 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.056 | Likely Benign | -0.82 | Neutral | 0.044 | Benign | 0.018 | Benign | 2.84 | Benign | 0.33 | Tolerated | 0.1220 | 0.4050 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3091A>T | M1031L 2D  AIThe SynGAP1 missense variant M1031L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Overall, the variant is most likely benign, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.865454 | Disordered | 0.995959 | Binding | 0.340 | 0.736 | 0.500 | -2.213 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.056 | Likely Benign | -0.82 | Neutral | 0.044 | Benign | 0.018 | Benign | 2.84 | Benign | 0.33 | Tolerated | 0.1220 | 0.4050 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3129G>C | R1043S 2D  AIThe SynGAP1 missense variant R1043S is not reported in ClinVar and has no entry in gnomAD. Functional prediction tools cluster into two groups: benign predictions include PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized; pathogenic predictions come from REVEL and SIFT. AlphaMissense‑Default is uncertain, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized scores benign, SGM‑Consensus is likely benign, and Foldetta results are unavailable. Taken together, the majority of evidence points to a benign impact for R1043S. This conclusion is consistent with the absence of a ClinVar assertion, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.978672 | Disordered | 0.954069 | Binding | 0.299 | 0.853 | 0.625 | -3.223 | Likely Benign | 0.457 | Ambiguous | Likely Benign | 0.509 | Likely Pathogenic | -2.10 | Neutral | 0.036 | Benign | 0.018 | Benign | 5.42 | Benign | 0.00 | Affected | 3.77 | 5 | 0.2727 | 0.4628 | -1 | 0 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||
| c.3129G>T | R1043S 2D  AIThe SynGAP1 missense variant R1043S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443681‑G‑T). Prediction tools that agree on a benign effect include PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are REVEL and SIFT. AlphaMissense‑Default remains uncertain, and no Foldetta stability result is available. High‑accuracy assessments: AlphaMissense‑Optimized classifies the variant as benign; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome; Foldetta data are missing, so it does not contribute to the evaluation. Based on the collective predictions, the variant is most likely benign, which is consistent with its ClinVar “Uncertain” classification and does not contradict the available evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.978672 | Disordered | 0.954069 | Binding | 0.299 | 0.853 | 0.625 | Uncertain | 1 | 6-33443681-G-T | 2 | 1.24e-6 | -3.223 | Likely Benign | 0.457 | Ambiguous | Likely Benign | 0.509 | Likely Pathogenic | -2.10 | Neutral | 0.036 | Benign | 0.018 | Benign | 5.42 | Benign | 0.00 | Affected | 3.77 | 5 | 0.2727 | 0.4628 | -1 | 0 | 3.7 | -69.11 | ||||||||||||||||||||||||||||||||
| c.3235A>G | S1079G 2D  AIThe SynGAP1 missense variant S1079G is reported in gnomAD (variant ID 6‑33443787‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign status. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of prediction tools and the high‑accuracy methods points to a benign effect. This conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.983887 | Binding | 0.307 | 0.900 | 0.750 | 6-33443787-A-G | 6 | 3.76e-6 | -3.552 | Likely Benign | 0.177 | Likely Benign | Likely Benign | 0.065 | Likely Benign | -1.50 | Neutral | 0.036 | Benign | 0.018 | Benign | 3.87 | Benign | 0.00 | Affected | 3.77 | 5 | 0.2151 | 0.4623 | 0 | 1 | 0.4 | -30.03 | ||||||||||||||||||||||||||||||||||
| c.35G>A | S12N 2D  AIThe SynGAP1 missense variant S12N is listed in ClinVar with no submitted interpretation and is present in gnomAD (variant ID 6‑33420299‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” No Foldetta stability result is available. Overall, the majority of evidence points to a benign effect, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.480142 | Structured | 0.490599 | Uncertain | 0.355 | 0.916 | 0.500 | 6-33420299-G-A | -4.946 | Likely Benign | 0.185 | Likely Benign | Likely Benign | 0.069 | Likely Benign | -0.41 | Neutral | 0.208 | Benign | 0.018 | Benign | 4.10 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1405 | 0.5004 | 1 | 1 | -2.7 | 27.03 | ||||||||||||||||||||||||||||||||||||
| c.3607C>A | H1203N 2D  AIThe SynGAP1 missense variant H1203N is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a neutral effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this view: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is likely benign, and Foldetta results are unavailable. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.618285 | Disordered | 0.527023 | Binding | 0.892 | 0.589 | 0.250 | -4.278 | Likely Benign | 0.109 | Likely Benign | Likely Benign | 0.181 | Likely Benign | -1.07 | Neutral | 0.002 | Benign | 0.018 | Benign | 5.61 | Benign | 0.31 | Tolerated | 0.1155 | 0.0914 | 2 | 1 | -0.3 | -23.04 | ||||||||||||||||||||||||||||||||||||||
| c.3726G>C | E1242D 2D  AIThe SynGAP1 missense variant E1242D is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this is consistent with the lack of ClinVar annotation—there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.557691 | Disordered | 0.456349 | Uncertain | 0.870 | 0.549 | 0.500 | -2.582 | Likely Benign | 0.158 | Likely Benign | Likely Benign | 0.031 | Likely Benign | -1.96 | Neutral | 0.020 | Benign | 0.018 | Benign | 2.37 | Pathogenic | 0.00 | Affected | 0.1594 | 0.2675 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||||||||
| c.3726G>T | E1242D 2D  AIThe SynGAP1 missense variant E1242D is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.557691 | Disordered | 0.456349 | Uncertain | 0.870 | 0.549 | 0.500 | -2.582 | Likely Benign | 0.158 | Likely Benign | Likely Benign | 0.032 | Likely Benign | -1.96 | Neutral | 0.020 | Benign | 0.018 | Benign | 2.37 | Pathogenic | 0.00 | Affected | 0.1594 | 0.2675 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||||||||
| c.3845A>C | E1282A 2D  AIThe SynGAP1 missense variant E1282A is not reported in ClinVar and is absent from gnomAD. Across the available in‑silico predictors, every tool examined—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently classifies the substitution as benign. No pathogenic predictions are present. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts a benign effect, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, was not available for this variant. Overall, the computational evidence overwhelmingly supports a benign impact, and this conclusion does not contradict any ClinVar status, as none exists for E1282A. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.817364 | Binding | 0.465 | 0.725 | 0.875 | -1.980 | Likely Benign | 0.097 | Likely Benign | Likely Benign | 0.118 | Likely Benign | 0.52 | Neutral | 0.026 | Benign | 0.018 | Benign | 2.94 | Benign | 0.38 | Tolerated | 0.3411 | 0.5358 | 0 | -1 | 5.3 | -58.04 | |||||||||||||||||||||||||||||||||||||||
| c.3892C>G | Q1298E 2D  AIThe SynGAP1 missense variant Q1298E is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this conclusion does not contradict any ClinVar annotation (none present). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.895297 | Binding | 0.410 | 0.821 | 0.750 | -3.530 | Likely Benign | 0.156 | Likely Benign | Likely Benign | 0.098 | Likely Benign | -1.14 | Neutral | 0.026 | Benign | 0.018 | Benign | 2.83 | Benign | 0.06 | Tolerated | 0.1235 | 0.1330 | 2 | 2 | 0.0 | 0.98 | |||||||||||||||||||||||||||||||||||||||
| c.53A>T | Y18F 2D  AIThe SynGAP1 missense variant Y18F is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.483068 | Structured | 0.446314 | Uncertain | 0.345 | 0.908 | 0.375 | -2.371 | Likely Benign | 0.160 | Likely Benign | Likely Benign | 0.059 | Likely Benign | -0.19 | Neutral | 0.115 | Benign | 0.018 | Benign | 4.18 | Benign | 0.00 | Affected | 0.2675 | 0.3322 | 7 | 3 | 4.1 | -16.00 | |||||||||||||||||||||||||||||||||||||||
| c.679G>A | G227R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G227R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. In contrast, the majority of other in silico predictors classify the variant as pathogenic: SGM‑Consensus, REVEL, FoldX, Rosetta, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized (premPS is inconclusive). High‑accuracy assessments further support a deleterious impact: AlphaMissense‑Optimized predicts pathogenic, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates likely pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) reports a pathogenic effect. Overall, the preponderance of evidence points to a pathogenic classification for G227R, with no conflict from ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.106997 | Structured | 0.329995 | Uncertain | 0.800 | 0.329 | 0.250 | -9.776 | Likely Pathogenic | 0.994 | Likely Pathogenic | Likely Pathogenic | 2.31 | Destabilizing | 0.3 | 5.29 | Destabilizing | 3.80 | Destabilizing | 0.90 | Ambiguous | 0.765 | Likely Pathogenic | -6.49 | Deleterious | 0.020 | Benign | 0.018 | Benign | 6.02 | Benign | 0.01 | Affected | 3.43 | 12 | 0.0997 | 0.4195 | -2 | -3 | -4.1 | 99.14 | |||||||||||||||||||||||||||
| c.679G>C | G227R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G227R is not reported in ClinVar (no ClinVar ID) but is present in gnomAD (ID 6‑33435530‑G‑C). Prediction tools that agree on a benign effect include polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect comprise REVEL, FoldX, Rosetta, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and Foldetta. The premPS score is uncertain and does not influence the consensus. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, the SGM‑Consensus as pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) as pathogenic. Based on the overwhelming agreement among high‑confidence predictors, the variant is most likely pathogenic; this conclusion is not contradicted by ClinVar, which contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.106997 | Structured | 0.329995 | Uncertain | 0.800 | 0.329 | 0.250 | 6-33435530-G-C | 1 | 6.20e-7 | -9.776 | Likely Pathogenic | 0.994 | Likely Pathogenic | Likely Pathogenic | 2.31 | Destabilizing | 0.3 | 5.29 | Destabilizing | 3.80 | Destabilizing | 0.90 | Ambiguous | 0.765 | Likely Pathogenic | -6.49 | Deleterious | 0.020 | Benign | 0.018 | Benign | 6.02 | Benign | 0.01 | Affected | 3.43 | 12 | 0.0997 | 0.4195 | -2 | -3 | -4.1 | 99.14 | ||||||||||||||||||||||||
| c.7A>G | R3G 2D  AIThe SynGAP1 missense variant R3G is reported in gnomAD (ID 6‑33420271‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it as pathogenic, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also likely benign; Foldetta results are unavailable. Taken together, the preponderance of evidence points to a benign impact. This conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.538167 | Disordered | 0.550331 | Binding | 0.358 | 0.920 | 0.875 | 6-33420271-A-G | -3.093 | Likely Benign | 0.160 | Likely Benign | Likely Benign | 0.099 | Likely Benign | -0.20 | Neutral | 0.115 | Benign | 0.018 | Benign | 3.99 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3559 | 0.4114 | -2 | -3 | 4.1 | -99.14 | ||||||||||||||||||||||||||||||||||||
| c.867G>A | M289I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M289I is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while FoldX and AlphaMissense‑Default are uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign prediction; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. Overall, the preponderance of evidence indicates that M289I is most likely benign, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.127496 | Structured | 0.403499 | Uncertain | 0.886 | 0.276 | 0.000 | -6.010 | Likely Benign | 0.505 | Ambiguous | Likely Benign | 0.62 | Ambiguous | 0.1 | -0.23 | Likely Benign | 0.20 | Likely Benign | 0.36 | Likely Benign | 0.040 | Likely Benign | -0.72 | Neutral | 0.005 | Benign | 0.018 | Benign | 1.78 | Pathogenic | 0.31 | Tolerated | 0.1001 | 0.2758 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.867G>C | M289I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M289I is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while FoldX and AlphaMissense‑Default are uncertain. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign verdict; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. Overall, the preponderance of evidence indicates that M289I is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.127496 | Structured | 0.403499 | Uncertain | 0.886 | 0.276 | 0.000 | -6.010 | Likely Benign | 0.505 | Ambiguous | Likely Benign | 0.62 | Ambiguous | 0.1 | -0.23 | Likely Benign | 0.20 | Likely Benign | 0.36 | Likely Benign | 0.040 | Likely Benign | -0.72 | Neutral | 0.005 | Benign | 0.018 | Benign | 1.78 | Pathogenic | 0.31 | Tolerated | 0.1001 | 0.2758 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.867G>T | M289I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M289I is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while FoldX and AlphaMissense‑Default are uncertain. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign verdict; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. Overall, the preponderance of evidence indicates that M289I is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.127496 | Structured | 0.403499 | Uncertain | 0.886 | 0.276 | 0.000 | -6.010 | Likely Benign | 0.505 | Ambiguous | Likely Benign | 0.62 | Ambiguous | 0.1 | -0.23 | Likely Benign | 0.20 | Likely Benign | 0.36 | Likely Benign | 0.040 | Likely Benign | -0.72 | Neutral | 0.005 | Benign | 0.018 | Benign | 1.78 | Pathogenic | 0.31 | Tolerated | 0.1001 | 0.2758 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.8G>C | R3T 2D  AIThe SynGAP1 missense variant R3T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.538167 | Disordered | 0.550331 | Binding | 0.358 | 0.920 | 0.875 | -3.693 | Likely Benign | 0.285 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -0.55 | Neutral | 0.208 | Benign | 0.018 | Benign | 4.01 | Benign | 0.00 | Affected | 0.1809 | 0.5145 | -1 | -1 | 3.8 | -55.08 | |||||||||||||||||||||||||||||||||||||||
| c.1909T>A | S637T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S637T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Functional prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive result is from Rosetta, which is treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.076542 | Structured | 0.083482 | Uncertain | 0.920 | 0.253 | 0.000 | -4.116 | Likely Benign | 0.092 | Likely Benign | Likely Benign | 0.36 | Likely Benign | 0.1 | -0.91 | Ambiguous | -0.28 | Likely Benign | -0.39 | Likely Benign | 0.067 | Likely Benign | -0.19 | Neutral | 0.086 | Benign | 0.019 | Benign | 3.45 | Benign | 0.19 | Tolerated | 0.1675 | 0.4572 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.1933T>A | F645I 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant F645I is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show mixed results: benign predictions come from REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM, while pathogenic predictions are given by premPS, PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. Four tools (FoldX, Rosetta, Foldetta, AlphaMissense‑Optimized) report uncertain or inconclusive outcomes. High‑accuracy assessments indicate that AlphaMissense‑Optimized is uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is pathogenic, and Foldetta is uncertain. Overall, the majority of evaluated predictors (five pathogenic vs four benign) lean toward a pathogenic effect. Therefore, the variant is most likely pathogenic, and this assessment does not contradict ClinVar status, which currently has no entry for F645I. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.046336 | Structured | 0.276445 | Uncertain | 0.921 | 0.325 | 0.000 | -13.055 | Likely Pathogenic | 0.878 | Likely Pathogenic | Ambiguous | 1.12 | Ambiguous | 0.2 | 1.68 | Ambiguous | 1.40 | Ambiguous | 1.01 | Destabilizing | 0.299 | Likely Benign | -4.24 | Deleterious | 0.190 | Benign | 0.019 | Benign | 3.44 | Benign | 0.04 | Affected | 0.1920 | 0.2891 | 1 | 0 | 1.7 | -34.02 | |||||||||||||||||||||||||||||
| c.220A>C | S74R 2D  AIThe SynGAP1 missense variant S74R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, and the SGM‑Consensus (majority of the high‑accuracy tools) also indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign effect, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.450156 | Uncertain | 0.294 | 0.831 | 0.500 | -3.271 | Likely Benign | 0.418 | Ambiguous | Likely Benign | 0.065 | Likely Benign | -1.34 | Neutral | 0.361 | Benign | 0.019 | Benign | 4.08 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0943 | 0.3562 | -1 | 0 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||
| c.222C>A | S74R 2D  AIThe SynGAP1 missense variant S74R is catalogued in gnomAD (ID 6‑33425830‑C‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all report benign or likely benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments confirm the benign consensus: AlphaMissense‑Optimized is benign, and the SGM‑Consensus, derived from the majority of the high‑confidence predictors, is benign. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that S74R is most likely benign, and this assessment does not contradict any ClinVar status, as none is reported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.450156 | Uncertain | 0.294 | 0.831 | 0.500 | 6-33425830-C-A | 1 | 6.20e-7 | -3.271 | Likely Benign | 0.418 | Ambiguous | Likely Benign | 0.070 | Likely Benign | -1.34 | Neutral | 0.361 | Benign | 0.019 | Benign | 4.08 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0943 | 0.3562 | -1 | 0 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||||
| c.222C>G | S74R 2D  AIThe SynGAP1 missense variant S74R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority of the high‑accuracy tools) is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.450156 | Uncertain | 0.294 | 0.831 | 0.500 | -3.271 | Likely Benign | 0.418 | Ambiguous | Likely Benign | 0.070 | Likely Benign | -1.34 | Neutral | 0.361 | Benign | 0.019 | Benign | 4.08 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0943 | 0.3562 | -1 | 0 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||
| c.2401G>A | G801S 2D  AIThe SynGAP1 missense variant G801S is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. All available in‑silico predictors classify the change as benign: SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this benign prediction: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is also Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant. Overall, the evidence strongly supports a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.874069 | Disordered | 0.636323 | Binding | 0.320 | 0.892 | 0.625 | Uncertain | 1 | -3.665 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.039 | Likely Benign | -0.41 | Neutral | 0.009 | Benign | 0.019 | Benign | 2.76 | Benign | 0.48 | Tolerated | 4.32 | 2 | 0.2325 | 0.4755 | 0 | 1 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||||
| c.2506A>G | S836G 2D  AIThe SynGAP1 missense variant S836G is listed in ClinVar (ID 537003.0) with an uncertain significance annotation and is observed in the gnomAD database (variant ID 6‑33443058‑A‑G). Consensus from multiple in silico predictors indicates a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool in the dataset predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is labeled Likely Benign. Foldetta, a protein‑folding stability predictor, did not return a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign classification, which does not conflict with the ClinVar uncertain designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.525368 | Disordered | 0.634582 | Binding | 0.269 | 0.859 | 0.250 | Uncertain | 1 | 6-33443058-A-G | 4 | 2.48e-6 | -4.749 | Likely Benign | 0.112 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -1.65 | Neutral | 0.006 | Benign | 0.019 | Benign | 2.54 | Benign | 0.39 | Tolerated | 3.77 | 5 | 0.2567 | 0.4089 | 1 | 0 | 0.4 | -30.03 | ||||||||||||||||||||||||||||||||
| c.2543G>A | G848D 2D  AIThe SynGAP1 missense variant G848D is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the evidence strongly supports a benign classification, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.724957 | Disordered | 0.563942 | Binding | 0.287 | 0.816 | 0.500 | -0.059 | Likely Benign | 0.318 | Likely Benign | Likely Benign | 0.121 | Likely Benign | 2.94 | Neutral | 0.008 | Benign | 0.019 | Benign | 2.95 | Benign | 0.81 | Tolerated | 0.1702 | 0.1392 | 1 | -1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||||||||||||
| c.2593G>T | A865S 2D  AIThe SynGAP1 missense variant A865S is reported as “Likely Benign” in SGM‑Consensus and has no ClinVar entry or gnomAD allele. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is also benign. Foldetta results are not available. Overall, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.521092 | Disordered | 0.626222 | Binding | 0.271 | 0.788 | 0.250 | -3.102 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.050 | Likely Benign | 0.20 | Neutral | 0.009 | Benign | 0.019 | Benign | 2.78 | Benign | 0.52 | Tolerated | 0.2314 | 0.4876 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.2594C>G | A865G 2D  AIThe SynGAP1 missense variant A865G is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Based on the collective predictions, the variant is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.521092 | Disordered | 0.626222 | Binding | 0.271 | 0.788 | 0.250 | -3.412 | Likely Benign | 0.107 | Likely Benign | Likely Benign | 0.027 | Likely Benign | -0.74 | Neutral | 0.009 | Benign | 0.019 | Benign | 2.69 | Benign | 0.51 | Tolerated | 0.2344 | 0.4683 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.277C>G | R93G 2D  AIThe SynGAP1 missense variant R93G is listed in ClinVar (ID 2504251.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized scores the variant as benign, and the SGM‑Consensus (derived from the same set of predictors) labels it “Likely Benign”; Foldetta results are unavailable. Overall, the preponderance of evidence indicates that R93G is most likely benign, which does not contradict the current ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.549151 | Binding | 0.290 | 0.874 | 0.625 | Uncertain | 1 | -2.674 | Likely Benign | 0.400 | Ambiguous | Likely Benign | 0.093 | Likely Benign | -1.69 | Neutral | 0.103 | Benign | 0.019 | Benign | 3.99 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3809 | 0.3814 | -2 | -3 | 4.1 | -99.14 | |||||||||||||||||||||||||||||||||||
| c.278G>T | R93L 2D  AIThe SynGAP1 R93L missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, SGM‑Consensus indicates Likely Benign, and Foldetta results are unavailable. Overall, the preponderance of evidence points to the variant being most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.549151 | Binding | 0.290 | 0.874 | 0.625 | -2.850 | Likely Benign | 0.425 | Ambiguous | Likely Benign | 0.064 | Likely Benign | -1.72 | Neutral | 0.103 | Benign | 0.019 | Benign | 4.00 | Benign | 0.00 | Affected | 0.2197 | 0.4861 | -3 | -2 | 8.3 | -43.03 | |||||||||||||||||||||||||||||||||||||||
| c.3835G>T | A1279S 2D  AIThe SynGAP1 missense variant A1279S is catalogued in gnomAD (ID 6‑33447883‑G‑T) but has no ClinVar entry. Across the spectrum of in‑silico predictors, every tool listed—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently scores the substitution as benign. No pathogenic predictions are reported. Grouping by agreement, the benign‑predicting tools comprise the entire set, while the pathogenic group is empty. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar classification (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.842060 | Disordered | 0.814139 | Binding | 0.485 | 0.724 | 0.750 | 6-33447883-G-T | -4.281 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.134 | Likely Benign | 0.37 | Neutral | 0.019 | Benign | 0.019 | Benign | 2.74 | Benign | 0.16 | Tolerated | 3.77 | 5 | 0.2166 | 0.4583 | 1 | 1 | -2.6 | 16.00 | ||||||||||||||||||||||||||||||||||||
| c.265C>G | P89A 2D  AIThe SynGAP1 missense variant P89A is listed in ClinVar (ID 1031674) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain, and Foldetta results are unavailable. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign outcome. High‑accuracy assessments therefore indicate a benign or uncertain impact: AlphaMissense‑Optimized is inconclusive, SGM‑Consensus is likely benign, and Foldetta data are missing. Overall, the balance of evidence points to the variant being most likely benign, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.545797 | Binding | 0.316 | 0.865 | 0.500 | Conflicting | 3 | -5.778 | Likely Benign | 0.920 | Likely Pathogenic | Ambiguous | 0.095 | Likely Benign | -2.47 | Neutral | 0.225 | Benign | 0.020 | Benign | 3.77 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3407 | 0.3768 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||
| c.265C>T | P89S 2D  AIThe SynGAP1 missense variant P89S (ClinVar ID 4801335) is listed as “Uncertain” in ClinVar and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are AlphaMissense‑Default, AlphaMissense‑Optimized, and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus (majority vote) remains likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of predictions (six benign vs. three pathogenic) and the SGM‑Consensus support a benign interpretation, which does not contradict the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.545797 | Binding | 0.316 | 0.865 | 0.500 | Uncertain | 1 | -5.198 | Likely Benign | 0.979 | Likely Pathogenic | Likely Pathogenic | 0.099 | Likely Benign | -2.40 | Neutral | 0.225 | Benign | 0.020 | Benign | 3.76 | Benign | 0.00 | Affected | 0.3459 | 0.4191 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||
| c.3828C>A | D1276E 2D  AIThe SynGAP1 missense variant D1276E is catalogued in gnomAD (ID 6‑33447876‑C‑A) but has no ClinVar submission. Functional prediction tools show a split assessment: benign calls come from REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, and AlphaMissense‑Optimized, whereas pathogenic calls arise from PROVEAN, SIFT, and FATHMM; AlphaMissense‑Default remains uncertain. A high‑accuracy consensus (SGM) that aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN yields a pathogenic verdict. AlphaMissense‑Optimized alone predicts benign, and no Foldetta stability data are available. Overall, the majority of individual predictors favor a benign effect, but the SGM consensus contradicts this by labeling the variant pathogenic. Because ClinVar contains no classification, there is no external evidence to resolve the discrepancy. Thus, based on the current computational evidence, the variant is most likely benign, though the SGM consensus suggests a possible pathogenic interpretation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.666105 | Disordered | 0.802156 | Binding | 0.636 | 0.705 | 0.625 | 6-33447876-C-A | -0.388 | Likely Benign | 0.416 | Ambiguous | Likely Benign | 0.091 | Likely Benign | -2.64 | Deleterious | 0.027 | Benign | 0.020 | Benign | 1.26 | Pathogenic | 0.00 | Affected | 3.77 | 5 | 0.1027 | 0.5365 | 2 | 3 | 0.0 | 14.03 | |||||||||||||||||||||||||||||||||||||
| c.3828C>G | D1276E 2D  AIThe SynGAP1 missense variant D1276E is not reported in ClinVar and is absent from gnomAD. Consensus from routine in silico predictors shows a split: benign calls from REVEL, polyPhen‑2 (HumDiv and HumVar), and ESM1b, versus pathogenic calls from PROVEAN, SIFT, and FATHMM; AlphaMissense‑Default remains uncertain. High‑accuracy assessments give a benign result from AlphaMissense‑Optimized, but the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—leans pathogenic. Foldetta, a protein‑folding stability method, has no available output for this residue. Overall, the balance of evidence tilts toward a benign interpretation, and this conclusion is not in conflict with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.666105 | Disordered | 0.802156 | Binding | 0.636 | 0.705 | 0.625 | -0.388 | Likely Benign | 0.416 | Ambiguous | Likely Benign | 0.092 | Likely Benign | -2.64 | Deleterious | 0.027 | Benign | 0.020 | Benign | 1.26 | Pathogenic | 0.00 | Affected | 3.77 | 5 | 0.1027 | 0.5365 | 2 | 3 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||||||||
| c.394T>A | F132I 2D  AISynGAP1 missense variant F132I is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (two pathogenic, two benign) and Foldetta results are unavailable. Because the majority of consensus tools predict benign and no ClinVar evidence contradicts this, the variant is most likely benign, although the pathogenic signal from AlphaMissense‑Optimized and the inconclusive SGM Consensus leave some uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.450668 | Structured | 0.727897 | Binding | 0.345 | 0.892 | 0.250 | -6.245 | Likely Benign | 0.974 | Likely Pathogenic | Likely Pathogenic | 0.211 | Likely Benign | -3.23 | Deleterious | 0.084 | Benign | 0.020 | Benign | 3.36 | Benign | 0.00 | Affected | 0.2306 | 0.1698 | 1 | 0 | 1.7 | -34.02 | ||||||||||||||||||||||||||||||||||||||||
| c.538T>A | S180T 2D  AIThe SynGAP1 missense variant S180T is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, ESM1b, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive (2 pathogenic vs. 2 benign). Foldetta results are unavailable. Overall, the majority of predictions (six benign vs. three pathogenic) support a benign classification. This consensus does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Thus, the variant is most likely benign based on current predictive evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.509769 | Disordered | 0.442877 | Uncertain | 0.320 | 0.616 | 0.500 | -9.851 | Likely Pathogenic | 0.669 | Likely Pathogenic | Likely Benign | 0.114 | Likely Benign | -2.07 | Neutral | 0.057 | Benign | 0.020 | Benign | 3.86 | Benign | 0.02 | Affected | 0.1238 | 0.5171 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.5G>T | S2I 2D  AIThe SynGAP1 missense variant S2I is catalogued in gnomAD (6‑33420269‑G‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all report benign or likely benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments confirm the benign consensus: AlphaMissense‑Optimized is benign, and the SGM‑Consensus, derived from the majority of the high‑confidence predictors, is benign. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that S2I is most likely benign, and this assessment does not contradict any ClinVar status, as none is reported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.543646 | Binding | 0.382 | 0.922 | 0.750 | 6-33420269-G-T | -4.947 | Likely Benign | 0.439 | Ambiguous | Likely Benign | 0.031 | Likely Benign | -0.59 | Neutral | 0.212 | Benign | 0.020 | Benign | 4.04 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1021 | 0.5980 | -2 | -1 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||||||||
| c.703T>A | S235T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S235T is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome, while FoldX, Rosetta, and AlphaMissense‑Default are uncertain. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign majority; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. No prediction or folding result is missing or inconclusive. Based on the overall consensus of the available tools, the variant is most likely benign, and this conclusion does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.250310 | Structured | 0.319150 | Uncertain | 0.743 | 0.331 | 0.000 | -9.422 | Likely Pathogenic | 0.451 | Ambiguous | Likely Benign | 0.80 | Ambiguous | 0.0 | -0.54 | Ambiguous | 0.13 | Likely Benign | 0.21 | Likely Benign | 0.453 | Likely Benign | -2.02 | Neutral | 0.057 | Benign | 0.020 | Benign | 5.75 | Benign | 0.13 | Tolerated | 0.1674 | 0.6337 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||
| c.106C>A | H36N 2D  AIThe SynGAP1 missense variant H36N is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact. The variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -3.614 | Likely Benign | 0.085 | Likely Benign | Likely Benign | 0.027 | Likely Benign | -0.83 | Neutral | 0.019 | Benign | 0.021 | Benign | 4.21 | Benign | 0.00 | Affected | 0.2353 | 0.4258 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.1320T>A | N440K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N440K is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Tools that predict a pathogenic effect are ESM1b and AlphaMissense‑Default. The remaining methods—FoldX, Rosetta, Foldetta, and AlphaMissense‑Optimized—yield uncertain or inconclusive results and are treated as unavailable. High‑accuracy assessments are likewise inconclusive: AlphaMissense‑Optimized is uncertain; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is a 2‑vs‑2 tie, and Foldetta is uncertain. Based on the overall distribution of predictions, the variant is most likely benign, and this assessment does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.191378 | Structured | 0.267204 | Uncertain | 0.929 | 0.245 | 0.000 | -10.114 | Likely Pathogenic | 0.895 | Likely Pathogenic | Ambiguous | 0.92 | Ambiguous | 0.1 | 1.04 | Ambiguous | 0.98 | Ambiguous | 0.40 | Likely Benign | 0.058 | Likely Benign | -1.97 | Neutral | 0.206 | Benign | 0.021 | Benign | 3.50 | Benign | 0.19 | Tolerated | 0.1660 | 0.2550 | 1 | 0 | -0.4 | 14.07 | ||||||||||||||||||||||||||||||
| c.1320T>G | N440K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N440K is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Tools that predict a pathogenic effect are ESM1b and AlphaMissense‑Default. The remaining methods—FoldX, Rosetta, Foldetta, and AlphaMissense‑Optimized—yield uncertain or inconclusive results and are treated as unavailable. High‑accuracy assessments are likewise inconclusive: AlphaMissense‑Optimized is uncertain; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is a 2‑vs‑2 tie, and Foldetta is uncertain. Based on the overall distribution of predictions, the variant is most likely benign, and this assessment does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.191378 | Structured | 0.267204 | Uncertain | 0.929 | 0.245 | 0.000 | -10.114 | Likely Pathogenic | 0.895 | Likely Pathogenic | Ambiguous | 0.92 | Ambiguous | 0.1 | 1.04 | Ambiguous | 0.98 | Ambiguous | 0.40 | Likely Benign | 0.057 | Likely Benign | -1.97 | Neutral | 0.206 | Benign | 0.021 | Benign | 3.50 | Benign | 0.19 | Tolerated | 0.1660 | 0.2550 | 1 | 0 | -0.4 | 14.07 | ||||||||||||||||||||||||||||||
| c.1852C>G | Q618E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant Q618E is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are ESM1b and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive due to a 2‑vs‑2 split. Overall, the preponderance of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation and gnomAD presence, and there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.175930 | Structured | 0.138725 | Uncertain | 0.904 | 0.240 | 0.000 | -12.535 | Likely Pathogenic | 0.162 | Likely Benign | Likely Benign | 0.46 | Likely Benign | 0.1 | 0.50 | Ambiguous | 0.48 | Likely Benign | 0.33 | Likely Benign | 0.233 | Likely Benign | -1.16 | Neutral | 0.046 | Benign | 0.021 | Benign | -1.17 | Pathogenic | 0.26 | Tolerated | 0.1003 | 0.1469 | 2 | 2 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||
| c.1936C>G | L646V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L646V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show a split assessment: benign predictions come from REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, AlphaMissense‑Default, AlphaMissense‑Optimized, and FATHMM, whereas pathogenic predictions arise from FoldX, Rosetta, premPS, SIFT, and ESM1b. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, resolves to “Likely Benign” (3 benign vs 1 pathogenic votes). High‑accuracy assessments further indicate AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as pathogenic. Overall, the majority of tools, including the high‑accuracy methods, lean toward a benign effect, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.048328 | Structured | 0.303751 | Uncertain | 0.941 | 0.344 | 0.000 | -8.995 | Likely Pathogenic | 0.332 | Likely Benign | Likely Benign | 3.82 | Destabilizing | 0.2 | 2.01 | Destabilizing | 2.92 | Destabilizing | 1.41 | Destabilizing | 0.125 | Likely Benign | -1.40 | Neutral | 0.040 | Benign | 0.021 | Benign | 3.21 | Benign | 0.03 | Affected | 0.2200 | 0.3767 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||
| c.2080G>T | A694S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A694S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree that the substitution is benign: REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The only inconclusive results come from Rosetta and premPS, which are listed as uncertain and do not influence the overall assessment. High‑accuracy predictors reinforce this view: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also reports benign. Consequently, the variant is most likely benign, and this prediction is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.127496 | Structured | 0.352199 | Uncertain | 0.938 | 0.269 | 0.000 | -0.326 | Likely Benign | 0.076 | Likely Benign | Likely Benign | 0.22 | Likely Benign | 0.1 | 0.59 | Ambiguous | 0.41 | Likely Benign | -0.53 | Ambiguous | 0.092 | Likely Benign | 0.70 | Neutral | 0.013 | Benign | 0.021 | Benign | 3.57 | Benign | 1.00 | Tolerated | 0.2725 | 0.4231 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.211G>A | D71N 2D  AIThe SynGAP1 D71N missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also reports Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.575842 | Disordered | 0.456046 | Uncertain | 0.350 | 0.799 | 0.375 | -4.279 | Likely Benign | 0.370 | Ambiguous | Likely Benign | 0.136 | Likely Benign | -1.25 | Neutral | 0.198 | Benign | 0.021 | Benign | 4.08 | Benign | 0.00 | Affected | 0.1331 | 0.6021 | 2 | 1 | 0.0 | -0.98 | |||||||||||||||||||||||||||||||||||||||
| c.2122C>A | L708I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L708I is not reported in ClinVar and is absent from gnomAD. All available in‑silico predictors classify the substitution as benign: SIFT, PolyPhen‑2 (HumDiv and HumVar), REVEL, PROVEAN, premPS, FoldX, Rosetta, AlphaMissense‑Default, AlphaMissense‑Optimized, ESM1b, and FATHMM. No tool predicts pathogenicity. High‑accuracy assessments concur: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also reports a benign effect. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.250310 | Structured | 0.365875 | Uncertain | 0.931 | 0.378 | 0.000 | -4.406 | Likely Benign | 0.116 | Likely Benign | Likely Benign | 0.42 | Likely Benign | 0.0 | -0.21 | Likely Benign | 0.11 | Likely Benign | -0.05 | Likely Benign | 0.108 | Likely Benign | 0.02 | Neutral | 0.022 | Benign | 0.021 | Benign | 3.34 | Benign | 0.20 | Tolerated | 0.0749 | 0.2729 | 2 | 2 | 0.7 | 0.00 | |||||||||||||||||||||||||||||
| c.212A>G | D71G 2D  AIThe SynGAP1 missense variant D71G is not reported in ClinVar (ClinVar ID None) but is present in gnomAD (ID 6‑33425820‑A‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this is consistent with the absence of a ClinVar pathogenic classification. Thus, the variant is most likely benign and does not contradict ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.575842 | Disordered | 0.456046 | Uncertain | 0.350 | 0.799 | 0.375 | 6-33425820-A-G | 1 | 6.20e-7 | -3.136 | Likely Benign | 0.439 | Ambiguous | Likely Benign | 0.111 | Likely Benign | -1.82 | Neutral | 0.171 | Benign | 0.021 | Benign | 4.03 | Benign | 0.00 | Affected | 4.32 | 1 | 0.3802 | 0.5704 | -1 | 1 | 3.1 | -58.04 | ||||||||||||||||||||||||||||||||||
| c.224A>C | E75A 2D  AIThe SynGAP1 missense variant E75A is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic call comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.443881 | Uncertain | 0.303 | 0.822 | 0.500 | -3.111 | Likely Benign | 0.194 | Likely Benign | Likely Benign | 0.055 | Likely Benign | -1.19 | Neutral | 0.345 | Benign | 0.021 | Benign | 4.05 | Benign | 0.00 | Affected | 0.4248 | 0.6413 | 0 | -1 | 5.3 | -58.04 | |||||||||||||||||||||||||||||||||||||||
| c.233G>T | R78L 2D  AIThe SynGAP1 missense variant R78L is listed in ClinVar (ID 3390541.0) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (which is a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as benign; the Foldetta protein‑folding stability analysis is unavailable for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the current ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.448183 | Uncertain | 0.304 | 0.866 | 0.500 | Uncertain | 1 | -3.389 | Likely Benign | 0.635 | Likely Pathogenic | Likely Benign | 0.062 | Likely Benign | -1.59 | Neutral | 0.385 | Benign | 0.021 | Benign | 3.84 | Benign | 0.00 | Affected | 0.1445 | 0.4276 | -3 | -2 | 8.3 | -43.03 | |||||||||||||||||||||||||||||||||||||
| c.346T>A | Y116N 2D  AIThe SynGAP1 missense variant Y116N is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.670235 | Binding | 0.381 | 0.878 | 0.625 | -1.706 | Likely Benign | 0.260 | Likely Benign | Likely Benign | 0.108 | Likely Benign | 0.21 | Neutral | 0.137 | Benign | 0.021 | Benign | 4.27 | Benign | 1.00 | Tolerated | 0.2592 | 0.0545 | -2 | -2 | -2.2 | -49.07 | |||||||||||||||||||||||||||||||||||||||
| c.385T>A | S129T 2D  AIThe SynGAP1 missense variant S129T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion is consistent with the lack of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.517562 | Disordered | 0.713635 | Binding | 0.311 | 0.880 | 0.625 | -4.296 | Likely Benign | 0.171 | Likely Benign | Likely Benign | 0.134 | Likely Benign | 0.12 | Neutral | 0.016 | Benign | 0.021 | Benign | 4.14 | Benign | 0.85 | Tolerated | 0.1251 | 0.5794 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.421A>G | I141V 2D  AIThe SynGAP1 I141V missense variant is catalogued in gnomAD (ID 6‑33432718‑A‑G) but has no ClinVar entry. Functional prediction tools cluster into two groups: benign (REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM) and pathogenic (SIFT, AlphaMissense‑Default). The high‑accuracy AlphaMissense‑Optimized score is uncertain, and the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, favors a benign outcome. Foldetta, a protein‑folding stability predictor that integrates FoldX‑MD and Rosetta outputs, has no reported result for this variant. Overall, the preponderance of evidence points to a benign effect, and this assessment aligns with the lack of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.465241 | Structured | 0.577021 | Binding | 0.367 | 0.877 | 0.500 | 6-33432718-A-G | 1 | 6.42e-7 | -4.030 | Likely Benign | 0.785 | Likely Pathogenic | Ambiguous | 0.125 | Likely Benign | -0.58 | Neutral | 0.016 | Benign | 0.021 | Benign | 3.74 | Benign | 0.03 | Affected | 3.61 | 5 | 0.1101 | 0.3464 | 3 | 4 | -0.3 | -14.03 | ||||||||||||||||||||||||||||||||||
| c.502C>G | H168D 2D  AIThe SynGAP1 missense variant H168D is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign consensus, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. This prediction is consistent with the lack of ClinVar annotation and does not contradict any existing clinical database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.433034 | Structured | 0.502450 | Binding | 0.402 | 0.678 | 0.125 | -8.519 | Likely Pathogenic | 0.481 | Ambiguous | Likely Benign | 0.131 | Likely Benign | -1.24 | Neutral | 0.016 | Benign | 0.021 | Benign | 4.24 | Benign | 0.08 | Tolerated | 0.2365 | 0.2017 | 1 | -1 | -0.3 | -22.05 | ||||||||||||||||||||||||||||||||||||||||
| c.503A>T | H168L 2D  AIThe SynGAP1 H168L missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while ESM1b is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.502450 | Binding | 0.402 | 0.678 | 0.125 | -7.936 | In-Between | 0.178 | Likely Benign | Likely Benign | 0.250 | Likely Benign | -2.36 | Neutral | 0.037 | Benign | 0.021 | Benign | 4.21 | Benign | 0.01 | Affected | 0.0752 | 0.5602 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.97C>A | Q33K 2D  AIThe SynGAP1 missense variant Q33K is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. This conclusion is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.436712 | Uncertain | 0.342 | 0.860 | 0.375 | -0.317 | Likely Benign | 0.217 | Likely Benign | Likely Benign | 0.026 | Likely Benign | -0.53 | Neutral | 0.019 | Benign | 0.021 | Benign | 4.24 | Benign | 0.00 | Affected | 0.2366 | 0.4514 | 1 | 1 | -0.4 | 0.04 | |||||||||||||||||||||||||||||||||||||||
| c.1048G>C | V350L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V350L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only FATHMM predicts a pathogenic outcome, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates a likely benign status. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, and Foldetta (combining FoldX‑MD and Rosetta stability outputs) as uncertain. No other tools provide conflicting evidence. Based on the preponderance of benign predictions and the lack of pathogenic support, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.179055 | Structured | 0.353227 | Uncertain | 0.931 | 0.371 | 0.000 | -5.941 | Likely Benign | 0.238 | Likely Benign | Likely Benign | -1.31 | Ambiguous | 0.1 | -0.34 | Likely Benign | -0.83 | Ambiguous | 0.36 | Likely Benign | 0.042 | Likely Benign | -1.63 | Neutral | 0.068 | Benign | 0.022 | Benign | 2.37 | Pathogenic | 0.27 | Tolerated | 0.1173 | 0.4958 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||
| c.1048G>T | V350L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V350L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only FATHMM predicts a pathogenic outcome, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates a likely benign status. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) is inconclusive. No other tools provide conflicting evidence. Based on the preponderance of predictions, V350L is most likely benign, and this conclusion does not contradict any ClinVar status because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.179055 | Structured | 0.353227 | Uncertain | 0.931 | 0.371 | 0.000 | -5.941 | Likely Benign | 0.238 | Likely Benign | Likely Benign | -1.31 | Ambiguous | 0.1 | -0.34 | Likely Benign | -0.83 | Ambiguous | 0.36 | Likely Benign | 0.042 | Likely Benign | -1.63 | Neutral | 0.068 | Benign | 0.022 | Benign | 2.37 | Pathogenic | 0.27 | Tolerated | 0.1173 | 0.4958 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||
| c.1450T>C | F484L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F484L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show a split opinion: benign predictions come from REVEL, polyPhen‑2 (HumDiv and HumVar), and FATHMM, while pathogenic predictions arise from SGM‑Consensus (Likely Pathogenic), premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The high‑accuracy consensus, SGM‑Consensus, is derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN (3 pathogenic vs. 1 benign) and therefore also indicates pathogenicity. AlphaMissense‑Optimized independently predicts pathogenicity. Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is inconclusive. Overall, seven tools predict pathogenicity versus four predicting benign, and the high‑accuracy consensus supports a pathogenic interpretation. Thus, the variant is most likely pathogenic, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.182256 | Structured | 0.403079 | Uncertain | 0.798 | 0.245 | 0.125 | -11.052 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 1.52 | Ambiguous | 0.0 | 1.32 | Ambiguous | 1.42 | Ambiguous | 1.20 | Destabilizing | 0.275 | Likely Benign | -5.64 | Deleterious | 0.054 | Benign | 0.022 | Benign | 3.20 | Benign | 0.04 | Affected | 0.1737 | 0.2956 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||
| c.1452C>A | F484L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F484L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show a split opinion: benign predictions come from REVEL, polyPhen‑2 (HumDiv and HumVar), and FATHMM, while pathogenic predictions arise from SGM‑Consensus (Likely Pathogenic), premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The high‑accuracy consensus, SGM‑Consensus, is derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN (3 pathogenic vs. 1 benign) and therefore also indicates pathogenicity. AlphaMissense‑Optimized independently predicts pathogenicity. Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is inconclusive. Overall, seven tools predict pathogenicity versus four predicting benign, and the high‑accuracy consensus supports a pathogenic interpretation. Thus, the variant is most likely pathogenic, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.182256 | Structured | 0.403079 | Uncertain | 0.798 | 0.245 | 0.125 | -11.052 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 1.52 | Ambiguous | 0.0 | 1.32 | Ambiguous | 1.42 | Ambiguous | 1.20 | Destabilizing | 0.214 | Likely Benign | -5.64 | Deleterious | 0.054 | Benign | 0.022 | Benign | 3.20 | Benign | 0.04 | Affected | 0.1737 | 0.2956 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||
| c.1452C>G | F484L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F484L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show a split opinion: benign predictions come from REVEL, polyPhen‑2 (HumDiv and HumVar), and FATHMM, while pathogenic predictions arise from SGM‑Consensus (Likely Pathogenic), premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments further support a deleterious effect: AlphaMissense‑Optimized predicts pathogenic, and the SGM‑Consensus (a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates likely pathogenic. Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, yields an uncertain result. Overall, seven tools predict pathogenicity versus four predicting benign, and the high‑accuracy methods reinforce the pathogenic signal. Thus, the variant is most likely pathogenic, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.182256 | Structured | 0.403079 | Uncertain | 0.798 | 0.245 | 0.125 | -11.052 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 1.52 | Ambiguous | 0.0 | 1.32 | Ambiguous | 1.42 | Ambiguous | 1.20 | Destabilizing | 0.214 | Likely Benign | -5.64 | Deleterious | 0.054 | Benign | 0.022 | Benign | 3.20 | Benign | 0.04 | Affected | 0.1737 | 0.2956 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||
| c.1585A>C | I529L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I529L is listed in gnomAD (variant ID 6‑33438828‑A‑C) but has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments further support benignity: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote) is benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is benign. No prediction or folding result is missing or inconclusive. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.318242 | Structured | 0.019545 | Uncertain | 0.901 | 0.403 | 0.000 | 6-33438828-A-C | 1 | 6.20e-7 | 0.920 | Likely Benign | 0.066 | Likely Benign | Likely Benign | -0.13 | Likely Benign | 0.0 | -0.11 | Likely Benign | -0.12 | Likely Benign | -0.21 | Likely Benign | 0.309 | Likely Benign | 0.00 | Neutral | 0.001 | Benign | 0.022 | Benign | -1.24 | Pathogenic | 0.48 | Tolerated | 3.37 | 35 | 0.0819 | 0.3789 | 2 | 2 | -0.7 | 0.00 | ||||||||||||||||||||||||
| c.1609G>C | A537P 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A537P is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are FATHMM and Rosetta; FoldX and Foldetta are inconclusive. The high‑accuracy consensus (SGM‑Consensus) derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN indicates a likely benign outcome, while AlphaMissense‑Optimized also predicts benign. Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is uncertain. Overall, the majority of evidence points to a benign impact for A537P, and this assessment does not contradict the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.116183 | Structured | 0.037313 | Uncertain | 0.928 | 0.362 | 0.000 | 0.404 | Likely Benign | 0.102 | Likely Benign | Likely Benign | -0.61 | Ambiguous | 0.6 | 2.43 | Destabilizing | 0.91 | Ambiguous | -0.18 | Likely Benign | 0.218 | Likely Benign | 0.67 | Neutral | 0.020 | Benign | 0.022 | Benign | -1.29 | Pathogenic | 0.35 | Tolerated | 0.2152 | 0.3807 | 1 | -1 | -3.4 | 26.04 | |||||||||||||||||||||||||||||
| c.1919C>T | T640I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T640I is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only PROVEAN predicts a pathogenic outcome. The remaining tools (FoldX, Rosetta, ESM1b, AlphaMissense‑Default, and Foldetta) give uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is unavailable due to a tie, and Foldetta also yields an uncertain stability change. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar status, which has no classification for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.066181 | Structured | 0.137043 | Uncertain | 0.893 | 0.284 | 0.000 | -7.256 | In-Between | 0.358 | Ambiguous | Likely Benign | 0.70 | Ambiguous | 0.1 | 0.66 | Ambiguous | 0.68 | Ambiguous | 0.28 | Likely Benign | 0.193 | Likely Benign | -2.75 | Deleterious | 0.322 | Benign | 0.022 | Benign | 3.46 | Benign | 0.12 | Tolerated | 0.1030 | 0.4793 | 0 | -1 | 5.2 | 12.05 | ||||||||||||||||||||||||||||||
| c.2221C>A | P741T 2D  AIThe SynGAP1 missense variant P741T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.493550 | Uncertain | 0.354 | 0.859 | 0.875 | -4.626 | Likely Benign | 0.061 | Likely Benign | Likely Benign | 0.077 | Likely Benign | -0.54 | Neutral | 0.010 | Benign | 0.022 | Benign | 2.86 | Benign | 0.05 | Affected | 0.1209 | 0.5080 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.2396C>G | P799R 2D  AIThe SynGAP1 missense variant P799R is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it to be pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a “Likely Benign” outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) likewise indicates benign. No Foldetta stability analysis is available, so it does not influence the assessment. Overall, the preponderance of evidence points to the variant being most likely benign, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.871313 | Disordered | 0.537892 | Binding | 0.400 | 0.894 | 0.750 | -5.100 | Likely Benign | 0.141 | Likely Benign | Likely Benign | 0.029 | Likely Benign | -0.49 | Neutral | 0.029 | Benign | 0.022 | Benign | 4.27 | Benign | 0.00 | Affected | 0.1425 | 0.2905 | 0 | -2 | -2.9 | 59.07 | ||||||||||||||||||||||||||||||||||||||
| c.2420A>T | Y807F 2D  AIThe SynGAP1 missense variant Y807F is listed in ClinVar (ID 1491782.0) with an “Uncertain” status and is not reported in gnomAD. All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates a likely benign effect. No tool in the set predicts pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (derived from the four high‑accuracy predictors) is benign. Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, has no available result for this variant. Overall, the computational evidence strongly supports a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.699094 | Disordered | 0.853760 | Binding | 0.336 | 0.901 | 0.500 | Uncertain | 1 | -3.667 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.057 | Likely Benign | 0.14 | Neutral | 0.012 | Benign | 0.022 | Benign | 2.92 | Benign | 0.98 | Tolerated | 3.77 | 5 | 0.2485 | 0.2500 | 7 | 3 | 4.1 | -16.00 | ||||||||||||||||||||||||||||||||||
| c.2426G>T | S809I 2D  AIThe SynGAP1 missense variant S809I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Consensus from standard in silico predictors shows a split: six tools (REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Optimized) predict a benign effect, while two (SIFT, AlphaMissense‑Default) predict pathogenicity; ESM1b is uncertain. High‑accuracy assessment further supports a benign outcome: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign classification, and Foldetta data are unavailable. Overall, the preponderance of evidence indicates the variant is most likely benign, and this assessment does not contradict the absence of a ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | SH3-binding motif | 0.626927 | Disordered | 0.853218 | Binding | 0.330 | 0.907 | 0.500 | -7.708 | In-Between | 0.632 | Likely Pathogenic | Likely Benign | 0.087 | Likely Benign | -1.93 | Neutral | 0.065 | Benign | 0.022 | Benign | 2.50 | Benign | 0.01 | Affected | 0.1039 | 0.5984 | -1 | -2 | 5.3 | 26.08 | |||||||||||||||||||||||||||||||||||||||
| c.24C>G | I8M 2D  AIThe SynGAP1 missense variant I8M is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus, SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign status. Foldetta, a protein‑folding stability method that combines FoldX‑MD and Rosetta outputs, was not available for this variant. Overall, the majority of evidence points to a benign effect, and this assessment does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.543080 | Binding | 0.341 | 0.916 | 0.625 | -3.957 | Likely Benign | 0.140 | Likely Benign | Likely Benign | 0.085 | Likely Benign | -0.07 | Neutral | 0.296 | Benign | 0.022 | Benign | 4.02 | Benign | 0.00 | Affected | 0.0626 | 0.3034 | 2 | 1 | -2.6 | 18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3005A>G | H1002R 2D  AIThe SynGAP1 missense variant H1002R is listed in gnomAD (ID 6‑33443557‑A‑G) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable. Taken together, the majority of evidence points to a benign impact, and this conclusion is not contradicted by any ClinVar classification (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.784345 | Disordered | 0.953758 | Binding | 0.285 | 0.900 | 0.500 | 6-33443557-A-G | 1 | 6.20e-7 | -3.624 | Likely Benign | 0.609 | Likely Pathogenic | Likely Benign | 0.082 | Likely Benign | -1.52 | Neutral | 0.012 | Benign | 0.022 | Benign | 2.76 | Benign | 0.25 | Tolerated | 4.32 | 4 | 0.2160 | 0.2978 | 0 | 2 | -1.3 | 19.05 | ||||||||||||||||||||||||||||||||||
| c.3041G>C | G1014A 2D  AIThe SynGAP1 missense variant G1014A is not reported in ClinVar and is absent from gnomAD. Prediction tools that assess sequence conservation and functional impact (REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized) all classify the variant as benign. No tool predicts pathogenicity. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and Foldetta results are unavailable. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.771762 | Disordered | 0.914808 | Binding | 0.293 | 0.835 | 0.625 | -3.520 | Likely Benign | 0.114 | Likely Benign | Likely Benign | 0.039 | Likely Benign | -1.06 | Neutral | 0.025 | Benign | 0.022 | Benign | 2.78 | Benign | 0.36 | Tolerated | 0.3677 | 0.4682 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3212G>C | G1071A 2D  AIThe SynGAP1 missense variant G1071A is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM Consensus—derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—also indicates likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign effect, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.983740 | Binding | 0.313 | 0.905 | 0.875 | -1.825 | Likely Benign | 0.229 | Likely Benign | Likely Benign | 0.070 | Likely Benign | -1.34 | Neutral | 0.025 | Benign | 0.022 | Benign | 4.12 | Benign | 0.05 | Affected | 0.3376 | 0.5084 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3212G>T | G1071V 2D  AIThe SynGAP1 missense variant G1071V is catalogued in gnomAD (ID 6‑33443764‑G‑T) but has no ClinVar entry. In silico prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign or likely benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign classification. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the consensus of the majority of tools, including the high‑accuracy methods, supports a benign interpretation. This prediction does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.983740 | Binding | 0.313 | 0.905 | 0.875 | 6-33443764-G-T | 3 | 1.87e-6 | -2.901 | Likely Benign | 0.300 | Likely Benign | Likely Benign | 0.110 | Likely Benign | -2.42 | Neutral | 0.057 | Benign | 0.022 | Benign | 4.14 | Benign | 0.01 | Affected | 3.77 | 5 | 0.1408 | 0.3634 | -3 | -1 | 4.6 | 42.08 | ||||||||||||||||||||||||||||||||||
| c.3347G>A | G1116E 2D  AIThe SynGAP1 missense variant G1116E is reported in gnomAD (variant ID 6-33443899‑G‑A) but has no ClinVar entry. Functional prediction tools uniformly favor a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign, while AlphaMissense‑Default remains uncertain. No tool predicts pathogenicity. The high‑accuracy consensus (SGM‑Consensus) also indicates a likely benign outcome, and AlphaMissense‑Optimized corroborates this. Foldetta, a protein‑folding stability predictor, was not available for this variant. Overall, the collective evidence strongly supports a benign impact, and this conclusion is not contradicted by any ClinVar classification (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.889439 | Disordered | 0.873279 | Binding | 0.320 | 0.909 | 0.750 | 6-33443899-G-A | -6.375 | Likely Benign | 0.375 | Ambiguous | Likely Benign | 0.345 | Likely Benign | -0.33 | Neutral | 0.043 | Benign | 0.022 | Benign | 4.06 | Benign | 0.07 | Tolerated | 4.32 | 2 | 0.1467 | 0.4069 | -2 | 0 | -3.1 | 72.06 | ||||||||||||||||||||||||||||||||||||
| c.3347G>C | G1116A 2D  AIThe SynGAP1 missense variant G1116A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.889439 | Disordered | 0.873279 | Binding | 0.320 | 0.909 | 0.750 | -5.753 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.288 | Likely Benign | -0.24 | Neutral | 0.010 | Benign | 0.022 | Benign | 4.08 | Benign | 0.24 | Tolerated | 0.3417 | 0.4944 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3400A>C | T1134P 2D  AIThe SynGAP1 missense variant T1134P is not reported in ClinVar and is absent from gnomAD. All evaluated in‑silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. The high‑accuracy consensus methods likewise support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta results are unavailable. Overall, the variant is most likely benign, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.813034 | Binding | 0.335 | 0.885 | 0.875 | -2.993 | Likely Benign | 0.098 | Likely Benign | Likely Benign | 0.164 | Likely Benign | -0.57 | Neutral | 0.013 | Benign | 0.022 | Benign | 5.41 | Benign | 0.07 | Tolerated | 0.2001 | 0.5165 | 0 | -1 | -0.9 | -3.99 | |||||||||||||||||||||||||||||||||||||||
| c.3445C>G | P1149A 2D  AIThe SynGAP1 missense variant P1149A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification, so there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.786938 | Binding | 0.424 | 0.837 | 0.625 | -3.179 | Likely Benign | 0.107 | Likely Benign | Likely Benign | 0.037 | Likely Benign | -0.72 | Neutral | 0.025 | Benign | 0.022 | Benign | 2.76 | Benign | 0.07 | Tolerated | 0.3403 | 0.4352 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||||||
| c.3787A>C | I1263L 2D  AIThe SynGAP1 missense variant I1263L is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (two benign, two pathogenic). Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Coiled-coil | 0.425610 | Structured | 0.740957 | Binding | 0.867 | 0.574 | 0.000 | -1.210 | Likely Benign | 0.699 | Likely Pathogenic | Likely Benign | 0.194 | Likely Benign | -1.66 | Neutral | 0.011 | Benign | 0.022 | Benign | 1.94 | Pathogenic | 0.00 | Affected | 0.0717 | 0.2890 | 2 | 2 | -0.7 | 0.00 | |||||||||||||||||||||||||||||||||||||||
| c.3904T>G | L1302V 2D  AIThe SynGAP1 missense variant L1302V is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, AlphaMissense‑Default, AlphaMissense‑Optimized, and ESM1b. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.784345 | Disordered | 0.889642 | Binding | 0.429 | 0.842 | 0.875 | -4.140 | Likely Benign | 0.082 | Likely Benign | Likely Benign | 0.214 | Likely Benign | -1.71 | Neutral | 0.004 | Benign | 0.022 | Benign | 1.58 | Pathogenic | 0.00 | Affected | 0.1603 | 0.2686 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.39C>G | I13M 2D  AIThe SynGAP1 missense variant I13M is reported in gnomAD (ID 6‑33420303‑C‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus itself is likely benign. Foldetta results are not available, so they do not influence the assessment. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar status (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.486429 | Structured | 0.482657 | Uncertain | 0.318 | 0.916 | 0.375 | 6-33420303-C-G | 1 | 6.49e-7 | -4.097 | Likely Benign | 0.170 | Likely Benign | Likely Benign | 0.093 | Likely Benign | 0.16 | Neutral | 0.296 | Benign | 0.022 | Benign | 4.04 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0883 | 0.3806 | 1 | 2 | -2.6 | 18.03 | ||||||||||||||||||||||||||||||||||
| c.518T>A | L173Q 2D  AIThe SynGAP1 missense variant L173Q is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM. Only AlphaMissense‑Default predicts a pathogenic outcome, while AlphaMissense‑Optimized is uncertain. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence from multiple in silico predictors and the SGM‑Consensus indicates that the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.534167 | Disordered | 0.491566 | Uncertain | 0.390 | 0.631 | 0.375 | -5.605 | Likely Benign | 0.850 | Likely Pathogenic | Ambiguous | 0.133 | Likely Benign | -1.72 | Neutral | 0.022 | Benign | 0.022 | Benign | 3.94 | Benign | 0.08 | Tolerated | 0.1028 | 0.0919 | -2 | -2 | -7.3 | 14.97 | |||||||||||||||||||||||||||||||||||||||
| c.529T>C | F177L 2D  AIThe SynGAP1 missense variant F177L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, and FATHMM. Tools that predict a pathogenic effect are AlphaMissense‑Default and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Pathogenic, while the SGM‑Consensus remains Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence (seven benign predictions versus two pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.461817 | Uncertain | 0.357 | 0.598 | 0.500 | -5.564 | Likely Benign | 0.994 | Likely Pathogenic | Likely Pathogenic | 0.184 | Likely Benign | 0.24 | Neutral | 0.022 | Benign | 0.022 | Benign | 4.41 | Benign | 1.00 | Tolerated | 0.2376 | 0.4280 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.531T>A | F177L 2D  AIThe SynGAP1 missense variant F177L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, and FATHMM. Tools that predict a pathogenic effect are AlphaMissense‑Default and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Pathogenic, while the SGM‑Consensus remains Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence (seven benign predictions versus two pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.461817 | Uncertain | 0.357 | 0.598 | 0.500 | -5.564 | Likely Benign | 0.994 | Likely Pathogenic | Likely Pathogenic | 0.173 | Likely Benign | 0.24 | Neutral | 0.022 | Benign | 0.022 | Benign | 4.41 | Benign | 1.00 | Tolerated | 0.2376 | 0.4280 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.531T>G | F177L 2D  AIThe SynGAP1 missense variant F177L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, and FATHMM. Tools that predict a pathogenic effect are AlphaMissense‑Default and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Pathogenic, while the SGM‑Consensus remains Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence (seven benign predictions versus two pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.461817 | Uncertain | 0.357 | 0.598 | 0.500 | -5.564 | Likely Benign | 0.994 | Likely Pathogenic | Likely Pathogenic | 0.173 | Likely Benign | 0.24 | Neutral | 0.022 | Benign | 0.022 | Benign | 4.41 | Benign | 1.00 | Tolerated | 0.2376 | 0.4280 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.1051G>T | A351S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A351S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: SGM‑Consensus (Likely Benign), REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. Only FATHMM predicts a pathogenic outcome. High‑accuracy methods—AlphaMissense‑Optimized, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and Foldetta (integrating FoldX‑MD and Rosetta)—all report benign or likely benign. No prediction or stability result is missing or inconclusive. Consequently, the variant is most likely benign; this assessment does not contradict any ClinVar annotation, as none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.216401 | Structured | 0.362025 | Uncertain | 0.925 | 0.342 | 0.000 | -4.823 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.24 | Likely Benign | 0.0 | 0.25 | Likely Benign | 0.25 | Likely Benign | 0.27 | Likely Benign | 0.016 | Likely Benign | -1.42 | Neutral | 0.080 | Benign | 0.023 | Benign | 1.88 | Pathogenic | 0.13 | Tolerated | 0.2558 | 0.5334 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.119A>C | D40A 2D  AIThe SynGAP1 missense variant D40A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic call comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign consensus. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are not available. Taken together, the majority of evidence supports a benign interpretation, and this is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.384043 | Structured | 0.432002 | Uncertain | 0.319 | 0.769 | 0.375 | -3.630 | Likely Benign | 0.339 | Likely Benign | Likely Benign | 0.122 | Likely Benign | -1.22 | Neutral | 0.006 | Benign | 0.023 | Benign | 4.05 | Benign | 0.00 | Affected | 0.4299 | 0.7993 | 0 | -2 | 5.3 | -44.01 | |||||||||||||||||||||||||||||||||||||||
| c.1594A>G | T532A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T532A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only FATHMM predicts a pathogenic outcome. Grouping by consensus, the benign‑predicting tools outnumber the single pathogenic prediction. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) reports “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. No prediction or stability result is missing or inconclusive. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.275179 | Structured | 0.021478 | Uncertain | 0.889 | 0.385 | 0.000 | -2.907 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.16 | Likely Benign | 0.0 | 0.13 | Likely Benign | 0.15 | Likely Benign | 0.11 | Likely Benign | 0.165 | Likely Benign | -0.80 | Neutral | 0.032 | Benign | 0.023 | Benign | -1.15 | Pathogenic | 0.14 | Tolerated | 0.3573 | 0.3114 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||
| c.185A>C | D62A 2D  AIThe SynGAP1 D62A missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.425610 | Structured | 0.476010 | Uncertain | 0.575 | 0.720 | 0.125 | -3.983 | Likely Benign | 0.318 | Likely Benign | Likely Benign | 0.070 | Likely Benign | -1.67 | Neutral | 0.006 | Benign | 0.023 | Benign | 4.09 | Benign | 0.00 | Affected | 0.4152 | 0.5972 | 0 | -2 | 5.3 | -44.01 | |||||||||||||||||||||||||||||||||||||||
| c.2008C>G | L670V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense change L670V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score all indicate benign. Only ESM1b predicts pathogenicity, while FoldX, Rosetta, and Foldetta provide uncertain or inconclusive stability results. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is benign; and Foldetta remains uncertain. Taken together, the overwhelming majority of evidence supports a benign interpretation, and this conclusion does not conflict with the absence of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.161087 | Structured | 0.090855 | Uncertain | 0.812 | 0.385 | 0.000 | -9.829 | Likely Pathogenic | 0.113 | Likely Benign | Likely Benign | 1.45 | Ambiguous | 0.1 | 1.05 | Ambiguous | 1.25 | Ambiguous | 0.15 | Likely Benign | 0.025 | Likely Benign | -0.60 | Neutral | 0.127 | Benign | 0.023 | Benign | 3.67 | Benign | 0.74 | Tolerated | 0.1558 | 0.4108 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||
| c.224A>G | E75G 2D  AIThe SynGAP1 missense variant E75G is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized scores the variant as benign, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—labels it “Likely Benign.” No Foldetta stability prediction is available, so it does not influence the assessment. Overall, the preponderance of evidence indicates that E75G is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.443881 | Uncertain | 0.303 | 0.822 | 0.500 | -2.991 | Likely Benign | 0.175 | Likely Benign | Likely Benign | 0.091 | Likely Benign | -1.74 | Neutral | 0.345 | Benign | 0.023 | Benign | 4.01 | Benign | 0.00 | Affected | 0.3078 | 0.5738 | 0 | -2 | 3.1 | -72.06 | |||||||||||||||||||||||||||||||||||||||
| c.2710A>G | M904V 2D  AIThe SynGAP1 missense variant M904V is reported in ClinVar (ID 833650.0) as benign and is present in gnomAD (variant ID 6‑33443262‑A‑G). All evaluated in‑silico predictors classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence strongly supports a benign classification, which aligns with the ClinVar status and shows no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.589073 | Binding | 0.350 | 0.920 | 0.250 | Likely Benign | 2 | 6-33443262-A-G | 77 | 4.78e-5 | -2.907 | Likely Benign | 0.112 | Likely Benign | Likely Benign | 0.058 | Likely Benign | -0.33 | Neutral | 0.039 | Benign | 0.023 | Benign | 2.80 | Benign | 0.10 | Tolerated | 3.77 | 5 | 0.3724 | 0.3948 | 2 | 1 | 2.3 | -32.06 | ||||||||||||||||||||||||||||||||
| c.2712G>A | M904I 2D  AIThe SynGAP1 missense variant M904I is not reported in ClinVar and has no entries in gnomAD, indicating it is not catalogued in these databases. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM all return benign scores, and AlphaMissense‑Optimized also predicts a benign effect. No tool predicts pathogenicity, and the only uncertain result comes from AlphaMissense‑Default, which is inconclusive. The high‑accuracy consensus methods corroborate this benign assessment: the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and AlphaMissense‑Optimized explicitly labels the variant as benign. Foldetta, a protein‑folding stability predictor, has no available result for this residue. Taken together, the evidence overwhelmingly supports a benign impact for M904I, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Thus, the variant is most likely benign, and this conclusion does not contradict the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.589073 | Binding | 0.350 | 0.920 | 0.250 | -3.845 | Likely Benign | 0.385 | Ambiguous | Likely Benign | 0.023 | Likely Benign | -0.48 | Neutral | 0.039 | Benign | 0.023 | Benign | 2.78 | Benign | 0.07 | Tolerated | 0.1694 | 0.3633 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2712G>C | M904I 2D  AIThe SynGAP1 missense variant M904I is not reported in ClinVar and has no entries in gnomAD, indicating it is not catalogued in these databases. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM all return benign scores, and AlphaMissense‑Optimized also predicts a benign effect. No tool predicts pathogenicity, and the only uncertain result comes from AlphaMissense‑Default, which is inconclusive. The high‑accuracy consensus methods corroborate this benign assessment: the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and AlphaMissense‑Optimized also indicates a benign outcome. Foldetta, a protein‑folding stability predictor, has no available result for this variant. Taken together, the evidence overwhelmingly supports a benign impact, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.589073 | Binding | 0.350 | 0.920 | 0.250 | -3.845 | Likely Benign | 0.385 | Ambiguous | Likely Benign | 0.023 | Likely Benign | -0.48 | Neutral | 0.039 | Benign | 0.023 | Benign | 2.78 | Benign | 0.07 | Tolerated | 0.1694 | 0.3633 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2712G>T | M904I 2D  AIThe SynGAP1 missense variant M904I is not reported in ClinVar and has no entries in gnomAD, indicating it is not catalogued in these databases. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM all return benign scores, and AlphaMissense‑Optimized also predicts a benign effect. No tool predicts pathogenicity, and the only uncertain result comes from AlphaMissense‑Default, which is inconclusive. The high‑accuracy consensus methods corroborate this benign assessment: the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and AlphaMissense‑Optimized also indicates a benign outcome. Foldetta, a protein‑folding stability predictor, has no available result for this variant. Taken together, the evidence overwhelmingly supports a benign impact, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.671169 | Disordered | 0.589073 | Binding | 0.350 | 0.920 | 0.250 | -3.845 | Likely Benign | 0.385 | Ambiguous | Likely Benign | 0.023 | Likely Benign | -0.48 | Neutral | 0.039 | Benign | 0.023 | Benign | 2.78 | Benign | 0.07 | Tolerated | 0.1694 | 0.3633 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2902G>A | G968S 2D  AIThe SynGAP1 missense variant G968S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign, and this conclusion is not contradicted by any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.978316 | Disordered | 0.961360 | Binding | 0.327 | 0.896 | 0.750 | -4.484 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.124 | Likely Benign | -0.35 | Neutral | 0.058 | Benign | 0.023 | Benign | 4.22 | Benign | 0.37 | Tolerated | 0.2482 | 0.5699 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.2977C>T | P993S 2D  AIThe SynGAP1 missense variant P993S is reported in gnomAD (ID 6‑33443529‑C‑T) and has no ClinVar entry. All evaluated in silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are unavailable. Based on the unanimous benign predictions and lack of ClinVar pathogenicity, the variant is most likely benign and does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.852992 | Disordered | 0.923979 | Binding | 0.319 | 0.908 | 0.750 | 6-33443529-C-T | 3 | 1.86e-6 | -3.665 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.054 | Likely Benign | -0.13 | Neutral | 0.011 | Benign | 0.023 | Benign | 4.26 | Benign | 0.59 | Tolerated | 4.32 | 2 | 0.3421 | 0.5465 | -1 | 1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||||
| c.3289C>T | P1097S 2D  AIThe SynGAP1 missense variant P1097S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.974957 | Binding | 0.384 | 0.858 | 1.000 | -3.748 | Likely Benign | 0.116 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -1.04 | Neutral | 0.025 | Benign | 0.023 | Benign | 2.62 | Benign | 0.70 | Tolerated | 0.3332 | 0.5691 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.3352A>C | S1118R 2D  AIThe SynGAP1 missense variant S1118R is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that S1118R is most likely benign, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.876521 | Disordered | 0.828802 | Binding | 0.310 | 0.919 | 0.750 | -2.670 | Likely Benign | 0.553 | Ambiguous | Likely Benign | 0.175 | Likely Benign | -0.74 | Neutral | 0.034 | Benign | 0.023 | Benign | 5.17 | Benign | 0.05 | Affected | 4.32 | 2 | 0.1314 | 0.3431 | -1 | 0 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||||
| c.3354C>A | S1118R 2D  AIThe SynGAP1 missense variant S1118R (ClinVar ID 2656489.0) is listed as ClinVar status Uncertain and is not present in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only SIFT predicts a pathogenic effect, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this is consistent with the ClinVar Uncertain designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.876521 | Disordered | 0.828802 | Binding | 0.310 | 0.919 | 0.750 | Uncertain | 1 | -2.670 | Likely Benign | 0.553 | Ambiguous | Likely Benign | 0.166 | Likely Benign | -0.74 | Neutral | 0.034 | Benign | 0.023 | Benign | 5.17 | Benign | 0.05 | Affected | 4.32 | 2 | 0.1314 | 0.3431 | -1 | 0 | -3.7 | 69.11 | |||||||||||||||||||||||||||||||||||
| c.3354C>G | S1118R 2D  AIThe SynGAP1 missense variant S1118R is listed in gnomAD (ID 6‑33443906‑C‑G) but has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote). Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.876521 | Disordered | 0.828802 | Binding | 0.310 | 0.919 | 0.750 | 6-33443906-C-G | -2.670 | Likely Benign | 0.553 | Ambiguous | Likely Benign | 0.165 | Likely Benign | -0.74 | Neutral | 0.034 | Benign | 0.023 | Benign | 5.17 | Benign | 0.05 | Affected | 4.32 | 2 | 0.1314 | 0.3431 | -1 | 0 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||||||
| c.416G>C | S139T 2D  AIThe SynGAP1 missense variant S139T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The high‑accuracy consensus (SGM‑Consensus) also indicates a likely benign classification, and AlphaMissense‑Optimized independently predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, was not available for this variant. Overall, the majority of computational evidence supports a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.553315 | Disordered | 0.600637 | Binding | 0.353 | 0.900 | 0.250 | -4.518 | Likely Benign | 0.463 | Ambiguous | Likely Benign | 0.122 | Likely Benign | -1.70 | Neutral | 0.034 | Benign | 0.023 | Benign | 4.13 | Benign | 0.25 | Tolerated | 0.1655 | 0.4416 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.680G>C | G227A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G227A is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Those that agree on a pathogenic effect are REVEL, FoldX, Rosetta, Foldetta, PROVEAN, SIFT, and AlphaMissense‑Default. Predictions that remain uncertain are premPS, ESM1b, and AlphaMissense‑Optimized. High‑accuracy assessments further support pathogenicity: the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a pathogenic majority, and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts pathogenic. No prediction or stability result is missing or inconclusive. Based on the overall consensus of the majority of tools and the high‑accuracy predictions, the variant is most likely pathogenic, and this assessment does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.106997 | Structured | 0.329995 | Uncertain | 0.800 | 0.329 | 0.250 | -7.344 | In-Between | 0.920 | Likely Pathogenic | Ambiguous | 2.45 | Destabilizing | 0.4 | 5.14 | Destabilizing | 3.80 | Destabilizing | 0.78 | Ambiguous | 0.682 | Likely Pathogenic | -5.08 | Deleterious | 0.097 | Benign | 0.023 | Benign | 5.71 | Benign | 0.04 | Affected | 0.3987 | 0.5221 | 1 | 0 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||
| c.1054A>G | T352A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T352A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: SGM‑Consensus, REVEL, FoldX, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only FATHMM predicts pathogenic, while Rosetta remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign verdict; and Foldetta, integrating FoldX‑MD (benign) and Rosetta (uncertain), reports benign stability. Overall, the preponderance of evidence indicates that T352A is most likely benign, and this conclusion does not contradict the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.137348 | Structured | 0.367886 | Uncertain | 0.926 | 0.329 | 0.000 | -1.952 | Likely Benign | 0.055 | Likely Benign | Likely Benign | 0.12 | Likely Benign | 0.0 | 0.50 | Ambiguous | 0.31 | Likely Benign | 0.45 | Likely Benign | 0.032 | Likely Benign | -1.09 | Neutral | 0.031 | Benign | 0.024 | Benign | 1.73 | Pathogenic | 0.29 | Tolerated | 0.4341 | 0.4938 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||
| c.1075A>T | T359S 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant T359S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score. Only FATHMM predicts a pathogenic outcome, while FoldX, Rosetta, Foldetta, and premPS are inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also favors benign. Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, is uncertain. Overall, the majority of evidence points to a benign effect, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.281712 | Structured | 0.414952 | Uncertain | 0.939 | 0.480 | 0.250 | -4.624 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.50 | Ambiguous | 0.0 | 0.73 | Ambiguous | 0.62 | Ambiguous | 0.66 | Ambiguous | 0.100 | Likely Benign | -1.87 | Neutral | 0.146 | Benign | 0.024 | Benign | 1.76 | Pathogenic | 0.27 | Tolerated | 0.3637 | 0.4996 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||
| c.1187G>C | G396A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G396A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, PROVEAN, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also yields a benign verdict, while Foldetta (combining FoldX‑MD and Rosetta outputs) is inconclusive. Consequently, the variant is most likely benign based on the available predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.414856 | Structured | 0.394626 | Uncertain | 0.640 | 0.584 | 0.500 | -3.655 | Likely Benign | 0.103 | Likely Benign | Likely Benign | 1.74 | Ambiguous | 0.7 | 1.90 | Ambiguous | 1.82 | Ambiguous | 0.41 | Likely Benign | 0.274 | Likely Benign | -1.28 | Neutral | 0.062 | Benign | 0.024 | Benign | 3.93 | Benign | 0.84 | Tolerated | 0.3846 | 0.5255 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||
| c.1196C>G | A399G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A399G is not reported in ClinVar and is absent from gnomAD. Across a panel of in silico predictors, all available pathogenicity scores classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. Uncertain results from FoldX, Rosetta, Foldetta, and premPS are treated as unavailable. High‑accuracy assessments reinforce the benign prediction: AlphaMissense‑Optimized is benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates benign. Foldetta, which integrates FoldX‑MD and Rosetta outputs, is inconclusive and therefore not considered evidence. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar annotation (none present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.394753 | Structured | 0.407674 | Uncertain | 0.939 | 0.490 | 0.125 | -6.513 | Likely Benign | 0.215 | Likely Benign | Likely Benign | 1.03 | Ambiguous | 0.1 | 1.77 | Ambiguous | 1.40 | Ambiguous | 0.74 | Ambiguous | 0.153 | Likely Benign | -1.59 | Neutral | 0.062 | Benign | 0.024 | Benign | 5.39 | Benign | 0.13 | Tolerated | 0.2174 | 0.4532 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||
| c.1598C>T | A533V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A533V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that indicate a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, Foldetta, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are SIFT and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as likely benign, and Foldetta as benign, all supporting a non‑pathogenic interpretation. No prediction or folding‑stability result is missing or inconclusive. Based on the overall consensus of the available predictions, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar evidence exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.179055 | Structured | 0.026324 | Uncertain | 0.843 | 0.393 | 0.000 | -6.948 | Likely Benign | 0.148 | Likely Benign | Likely Benign | 0.23 | Likely Benign | 0.0 | 0.51 | Ambiguous | 0.37 | Likely Benign | 0.25 | Likely Benign | 0.170 | Likely Benign | -2.41 | Neutral | 0.149 | Benign | 0.024 | Benign | -1.28 | Pathogenic | 0.05 | Affected | 0.1243 | 0.6205 | 0 | 0 | 2.4 | 28.05 | |||||||||||||||||||||||||||||
| c.244C>G | L82V 2D  AIThe SynGAP1 missense variant L82V is not reported in ClinVar (ClinVar status: not listed) but is present in gnomAD (ID 6‑33425852‑C‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Those that predict a pathogenic effect are SIFT and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain, and no Foldetta stability result is available. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments therefore indicate a benign likelihood: AlphaMissense‑Optimized is inconclusive, SGM‑Consensus is likely benign, and Foldetta data are missing. Overall, the majority of predictions support a benign impact, and this is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.517720 | Binding | 0.284 | 0.890 | 0.375 | 6-33425852-C-G | -6.701 | Likely Benign | 0.914 | Likely Pathogenic | Ambiguous | 0.065 | Likely Benign | -1.13 | Neutral | 0.371 | Benign | 0.024 | Benign | 3.72 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1467 | 0.2353 | 1 | 2 | 0.4 | -14.03 | ||||||||||||||||||||||||||||||||||||
| c.292C>T | H98Y 2D  AIThe SynGAP1 missense variant H98Y is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.733139 | Disordered | 0.631713 | Binding | 0.348 | 0.872 | 0.625 | -4.050 | Likely Benign | 0.135 | Likely Benign | Likely Benign | 0.112 | Likely Benign | -0.86 | Neutral | 0.444 | Benign | 0.024 | Benign | 4.19 | Benign | 0.00 | Affected | 0.0950 | 0.5024 | 0 | 2 | 1.9 | 26.03 | |||||||||||||||||||||||||||||||||||||||
| c.293A>C | H98P 2D  AIThe SynGAP1 missense variant H98P is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. There is no ClinVar classification to contradict this conclusion. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.733139 | Disordered | 0.631713 | Binding | 0.348 | 0.872 | 0.625 | -1.460 | Likely Benign | 0.069 | Likely Benign | Likely Benign | 0.275 | Likely Benign | -0.83 | Neutral | 0.659 | Possibly Damaging | 0.024 | Benign | 4.20 | Benign | 0.00 | Affected | 0.2062 | 0.4669 | 0 | -2 | 1.6 | -40.02 | |||||||||||||||||||||||||||||||||||||||
| c.35G>C | S12T 2D  AIThe SynGAP1 missense variant S12T is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus, SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a benign effect. AlphaMissense‑Optimized independently scores the variant as benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this assessment does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.480142 | Structured | 0.490599 | Uncertain | 0.355 | 0.916 | 0.500 | -4.304 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.075 | Likely Benign | -0.16 | Neutral | 0.208 | Benign | 0.024 | Benign | 4.14 | Benign | 0.00 | Affected | 0.1487 | 0.6303 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3962C>G | P1321R 2D  AIThe SynGAP1 missense variant P1321R is reported in gnomAD (ID 6‑33451836‑C‑G) but has no ClinVar entry. Across a broad panel of in silico predictors, every tool examined—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—classifies the substitution as benign. No pathogenic predictions are present. Grouping the results, the benign consensus is unanimous; the pathogenic group is empty. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign status. Foldetta stability analysis is not available for this variant. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar classification (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.933505 | Binding | 0.463 | 0.828 | 0.875 | 6-33451836-C-G | 1 | 6.58e-7 | -5.310 | Likely Benign | 0.154 | Likely Benign | Likely Benign | 0.034 | Likely Benign | -1.10 | Neutral | 0.389 | Benign | 0.024 | Benign | 4.25 | Benign | 0.09 | Tolerated | 3.77 | 5 | 0.1722 | 0.3635 | -2 | 0 | -2.9 | 59.07 | ||||||||||||||||||||||||||||||||||
| c.1290G>A | M430I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M430I is catalogued in gnomAD (6‑33438195‑G‑A) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from FoldX, premPS, and AlphaMissense‑Default. High‑accuracy assessments reinforce the benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) reports “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) also indicates benign. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.071867 | Structured | 0.385298 | Uncertain | 0.952 | 0.306 | 0.000 | 6-33438195-G-A | 1 | 6.19e-7 | -4.655 | Likely Benign | 0.420 | Ambiguous | Likely Benign | 1.22 | Ambiguous | 0.1 | -0.29 | Likely Benign | 0.47 | Likely Benign | 0.65 | Ambiguous | 0.068 | Likely Benign | -1.62 | Neutral | 0.134 | Benign | 0.025 | Benign | 3.54 | Benign | 0.40 | Tolerated | 3.37 | 29 | 0.1524 | 0.4116 | 1 | 2 | 2.6 | -18.03 | ||||||||||||||||||||||||
| c.1290G>C | M430I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M430I is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the remaining predictions (FoldX, premPS, AlphaMissense‑Default) are uncertain. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized scores the variant as benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also classifies the variant as benign. Taken together, the consensus of both general and high‑accuracy predictors is that M430I is most likely benign, and this conclusion does not contradict the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.071867 | Structured | 0.385298 | Uncertain | 0.952 | 0.306 | 0.000 | -4.655 | Likely Benign | 0.420 | Ambiguous | Likely Benign | 1.22 | Ambiguous | 0.1 | -0.29 | Likely Benign | 0.47 | Likely Benign | 0.65 | Ambiguous | 0.068 | Likely Benign | -1.62 | Neutral | 0.134 | Benign | 0.025 | Benign | 3.54 | Benign | 0.40 | Tolerated | 3.37 | 29 | 0.1524 | 0.4116 | 1 | 2 | 2.6 | -18.03 | |||||||||||||||||||||||||||
| c.1290G>T | M430I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M430I is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, Rosetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome. Predictions that are inconclusive are FoldX, premPS, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, while Foldetta’s combined FoldX‑MD/Rosetta output is unavailable due to the uncertain FoldX result. Overall, the evidence overwhelmingly supports a benign classification for M430I, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.071867 | Structured | 0.385298 | Uncertain | 0.952 | 0.306 | 0.000 | -4.655 | Likely Benign | 0.420 | Ambiguous | Likely Benign | 1.22 | Ambiguous | 0.1 | -0.29 | Likely Benign | 0.47 | Likely Benign | 0.65 | Ambiguous | 0.068 | Likely Benign | -1.62 | Neutral | 0.134 | Benign | 0.025 | Benign | 3.54 | Benign | 0.40 | Tolerated | 3.37 | 29 | 0.1524 | 0.4116 | 1 | 2 | 2.6 | -18.03 | |||||||||||||||||||||||||||
| c.2101C>T | P701S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant P701S (ClinVar ID 2995856.0) is listed as “Uncertain” in ClinVar and is present in gnomAD (ID 6‑33441360‑C‑T). Prediction tools that agree on a benign effect include REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). No tool in the dataset predicts a pathogenic outcome; all remaining predictions are either benign or uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as Benign, the SGM‑Consensus as Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as Uncertain. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.074921 | Structured | 0.404318 | Uncertain | 0.918 | 0.345 | 0.000 | Uncertain | 1 | 6-33441360-C-T | 3 | 1.86e-6 | -4.375 | Likely Benign | 0.221 | Likely Benign | Likely Benign | 1.33 | Ambiguous | 0.0 | 0.12 | Likely Benign | 0.73 | Ambiguous | -0.36 | Likely Benign | 0.132 | Likely Benign | 0.78 | Neutral | 0.044 | Benign | 0.025 | Benign | 3.48 | Benign | 1.00 | Tolerated | 3.47 | 10 | 0.3149 | 0.3782 | -1 | 1 | 0.8 | -10.04 | 10.1016/j.ajhg.2020.11.011 | |||||||||||||||||||||
| c.2662G>T | A888S 2D  AIThe SynGAP1 missense variant A888S is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.784345 | Disordered | 0.575860 | Binding | 0.352 | 0.928 | 0.625 | -3.580 | Likely Benign | 0.082 | Likely Benign | Likely Benign | 0.034 | Likely Benign | -0.66 | Neutral | 0.017 | Benign | 0.025 | Benign | 2.64 | Benign | 0.00 | Affected | 0.2661 | 0.5626 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.3121C>A | P1041T 2D  AIThe SynGAP1 missense variant P1041T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this conclusion does not contradict any ClinVar annotation (none present). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.962114 | Disordered | 0.967463 | Binding | 0.345 | 0.833 | 0.625 | -5.009 | Likely Benign | 0.187 | Likely Benign | Likely Benign | 0.361 | Likely Benign | -2.45 | Neutral | 0.051 | Benign | 0.025 | Benign | 5.50 | Benign | 0.10 | Tolerated | 0.1632 | 0.6699 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.3229A>T | T1077S 2D  AIThe SynGAP1 missense variant T1077S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.903857 | Disordered | 0.988141 | Binding | 0.329 | 0.892 | 0.750 | -2.929 | Likely Benign | 0.201 | Likely Benign | Likely Benign | 0.114 | Likely Benign | -0.45 | Neutral | 0.068 | Benign | 0.025 | Benign | 4.32 | Benign | 0.06 | Tolerated | 0.2788 | 0.4635 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3236G>T | S1079I 2D  AIThe SynGAP1 missense variant S1079I is not reported in ClinVar (ClinVar status: none) and is absent from gnomAD (gnomAD ID: none). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 pathogenic vs. 2 benign). Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which currently has no classification for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.919029 | Disordered | 0.983887 | Binding | 0.307 | 0.900 | 0.750 | -4.732 | Likely Benign | 0.688 | Likely Pathogenic | Likely Benign | 0.093 | Likely Benign | -2.86 | Deleterious | 0.078 | Benign | 0.025 | Benign | 3.83 | Benign | 0.00 | Affected | 0.0921 | 0.4775 | -1 | -2 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||||||||||||
| c.328G>C | V110L 2D  AIThe SynGAP1 missense variant V110L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The high‑accuracy consensus, SGM‑Consensus, classifies the variant as Likely Benign, and AlphaMissense‑Optimized also reports a benign prediction. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence overwhelmingly supports a benign impact for V110L, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.622677 | Disordered | 0.665934 | Binding | 0.347 | 0.860 | 0.750 | -3.024 | Likely Benign | 0.357 | Ambiguous | Likely Benign | 0.058 | Likely Benign | -0.71 | Neutral | 0.158 | Benign | 0.025 | Benign | 4.20 | Benign | 0.50 | Tolerated | 0.1165 | 0.5145 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3365G>T | G1122V 2D  AIThe SynGAP1 missense variant G1122V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) reports likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign, and this assessment is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.896620 | Disordered | 0.814918 | Binding | 0.357 | 0.932 | 0.875 | -6.398 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.271 | Likely Benign | -1.18 | Neutral | 0.059 | Benign | 0.025 | Benign | 4.49 | Benign | 0.10 | Tolerated | 0.1381 | 0.3694 | -1 | -3 | 4.6 | 42.08 | |||||||||||||||||||||||||||||||||||||||
| c.364C>T | P122S 2D  AIThe SynGAP1 missense variant P122S is catalogued in gnomAD (ID 6‑33432229‑C‑T) but has no ClinVar entry. Across a broad panel of in silico predictors, every tool reports a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool in the set predicts pathogenicity, so the pathogenic group is empty. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized returns a benign prediction, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—labels the variant as Likely Benign. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status remains unavailable. Overall, the computational evidence overwhelmingly supports a benign classification, and this is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.618285 | Disordered | 0.672358 | Binding | 0.400 | 0.887 | 0.750 | 6-33432229-C-T | 1 | 6.20e-7 | -3.389 | Likely Benign | 0.098 | Likely Benign | Likely Benign | 0.091 | Likely Benign | -1.13 | Neutral | 0.036 | Benign | 0.025 | Benign | 4.22 | Benign | 0.31 | Tolerated | 3.61 | 5 | 0.4009 | 0.4759 | -1 | 1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||||
| c.3895C>G | L1299V 2D  AIThe SynGAP1 missense variant L1299V is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification—there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.771762 | Disordered | 0.896323 | Binding | 0.398 | 0.832 | 0.750 | -4.502 | Likely Benign | 0.121 | Likely Benign | Likely Benign | 0.385 | Likely Benign | -0.02 | Neutral | 0.124 | Benign | 0.025 | Benign | 2.89 | Benign | 0.09 | Tolerated | 0.1726 | 0.3544 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.439C>A | Q147K 2D  AIThe SynGAP1 missense variant Q147K is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments are inconclusive: the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a 2‑vs‑2 split, and Foldetta results are unavailable. Overall, the majority of available predictions lean toward a benign impact, and there is no ClinVar annotation to contradict this assessment. Thus, the variant is most likely benign based on current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.541878 | Disordered | 0.503877 | Binding | 0.349 | 0.840 | 0.625 | -12.562 | Likely Pathogenic | 0.950 | Likely Pathogenic | Ambiguous | 0.084 | Likely Benign | -2.12 | Neutral | 0.018 | Benign | 0.025 | Benign | 3.94 | Benign | 0.03 | Affected | 0.2012 | 0.3351 | 1 | 1 | -0.4 | 0.04 | ||||||||||||||||||||||||||||||||||||||||
| c.439C>G | Q147E 2D  AIThe SynGAP1 missense variant Q147E is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. The high‑accuracy AlphaMissense‑Optimized result is unavailable, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—yields a tie (2 pathogenic, 2 benign) and is therefore considered uncertain. Foldetta, which would assess protein‑folding stability, has no reported output for this variant, so its result is unavailable. Overall, the majority of evidence (five benign vs. three pathogenic) points to a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.541878 | Disordered | 0.503877 | Binding | 0.349 | 0.840 | 0.625 | -10.347 | Likely Pathogenic | 0.840 | Likely Pathogenic | Ambiguous | 0.093 | Likely Benign | -1.47 | Neutral | 0.018 | Benign | 0.025 | Benign | 3.94 | Benign | 0.02 | Affected | 0.1561 | 0.1838 | 2 | 2 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||||||||||||
| c.506A>C | D169A 2D  AIThe SynGAP1 D169A missense variant is not reported in ClinVar and has no entries in gnomAD, indicating it is not catalogued in these databases. Functional prediction tools show a split: benign calls come from REVEL, polyPhen‑2 (HumDiv and HumVar) and FATHMM, while pathogenic calls arise from PROVEAN, SIFT, ESM1b and AlphaMissense‑Default. Grouping by consensus, the benign set includes REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM; the pathogenic set includes PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. High‑accuracy assessments further support a deleterious effect: AlphaMissense‑Optimized is uncertain, but the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—labels the variant as Likely Pathogenic. Foldetta predictions are unavailable. Overall, the balance of evidence points to a pathogenic impact, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.418646 | Structured | 0.497160 | Uncertain | 0.420 | 0.675 | 0.125 | -11.065 | Likely Pathogenic | 0.874 | Likely Pathogenic | Ambiguous | 0.159 | Likely Benign | -3.15 | Deleterious | 0.018 | Benign | 0.025 | Benign | 4.10 | Benign | 0.01 | Affected | 0.4012 | 0.6809 | 0 | -2 | 5.3 | -44.01 | |||||||||||||||||||||||||||||||||||||||
| c.652T>C | F218L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F218L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and FATHMM. Those that predict a pathogenic effect comprise REVEL, Rosetta, premPS, PROVEAN, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). FoldX and Foldetta provide uncertain results and are therefore considered unavailable for interpretation. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, the SGM Consensus as likely pathogenic, and Foldetta as uncertain. Overall, the majority of evidence points to a pathogenic impact for F218L, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.281712 | Structured | 0.408725 | Uncertain | 0.848 | 0.272 | 0.000 | -8.861 | Likely Pathogenic | 0.999 | Likely Pathogenic | Likely Pathogenic | 0.96 | Ambiguous | 0.4 | 2.36 | Destabilizing | 1.66 | Ambiguous | 1.08 | Destabilizing | 0.546 | Likely Pathogenic | -3.80 | Deleterious | 0.158 | Benign | 0.025 | Benign | 5.90 | Benign | 0.15 | Tolerated | 0.2331 | 0.3241 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||
| c.654C>A | F218L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F218L is not reported in ClinVar (ClinVar status: none) and is absent from gnomAD (gnomAD ID: none). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and FATHMM. Tools that agree on a pathogenic effect comprise SGM‑Consensus (Likely Pathogenic), Rosetta, premPS, PROVEAN, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. FoldX and Foldetta are inconclusive and are treated as unavailable. High‑accuracy methods further support pathogenicity: AlphaMissense‑Optimized predicts pathogenic; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also predicts Likely Pathogenic; Foldetta remains uncertain. Overall, the majority of evidence points to a pathogenic impact. There is no ClinVar annotation to contradict this assessment, so the variant is most likely pathogenic. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.281712 | Structured | 0.408725 | Uncertain | 0.848 | 0.272 | 0.000 | -8.861 | Likely Pathogenic | 0.999 | Likely Pathogenic | Likely Pathogenic | 0.96 | Ambiguous | 0.4 | 2.36 | Destabilizing | 1.66 | Ambiguous | 1.08 | Destabilizing | 0.477 | Likely Benign | -3.80 | Deleterious | 0.158 | Benign | 0.025 | Benign | 5.90 | Benign | 0.15 | Tolerated | 0.2331 | 0.3241 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||
| c.654C>G | F218L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F218L is not reported in ClinVar (ClinVar status: none) and is absent from gnomAD (gnomAD ID: none). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and FATHMM. Tools that agree on a pathogenic effect comprise SGM‑Consensus (Likely Pathogenic), Rosetta, premPS, PROVEAN, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. FoldX and Foldetta are inconclusive and are treated as unavailable. High‑accuracy methods further support pathogenicity: AlphaMissense‑Optimized predicts pathogenic; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also predicts Likely Pathogenic; Foldetta remains uncertain. Overall, the majority of evidence points to a pathogenic impact. There is no ClinVar annotation to contradict this assessment, so the variant is most likely pathogenic. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.281712 | Structured | 0.408725 | Uncertain | 0.848 | 0.272 | 0.000 | -8.861 | Likely Pathogenic | 0.999 | Likely Pathogenic | Likely Pathogenic | 0.96 | Ambiguous | 0.4 | 2.36 | Destabilizing | 1.66 | Ambiguous | 1.08 | Destabilizing | 0.477 | Likely Benign | -3.80 | Deleterious | 0.158 | Benign | 0.025 | Benign | 5.90 | Benign | 0.15 | Tolerated | 0.2331 | 0.3241 | 2 | 0 | 1.0 | -34.02 | |||||||||||||||||||||||||||||
| c.694G>T | A232S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A232S is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Across the evaluated in‑silico predictors, all tools except AlphaMissense‑Default converge on a benign interpretation: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM all predict benign. AlphaMissense‑Default is uncertain, while AlphaMissense‑Optimized predicts benign. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign; and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.254060 | Structured | 0.307228 | Uncertain | 0.878 | 0.305 | 0.000 | -3.264 | Likely Benign | 0.344 | Ambiguous | Likely Benign | 0.23 | Likely Benign | 0.1 | -0.23 | Likely Benign | 0.00 | Likely Benign | -0.20 | Likely Benign | 0.378 | Likely Benign | 0.54 | Neutral | 0.057 | Benign | 0.025 | Benign | 5.95 | Benign | 1.00 | Tolerated | 0.2777 | 0.5300 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.1229G>C | S410T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S410T has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include AlphaMissense‑Default, AlphaMissense‑Optimized, ESM1b, FATHMM, PROVEAN, FoldX, Foldetta, SGM‑Consensus, REVEL, polyPhen‑2 (HumDiv and HumVar), SIFT, and premPS. No tool predicts a pathogenic outcome; the only inconclusive result is from Rosetta, which is treated as unavailable. High‑accuracy assessments are consistent: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also predicts benign. **Thus, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none is available).** Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.098513 | Structured | 0.349627 | Uncertain | 0.908 | 0.206 | 0.000 | -6.559 | Likely Benign | 0.123 | Likely Benign | Likely Benign | -0.32 | Likely Benign | 0.1 | -0.52 | Ambiguous | -0.42 | Likely Benign | 0.00 | Likely Benign | 0.066 | Likely Benign | -0.78 | Neutral | 0.080 | Benign | 0.026 | Benign | 4.24 | Benign | 0.84 | Tolerated | 0.1371 | 0.7159 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||
| c.1270G>C | V424L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V424L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Only AlphaMissense‑Default predicts a pathogenic outcome, while SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) reports a likely benign classification. Stability‑based methods are inconclusive: FoldX is uncertain, and Foldetta (combining FoldX‑MD and Rosetta outputs) is also uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar status because no ClinVar classification exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.050641 | Structured | 0.411431 | Uncertain | 0.973 | 0.248 | 0.000 | -4.941 | Likely Benign | 0.742 | Likely Pathogenic | Likely Benign | -0.90 | Ambiguous | 0.2 | -0.41 | Likely Benign | -0.66 | Ambiguous | 0.43 | Likely Benign | 0.159 | Likely Benign | -1.68 | Neutral | 0.327 | Benign | 0.026 | Benign | 4.50 | Benign | 0.59 | Tolerated | 0.1018 | 0.2680 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||
| c.1346G>A | S449N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S449N is not reported in ClinVar (ClinVar status: not listed) but is present in the gnomAD database (gnomAD ID: 6‑33438251‑G‑A). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from premPS (uncertain) and ESM1b (uncertain). High‑accuracy assessments reinforce the benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, is benign. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.254060 | Structured | 0.301437 | Uncertain | 0.958 | 0.251 | 0.000 | 6-33438251-G-A | 1 | 6.19e-7 | -7.692 | In-Between | 0.210 | Likely Benign | Likely Benign | 0.38 | Likely Benign | 0.1 | -0.03 | Likely Benign | 0.18 | Likely Benign | 0.81 | Ambiguous | 0.070 | Likely Benign | -2.31 | Neutral | 0.372 | Benign | 0.026 | Benign | 3.37 | Benign | 0.18 | Tolerated | 3.37 | 32 | 0.1085 | 0.3767 | 1 | 1 | -2.7 | 27.03 | ||||||||||||||||||||||||
| c.2336G>A | S779N 2D  AIThe SynGAP1 missense variant S779N is listed in gnomAD (ID 6‑33442494‑G‑A) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. A high‑accuracy consensus (SGM Consensus) derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN yields a benign majority. The AlphaMissense‑Optimized score is benign, and no Foldetta stability assessment is available. Taken together, the majority of evidence points to a benign impact. This conclusion is consistent with the absence of a ClinVar pathogenic classification, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.509769 | Disordered | 0.834974 | Binding | 0.321 | 0.890 | 0.375 | 6-33442494-G-A | -4.880 | Likely Benign | 0.384 | Ambiguous | Likely Benign | 0.087 | Likely Benign | -0.75 | Neutral | 0.021 | Benign | 0.026 | Benign | 2.30 | Pathogenic | 0.23 | Tolerated | 3.64 | 6 | 0.1327 | 0.4984 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||||||||||||
| c.2347A>G | M783V 2D  AIThe SynGAP1 missense variant M783V is catalogued in gnomAD (ID 6‑33442899‑A‑G) and has no ClinVar entry. All evaluated in‑silico predictors classify it as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. Foldetta results are unavailable. Consequently, the variant is most likely benign, and this prediction does not contradict any ClinVar status (none is assigned). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.736850 | Disordered | 0.738119 | Binding | 0.331 | 0.889 | 0.625 | 6-33442899-A-G | 1 | 6.21e-7 | -3.453 | Likely Benign | 0.086 | Likely Benign | Likely Benign | 0.065 | Likely Benign | -0.96 | Neutral | 0.072 | Benign | 0.026 | Benign | 2.85 | Benign | 0.12 | Tolerated | 3.64 | 6 | 0.3444 | 0.3710 | 1 | 2 | 2.3 | -32.06 | ||||||||||||||||||||||||||||||||||
| c.2383C>A | P795T 2D  AIThe SynGAP1 missense variant P795T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity, and the only uncertain result comes from ESM1b. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments further support this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also likely benign; Foldetta results are not available. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.972450 | Disordered | 0.410339 | Uncertain | 0.457 | 0.903 | 0.875 | -7.030 | In-Between | 0.060 | Likely Benign | Likely Benign | 0.068 | Likely Benign | -0.63 | Neutral | 0.036 | Benign | 0.026 | Benign | 4.26 | Benign | 0.09 | Tolerated | 0.1713 | 0.6381 | 0 | -1 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||||||
| c.2390C>G | P797R 2D  AIThe SynGAP1 missense variant P797R is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate a benign effect, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also reports “Likely Benign.” No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates a benign outcome; Foldetta results are not available. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.926919 | Disordered | 0.449970 | Uncertain | 0.561 | 0.902 | 0.875 | -5.642 | Likely Benign | 0.179 | Likely Benign | Likely Benign | 0.045 | Likely Benign | 0.00 | Neutral | 0.006 | Benign | 0.026 | Benign | 4.28 | Benign | 0.20 | Tolerated | 0.1549 | 0.3085 | 0 | -2 | -2.9 | 59.07 | ||||||||||||||||||||||||||||||||||||||
| c.2540A>G | Q847R 2D  AIThe SynGAP1 missense variant Q847R has no ClinVar entry and is not reported in gnomAD. Functional prediction tools show a split: benign calls come from REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, and AlphaMissense‑Optimized; pathogenic calls come from PROVEAN, SIFT, FATHMM, and AlphaMissense‑Default. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Pathogenic. High‑accuracy assessments further reveal AlphaMissense‑Optimized as benign, SGM‑Consensus as Likely Pathogenic, and Foldetta results are unavailable. Thus, the overall prediction leans toward benign based on the majority of tools, but the high‑accuracy SGM‑Consensus contradicts this by indicating likely pathogenic. No ClinVar annotation exists, so there is no conflict with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.653063 | Disordered | 0.577677 | Binding | 0.282 | 0.818 | 0.500 | -3.232 | Likely Benign | 0.662 | Likely Pathogenic | Likely Benign | 0.256 | Likely Benign | -2.63 | Deleterious | 0.014 | Benign | 0.026 | Benign | 2.30 | Pathogenic | 0.00 | Affected | 0.1404 | 0.1979 | 1 | 1 | -1.0 | 28.06 | |||||||||||||||||||||||||||||||||||||||
| c.2581T>A | S861T 2D  AIThe SynGAP1 missense variant S861T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. The high‑accuracy consensus, SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN), also reports a likely benign outcome. AlphaMissense‑Optimized likewise predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.557691 | Disordered | 0.540903 | Binding | 0.285 | 0.797 | 0.250 | -4.462 | Likely Benign | 0.096 | Likely Benign | Likely Benign | 0.022 | Likely Benign | -0.83 | Neutral | 0.043 | Benign | 0.026 | Benign | 3.99 | Benign | 0.14 | Tolerated | 0.1262 | 0.6245 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2611C>G | H871D 2D  AIThe SynGAP1 missense variant H871D is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.480142 | Structured | 0.679301 | Binding | 0.279 | 0.858 | 0.250 | -3.263 | Likely Benign | 0.257 | Likely Benign | Likely Benign | 0.245 | Likely Benign | -0.51 | Neutral | 0.016 | Benign | 0.026 | Benign | 2.80 | Benign | 0.31 | Tolerated | 0.2271 | 0.1617 | 1 | -1 | -0.3 | -22.05 | |||||||||||||||||||||||||||||||||||||||
| c.2842G>A | G948S 2D  AIThe SynGAP1 missense variant G948S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools that assess sequence conservation and structural impact uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this benign assessment: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta, a protein‑folding stability predictor, has no available result for this variant. Overall, the collective evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988505 | Disordered | 0.862121 | Binding | 0.365 | 0.919 | 0.750 | -4.760 | Likely Benign | 0.080 | Likely Benign | Likely Benign | 0.177 | Likely Benign | 1.09 | Neutral | 0.068 | Benign | 0.026 | Benign | 4.57 | Benign | 1.00 | Tolerated | 0.2466 | 0.5502 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.2912C>T | P971L 2D  AIThe SynGAP1 missense variant P971L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.950334 | Disordered | 0.951523 | Binding | 0.545 | 0.905 | 0.625 | -4.892 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.030 | Likely Benign | -1.57 | Neutral | 0.144 | Benign | 0.026 | Benign | 3.93 | Benign | 0.00 | Affected | 0.2046 | 0.5985 | -3 | -3 | 5.4 | 16.04 | |||||||||||||||||||||||||||||||||||||||
| c.2915C>T | P972L 2D  AIThe SynGAP1 missense variant P972L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.891961 | Disordered | 0.954150 | Binding | 0.472 | 0.904 | 0.625 | -4.399 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.020 | Likely Benign | -1.73 | Neutral | 0.036 | Benign | 0.026 | Benign | 4.24 | Benign | 0.02 | Affected | 0.2107 | 0.5447 | -3 | -3 | 5.4 | 16.04 | |||||||||||||||||||||||||||||||||||||||
| c.2921A>G | D974G 2D  AIThe SynGAP1 missense variant D974G is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this conclusion does not contradict any ClinVar annotation (none present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.964377 | Binding | 0.389 | 0.897 | 0.625 | -1.638 | Likely Benign | 0.188 | Likely Benign | Likely Benign | 0.203 | Likely Benign | -0.72 | Neutral | 0.036 | Benign | 0.026 | Benign | 4.18 | Benign | 0.06 | Tolerated | 0.3379 | 0.6771 | 1 | -1 | 3.1 | -58.04 | |||||||||||||||||||||||||||||||||||||||
| c.2922C>A | D974E 2D  AIThe SynGAP1 missense variant D974E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.964377 | Binding | 0.389 | 0.897 | 0.625 | -3.617 | Likely Benign | 0.109 | Likely Benign | Likely Benign | 0.078 | Likely Benign | -0.63 | Neutral | 0.036 | Benign | 0.026 | Benign | 4.26 | Benign | 0.05 | Affected | 0.2119 | 0.7516 | 3 | 2 | 0.0 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2922C>G | D974E 2D  AIThe SynGAP1 missense variant D974E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.964377 | Binding | 0.389 | 0.897 | 0.625 | -3.617 | Likely Benign | 0.109 | Likely Benign | Likely Benign | 0.078 | Likely Benign | -0.63 | Neutral | 0.036 | Benign | 0.026 | Benign | 4.26 | Benign | 0.05 | Affected | 0.2119 | 0.7516 | 3 | 2 | 0.0 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2972G>C | G991A 2D  AIThe SynGAP1 missense variant G991A is catalogued in gnomAD (ID 6‑33443524‑G‑C) and has no ClinVar entry. All evaluated in silico predictors report a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized are unanimous in predicting benign. No tool indicates pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign,” while Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.745909 | Disordered | 0.911393 | Binding | 0.286 | 0.920 | 0.750 | 6-33443524-G-C | 1 | 6.20e-7 | -3.585 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.064 | Likely Benign | -0.83 | Neutral | 0.002 | Benign | 0.026 | Benign | 4.20 | Benign | 0.62 | Tolerated | 4.32 | 2 | 0.3522 | 0.4528 | 0 | 1 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||||||
| c.29G>C | R10P 2D  AIThe SynGAP1 missense variant R10P is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33420293‑G‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority of the four high‑accuracy tools) is benign; Foldetta results are unavailable. Overall, the collective evidence points to a benign effect for R10P, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.534167 | Disordered | 0.513657 | Binding | 0.330 | 0.915 | 0.625 | Uncertain | 2 | 6-33420293-G-C | 2 | 1.30e-6 | -3.772 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.220 | Likely Benign | -0.05 | Neutral | 0.233 | Benign | 0.026 | Benign | 4.13 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2261 | 0.5245 | 0 | -2 | 2.9 | -59.07 | ||||||||||||||||||||||||||||||||
| c.318G>C | R106S 2D  AIThe SynGAP1 missense variant R106S is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, SGM‑Consensus as benign, and the Foldetta stability analysis is unavailable. Overall, the majority of predictions lean toward a benign impact, but the high‑accuracy tools provide conflicting evidence. Thus, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.663409 | Binding | 0.345 | 0.862 | 0.875 | -2.651 | Likely Benign | 0.961 | Likely Pathogenic | Likely Pathogenic | 0.093 | Likely Benign | -1.87 | Neutral | 0.131 | Benign | 0.026 | Benign | 3.68 | Benign | 0.00 | Affected | 0.3050 | 0.4129 | 0 | -1 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||||
| c.318G>T | R106S 2D  AIThe SynGAP1 missense variant R106S is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, SGM‑Consensus as benign, and the Foldetta stability analysis is unavailable. Overall, the majority of predictions lean toward a benign impact, but the high‑accuracy tools provide conflicting evidence. Thus, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.663409 | Binding | 0.345 | 0.862 | 0.875 | -2.651 | Likely Benign | 0.961 | Likely Pathogenic | Likely Pathogenic | 0.093 | Likely Benign | -1.87 | Neutral | 0.131 | Benign | 0.026 | Benign | 3.68 | Benign | 0.00 | Affected | 0.3050 | 0.4129 | 0 | -1 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||||
| c.31G>A | G11R 2D  AIThe SynGAP1 missense variant G11R is catalogued in gnomAD (ID 6‑33420295‑G‑A) but has no ClinVar entry. Functional prediction tools cluster into two groups: benign predictions include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Pathogenic predictions come from polyPhen‑2 HumDiv and SIFT, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar status (none is reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.444081 | Structured | 0.501027 | Binding | 0.348 | 0.915 | 0.375 | 6-33420295-G-A | -3.418 | Likely Benign | 0.428 | Ambiguous | Likely Benign | 0.102 | Likely Benign | -0.47 | Neutral | 0.498 | Possibly Damaging | 0.026 | Benign | 3.92 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1022 | 0.4596 | -2 | -3 | -4.1 | 99.14 | ||||||||||||||||||||||||||||||||||||
| c.31G>C | G11R 2D  AIThe SynGAP1 missense variant G11R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. AlphaMissense‑Default is uncertain, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates a likely benign outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.444081 | Structured | 0.501027 | Binding | 0.348 | 0.915 | 0.375 | -3.418 | Likely Benign | 0.428 | Ambiguous | Likely Benign | 0.109 | Likely Benign | -0.47 | Neutral | 0.498 | Possibly Damaging | 0.026 | Benign | 3.92 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1022 | 0.4596 | -2 | -3 | -4.1 | 99.14 | |||||||||||||||||||||||||||||||||||||
| c.3217T>A | S1073T 2D  AIThe SynGAP1 missense variant S1073T is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.916840 | Disordered | 0.985818 | Binding | 0.313 | 0.905 | 0.750 | -5.203 | Likely Benign | 0.169 | Likely Benign | Likely Benign | 0.161 | Likely Benign | -0.26 | Neutral | 0.025 | Benign | 0.026 | Benign | 4.06 | Benign | 0.55 | Tolerated | 0.1696 | 0.6509 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3244C>G | Q1082E 2D  AIThe SynGAP1 missense variant Q1082E is reported in gnomAD (ID 6‑33443796‑C‑G) and has no ClinVar entry. All evaluated in‑silico predictors classify it as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are unavailable. Based on the unanimous benign predictions and lack of ClinVar pathogenic annotation, the variant is most likely benign and does not contradict ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.819762 | Disordered | 0.979325 | Binding | 0.347 | 0.896 | 0.875 | 6-33443796-C-G | -3.437 | Likely Benign | 0.267 | Likely Benign | Likely Benign | 0.090 | Likely Benign | -0.87 | Neutral | 0.112 | Benign | 0.026 | Benign | 4.19 | Benign | 0.07 | Tolerated | 3.77 | 5 | 0.1485 | 0.3227 | 2 | 2 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||||||||
| c.3296A>C | Y1099S 2D  AIThe SynGAP1 missense variant Y1099S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this trend: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.974267 | Binding | 0.400 | 0.862 | 1.000 | -1.775 | Likely Benign | 0.139 | Likely Benign | Likely Benign | 0.156 | Likely Benign | -0.23 | Neutral | 0.149 | Benign | 0.026 | Benign | 2.90 | Benign | 0.62 | Tolerated | 0.4564 | 0.2298 | -3 | -2 | 0.5 | -76.10 | |||||||||||||||||||||||||||||||||||||||
| c.331C>T | P111S 2D  AIThe SynGAP1 missense variant P111S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods also support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta results are unavailable. Overall, the collective predictions strongly suggest that P111S is most likely benign, and this conclusion is not contradicted by any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.650020 | Binding | 0.438 | 0.858 | 0.750 | -2.773 | Likely Benign | 0.307 | Likely Benign | Likely Benign | 0.033 | Likely Benign | -1.03 | Neutral | 0.131 | Benign | 0.026 | Benign | 4.21 | Benign | 0.29 | Tolerated | 0.3515 | 0.4973 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.3328A>G | S1110G 2D  AIThe SynGAP1 missense variant S1110G is listed in ClinVar (ID 1722210.0) as benign and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion aligns with the ClinVar benign classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.912647 | Disordered | 0.934156 | Binding | 0.346 | 0.892 | 0.875 | Likely Benign | 1 | -4.674 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.035 | Likely Benign | -2.26 | Neutral | 0.036 | Benign | 0.026 | Benign | 2.19 | Pathogenic | 0.08 | Tolerated | 4.32 | 2 | 0.2559 | 0.4806 | 1 | 0 | 0.4 | -30.03 | |||||||||||||||||||||||||||||||||||
| c.3349G>A | G1117S 2D  AIThe SynGAP1 missense variant G1117S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.882776 | Disordered | 0.853192 | Binding | 0.323 | 0.914 | 0.750 | -3.890 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.211 | Likely Benign | -0.37 | Neutral | 0.032 | Benign | 0.026 | Benign | 5.08 | Benign | 0.17 | Tolerated | 0.2572 | 0.5111 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.3362G>C | S1121T 2D  AIThe SynGAP1 missense variant S1121T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is provided). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.894241 | Disordered | 0.810024 | Binding | 0.365 | 0.935 | 0.875 | -6.442 | Likely Benign | 0.069 | Likely Benign | Likely Benign | 0.105 | Likely Benign | -0.23 | Neutral | 0.011 | Benign | 0.026 | Benign | 5.45 | Benign | 0.00 | Affected | 0.2108 | 0.5582 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3400A>G | T1134A 2D  AIThe SynGAP1 missense variant T1134A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.813034 | Binding | 0.335 | 0.885 | 0.875 | -3.912 | Likely Benign | 0.090 | Likely Benign | Likely Benign | 0.172 | Likely Benign | -0.71 | Neutral | 0.036 | Benign | 0.026 | Benign | 5.70 | Benign | 0.31 | Tolerated | 0.3756 | 0.4424 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||||||||||||
| c.3406C>G | Q1136E 2D  AIThe SynGAP1 missense variant Q1136E is catalogued in gnomAD (ID 6‑33443958‑C‑G) and has no ClinVar entry. All evaluated in silico predictors uniformly classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic or likely pathogenic outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. Foldetta, a protein‑folding stability method, did not provide a result for this variant. Overall, the computational evidence strongly supports a benign classification, and this is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.903857 | Disordered | 0.775584 | Binding | 0.321 | 0.884 | 0.875 | 6-33443958-C-G | 4 | 2.71e-6 | -6.677 | Likely Benign | 0.277 | Likely Benign | Likely Benign | 0.209 | Likely Benign | -1.13 | Neutral | 0.022 | Benign | 0.026 | Benign | 5.46 | Benign | 0.11 | Tolerated | 4.32 | 2 | 0.1487 | 0.2886 | 2 | 2 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||||||
| c.3407A>T | Q1136L 2D  AIThe SynGAP1 missense variant Q1136L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate a benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this is consistent with the lack of ClinVar evidence; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.903857 | Disordered | 0.775584 | Binding | 0.321 | 0.884 | 0.875 | -6.020 | Likely Benign | 0.203 | Likely Benign | Likely Benign | 0.242 | Likely Benign | -2.42 | Neutral | 0.005 | Benign | 0.026 | Benign | 5.44 | Benign | 0.23 | Tolerated | 0.0861 | 0.6395 | -2 | -2 | 7.3 | -14.97 | |||||||||||||||||||||||||||||||||||||||
| c.3442A>G | M1148V 2D  AIThe SynGAP1 missense variant M1148V is catalogued in ClinVar with no submitted interpretation and is present in gnomAD (ID 6‑33444477‑A‑G). Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it to be pathogenic. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of available predictions indicates that M1148V is most likely benign, and this conclusion does not contradict any ClinVar status, as no pathogenic claim has been reported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.774279 | Binding | 0.343 | 0.835 | 0.875 | 6-33444477-A-G | 3 | 1.86e-6 | -2.358 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.057 | Likely Benign | -1.47 | Neutral | 0.016 | Benign | 0.026 | Benign | 2.59 | Benign | 0.00 | Affected | 4.32 | 2 | 0.3154 | 0.3371 | 1 | 2 | 2.3 | -32.06 | ||||||||||||||||||||||||||||||||||
| c.3530A>G | E1177G 2D  AIThe SynGAP1 missense variant E1177G is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact, and this is consistent with the lack of ClinVar annotation—there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.541878 | Disordered | 0.566503 | Binding | 0.542 | 0.705 | 0.250 | -3.948 | Likely Benign | 0.727 | Likely Pathogenic | Likely Benign | 0.389 | Likely Benign | -2.04 | Neutral | 0.012 | Benign | 0.026 | Benign | 5.45 | Benign | 0.02 | Affected | 0.2690 | 0.4094 | 0 | -2 | 3.1 | -72.06 | ||||||||||||||||||||||||||||||||||||||
| c.3531G>C | E1177D 2D  AIThe SynGAP1 missense variant E1177D is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it to be pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign outcome. High‑accuracy assessments further support this view: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta stability analysis is unavailable. Taken together, the majority of evidence indicates that E1177D is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.541878 | Disordered | 0.566503 | Binding | 0.542 | 0.705 | 0.250 | -4.195 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.210 | Likely Benign | -0.74 | Neutral | 0.029 | Benign | 0.026 | Benign | 5.42 | Benign | 0.05 | Affected | 0.1477 | 0.2735 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||||||||
| c.3531G>T | E1177D 2D  AIThe SynGAP1 missense variant E1177D is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of prediction tools and the high‑accuracy methods points to a benign impact. This conclusion is consistent with the lack of ClinVar evidence, so there is no contradiction with existing clinical annotations. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.541878 | Disordered | 0.566503 | Binding | 0.542 | 0.705 | 0.250 | -4.195 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.210 | Likely Benign | -0.74 | Neutral | 0.029 | Benign | 0.026 | Benign | 5.42 | Benign | 0.05 | Affected | 0.1477 | 0.2735 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||||||||
| c.361G>A | A121T 2D  AIThe SynGAP1 missense variant A121T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The prediction is consistent with the lack of ClinVar annotation, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.779859 | Disordered | 0.661304 | Binding | 0.362 | 0.888 | 0.750 | -3.955 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.059 | Likely Benign | -0.25 | Neutral | 0.063 | Benign | 0.026 | Benign | 4.10 | Benign | 0.05 | Affected | 0.1792 | 0.7305 | 1 | 0 | -2.5 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.362C>G | A121G 2D  AIThe SynGAP1 missense variant A121G is catalogued in gnomAD (ID 6‑33432227‑C‑G) but has no ClinVar entry. Across the spectrum of in‑silico predictors, every tool examined—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently classifies the substitution as benign. No pathogenic predictions are reported. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status remains unavailable. Overall, the evidence strongly supports a benign effect for A121G, and this conclusion does not contradict any ClinVar classification (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.779859 | Disordered | 0.661304 | Binding | 0.362 | 0.888 | 0.750 | 6-33432227-C-G | 4 | 2.48e-6 | -3.123 | Likely Benign | 0.099 | Likely Benign | Likely Benign | 0.073 | Likely Benign | -0.52 | Neutral | 0.118 | Benign | 0.026 | Benign | 4.06 | Benign | 0.07 | Tolerated | 3.61 | 5 | 0.2407 | 0.5271 | 0 | 1 | -2.2 | -14.03 | ||||||||||||||||||||||||||||||||||
| c.3832C>A | P1278T 2D  AIThe SynGAP1 missense variant P1278T is catalogued in gnomAD (ID 6‑33447880‑C‑A) but has no ClinVar entry. Across a broad panel of in silico predictors, every tool reports a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool in the set predicts pathogenicity, so the pathogenic group is empty. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized returns a benign prediction, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status remains unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, and this assessment does not contradict any ClinVar classification. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.788093 | Disordered | 0.806955 | Binding | 0.532 | 0.722 | 0.750 | 6-33447880-C-A | -5.505 | Likely Benign | 0.095 | Likely Benign | Likely Benign | 0.064 | Likely Benign | -0.60 | Neutral | 0.059 | Benign | 0.026 | Benign | 2.73 | Benign | 0.11 | Tolerated | 3.77 | 5 | 0.1825 | 0.4053 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||||
| c.3842C>T | A1281V 2D  AIThe SynGAP1 missense variant A1281V is reported in gnomAD (ID 6‑33447890‑C‑T) and has no ClinVar entry. Across the spectrum of in silico predictors, every tool examined—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently classifies the substitution as benign. No algorithm in the dataset returned a pathogenic or likely pathogenic label, so the pathogenic group is empty. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign status. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its influence is unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.821556 | Binding | 0.434 | 0.721 | 0.875 | 6-33447890-C-T | -4.021 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.037 | Likely Benign | -0.79 | Neutral | 0.057 | Benign | 0.026 | Benign | 2.65 | Benign | 0.15 | Tolerated | 4.32 | 4 | 0.1151 | 0.4578 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||||
| c.3844G>A | E1282K 2D  AIThe SynGAP1 missense variant E1282K is catalogued in gnomAD (ID 6‑33447892‑G‑A) but has no ClinVar entry. Across the spectrum of in‑silico predictors, every tool examined—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently classifies the substitution as benign. No pathogenic predictions are reported. Grouping by consensus, all listed tools fall into the benign category, with no tools indicating pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status remains unavailable. Overall, the computational evidence overwhelmingly supports a benign effect for E1282K, and this conclusion does not contradict any ClinVar classification (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.817364 | Binding | 0.465 | 0.725 | 0.875 | 6-33447892-G-A | -3.805 | Likely Benign | 0.226 | Likely Benign | Likely Benign | 0.173 | Likely Benign | -1.17 | Neutral | 0.126 | Benign | 0.026 | Benign | 2.73 | Benign | 0.16 | Tolerated | 3.77 | 5 | 0.1821 | 0.5809 | 1 | 0 | -0.4 | -0.94 | ||||||||||||||||||||||||||||||||||||
| c.3913A>T | T1305S 2D  AIThe SynGAP1 missense variant T1305S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.791621 | Disordered | 0.894658 | Binding | 0.390 | 0.873 | 0.875 | -2.813 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.053 | Likely Benign | 0.20 | Neutral | 0.003 | Benign | 0.026 | Benign | 2.83 | Benign | 0.08 | Tolerated | 0.3683 | 0.4988 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.391G>A | G131S 2D  AIThe SynGAP1 missense variant G131S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.429200 | Structured | 0.724779 | Binding | 0.302 | 0.891 | 0.250 | -2.953 | Likely Benign | 0.337 | Likely Benign | Likely Benign | 0.039 | Likely Benign | -2.75 | Deleterious | 0.115 | Benign | 0.026 | Benign | 4.10 | Benign | 0.02 | Affected | 0.2823 | 0.5338 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.4019C>A | T1340N 2D  AIThe SynGAP1 missense variant T1340N is reported in gnomAD (ID 6‑33451893‑C‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it as pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence points to a benign impact for T1340N. This conclusion is consistent with the absence of a ClinVar pathogenic classification, so there is no contradiction with existing clinical annotations. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.852992 | Disordered | 0.977899 | Binding | 0.444 | 0.697 | 0.750 | 6-33451893-C-A | -3.664 | Likely Benign | 0.154 | Likely Benign | Likely Benign | 0.071 | Likely Benign | -0.95 | Neutral | 0.092 | Benign | 0.026 | Benign | 4.09 | Benign | 0.02 | Affected | 3.77 | 5 | 0.1755 | 0.5309 | 0 | 0 | -2.8 | 13.00 | ||||||||||||||||||||||||||||||||||||
| c.512C>G | A171G 2D  AIThe SynGAP1 missense variant A171G is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta results are not available for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.562014 | Disordered | 0.492272 | Uncertain | 0.358 | 0.652 | 0.375 | -4.019 | Likely Benign | 0.301 | Likely Benign | Likely Benign | 0.050 | Likely Benign | -1.08 | Neutral | 0.063 | Benign | 0.026 | Benign | 4.14 | Benign | 0.05 | Affected | 0.1767 | 0.3278 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.512C>T | A171V 2D  AIThe SynGAP1 missense variant A171V is catalogued in gnomAD (ID 6‑33435154‑C‑T) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence points to a benign impact for A171V, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.562014 | Disordered | 0.492272 | Uncertain | 0.358 | 0.652 | 0.375 | 6-33435154-C-T | 1 | 6.20e-7 | -6.437 | Likely Benign | 0.434 | Ambiguous | Likely Benign | 0.052 | Likely Benign | -1.65 | Neutral | 0.118 | Benign | 0.026 | Benign | 4.15 | Benign | 0.03 | Affected | 3.74 | 4 | 0.0766 | 0.4897 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||
| c.95C>T | T32I 2D  AIThe SynGAP1 missense variant T32I is reported in gnomAD (ID 6‑33423504‑C‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus also reports likely benign; Foldetta results are not available. Overall, the preponderance of evidence points to a benign impact for T32I, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.437154 | Uncertain | 0.349 | 0.879 | 0.375 | 6-33423504-C-T | 1 | 6.20e-7 | -3.689 | Likely Benign | 0.213 | Likely Benign | Likely Benign | 0.024 | Likely Benign | -0.57 | Neutral | 0.049 | Benign | 0.026 | Benign | 4.26 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1081 | 0.6403 | -1 | 0 | 5.2 | 12.05 | ||||||||||||||||||||||||||||||||||
| c.1183G>A | G395R 2D  AIThe SynGAP1 missense variant G395R has no ClinVar record and is not reported in gnomAD. Prediction tools cluster into two groups: benign predictions come from REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized; pathogenic predictions come from SIFT, ESM1b, and AlphaMissense‑Default. Two tools, FoldX and Foldetta, yield uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta remains uncertain. Overall, the balance of evidence favors a benign effect, and this conclusion does not contradict any ClinVar annotation (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.513880 | Disordered | 0.396199 | Uncertain | 0.474 | 0.601 | 0.500 | -9.851 | Likely Pathogenic | 0.768 | Likely Pathogenic | Likely Benign | 1.26 | Ambiguous | 2.1 | -0.25 | Likely Benign | 0.51 | Ambiguous | 0.50 | Likely Benign | 0.430 | Likely Benign | -2.01 | Neutral | 0.037 | Benign | 0.027 | Benign | 4.23 | Benign | 0.02 | Affected | 0.0953 | 0.4313 | -3 | -2 | -4.1 | 99.14 | ||||||||||||||||||||||||||||||
| c.1183G>C | G395R 2D  AIThe SynGAP1 missense variant G395R has no ClinVar record and is not reported in gnomAD. Prediction tools cluster into two groups: benign predictions come from REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized; pathogenic predictions come from SIFT, ESM1b, and AlphaMissense‑Default. Two tools, FoldX and Foldetta, yield uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta remains uncertain. Overall, the balance of evidence favors a benign effect, and this conclusion does not contradict any ClinVar annotation (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.513880 | Disordered | 0.396199 | Uncertain | 0.474 | 0.601 | 0.500 | -9.851 | Likely Pathogenic | 0.768 | Likely Pathogenic | Likely Benign | 1.26 | Ambiguous | 2.1 | -0.25 | Likely Benign | 0.51 | Ambiguous | 0.50 | Likely Benign | 0.430 | Likely Benign | -2.01 | Neutral | 0.037 | Benign | 0.027 | Benign | 4.23 | Benign | 0.02 | Affected | 0.0953 | 0.4313 | -3 | -2 | -4.1 | 99.14 | ||||||||||||||||||||||||||||||
| c.2186A>G | N729S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N729S is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Across the available in‑silico predictors, the majority (REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized) classify the change as benign, while Rosetta and Foldetta return uncertain results. No tool predicts pathogenicity. High‑accuracy assessments reinforce this benign trend: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign,” and Foldetta’s stability prediction is inconclusive. Overall, the computational evidence strongly supports a benign effect, and this does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.750527 | Disordered | 0.426547 | Uncertain | 0.651 | 0.583 | 0.625 | Conflicting | 2 | -1.578 | Likely Benign | 0.066 | Likely Benign | Likely Benign | 0.14 | Likely Benign | 0.1 | 1.34 | Ambiguous | 0.74 | Ambiguous | -0.36 | Likely Benign | 0.063 | Likely Benign | -0.42 | Neutral | 0.221 | Benign | 0.027 | Benign | 3.38 | Benign | 0.93 | Tolerated | 3.59 | 7 | 0.3411 | 0.4854 | 1 | 1 | 2.7 | -27.03 | ||||||||||||||||||||||||||
| c.238A>G | K80E 2D  AIThe SynGAP1 K80E missense variant has no ClinVar record and is not listed in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The high‑accuracy consensus (SGM‑Consensus) is derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yielding a “Likely Benign” classification. AlphaMissense‑Optimized returns an uncertain result, and Foldetta (which would combine FoldX‑MD and Rosetta outputs) has no available data for this variant. Overall, the balance of evidence favors a benign impact, and this assessment does not contradict any ClinVar status because no ClinVar entry exists for K80E. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.477530 | Uncertain | 0.331 | 0.873 | 0.500 | -5.605 | Likely Benign | 0.943 | Likely Pathogenic | Ambiguous | 0.034 | Likely Benign | -0.91 | Neutral | 0.225 | Benign | 0.027 | Benign | 3.92 | Benign | 0.00 | Affected | 0.3850 | 0.0952 | 0 | 1 | 0.4 | 0.94 | |||||||||||||||||||||||||||||||||||||||
| c.262G>C | V88L 2D  AIThe SynGAP1 V88L variant has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Those that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized classifying the variant as pathogenic, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome; Foldetta results are unavailable. Overall, the majority of tools suggest a benign impact, but the high‑accuracy predictions are conflicting. The variant is most likely benign based on the collective evidence, and this assessment does not contradict any ClinVar status because no ClinVar classification exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.552910 | Binding | 0.323 | 0.870 | 0.500 | -5.808 | Likely Benign | 0.975 | Likely Pathogenic | Likely Pathogenic | 0.066 | Likely Benign | -0.79 | Neutral | 0.225 | Benign | 0.027 | Benign | 3.74 | Benign | 0.00 | Affected | 0.1457 | 0.4562 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.262G>T | V88L 2D  AIThe SynGAP1 missense variant V88L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM, whereas SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized classifying the variant as pathogenic, while the SGM Consensus (a majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect; a Foldetta stability analysis is unavailable. Overall, the majority of predictions lean toward a benign impact, and there is no ClinVar annotation to contradict this assessment. Thus, the variant is most likely benign based on the current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.552910 | Binding | 0.323 | 0.870 | 0.500 | -5.808 | Likely Benign | 0.975 | Likely Pathogenic | Likely Pathogenic | 0.066 | Likely Benign | -0.79 | Neutral | 0.225 | Benign | 0.027 | Benign | 3.74 | Benign | 0.00 | Affected | 0.1457 | 0.4562 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3093G>A | M1031I 2D  AIThe SynGAP1 missense variant M1031I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Taken together, the overwhelming majority of evidence points to a benign impact for M1031I. This conclusion is consistent with the lack of ClinVar annotation and does not contradict any existing clinical database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.865454 | Disordered | 0.995959 | Binding | 0.340 | 0.736 | 0.500 | -3.312 | Likely Benign | 0.739 | Likely Pathogenic | Likely Benign | 0.026 | Likely Benign | -0.99 | Neutral | 0.095 | Benign | 0.027 | Benign | 2.71 | Benign | 0.25 | Tolerated | 0.1128 | 0.3324 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3093G>C | M1031I 2D  AIThe SynGAP1 missense variant M1031I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Taken together, the overwhelming majority of evidence points to a benign impact for M1031I. This conclusion is consistent with the lack of ClinVar annotation and does not contradict any existing clinical database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.865454 | Disordered | 0.995959 | Binding | 0.340 | 0.736 | 0.500 | -3.312 | Likely Benign | 0.739 | Likely Pathogenic | Likely Benign | 0.025 | Likely Benign | -0.99 | Neutral | 0.095 | Benign | 0.027 | Benign | 2.71 | Benign | 0.25 | Tolerated | 0.1128 | 0.3324 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3093G>T | M1031I 2D  AIThe SynGAP1 missense variant M1031I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Taken together, the overwhelming majority of evidence points to a benign impact for M1031I. This conclusion is consistent with the lack of ClinVar annotation and does not contradict any existing clinical database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.865454 | Disordered | 0.995959 | Binding | 0.340 | 0.736 | 0.500 | -3.312 | Likely Benign | 0.739 | Likely Pathogenic | Likely Benign | 0.026 | Likely Benign | -0.99 | Neutral | 0.095 | Benign | 0.027 | Benign | 2.71 | Benign | 0.25 | Tolerated | 0.1128 | 0.3324 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3769T>A | S1257T 2D  AIThe SynGAP1 missense variant S1257T is predicted to be benign by all available in‑silico tools. Consensus predictors such as REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No tool predicts pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. ClinVar contains no entry for this variant, and it is not listed in gnomAD. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.541878 | Disordered | 0.482380 | Uncertain | 0.889 | 0.572 | 0.375 | -5.034 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.113 | Likely Benign | -1.01 | Neutral | 0.051 | Benign | 0.027 | Benign | 2.61 | Benign | 0.25 | Tolerated | 0.1034 | 0.4767 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||||||||||
| c.3770C>G | S1257C 2D  AIThe SynGAP1 missense variant S1257C is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the SGM‑Consensus score indicates a likely benign outcome. Only SIFT predicts a pathogenic effect, and ESM1b remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized reports benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also yields a likely benign verdict. Foldetta data are not available for this variant. Overall, the preponderance of evidence points to a benign impact, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.541878 | Disordered | 0.482380 | Uncertain | 0.889 | 0.572 | 0.375 | -7.741 | In-Between | 0.125 | Likely Benign | Likely Benign | 0.121 | Likely Benign | -2.15 | Neutral | 0.028 | Benign | 0.027 | Benign | 2.54 | Benign | 0.05 | Affected | 0.0761 | 0.4780 | 0 | -1 | 3.3 | 16.06 | ||||||||||||||||||||||||||||||||||||||
| c.55G>T | A19S 2D  AIThe SynGAP1 missense variant A19S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.529623 | Disordered | 0.443393 | Uncertain | 0.378 | 0.906 | 0.500 | -2.905 | Likely Benign | 0.107 | Likely Benign | Likely Benign | 0.029 | Likely Benign | -0.21 | Neutral | 0.225 | Benign | 0.027 | Benign | 4.17 | Benign | 0.00 | Affected | 0.3273 | 0.6260 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.1169G>C | G390A 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant G390A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, FATHMM, and Rosetta. The high‑accuracy assessment shows AlphaMissense‑Optimized as benign, the SGM Consensus (derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) as likely benign, and Foldetta (combining FoldX‑MD and Rosetta stability outputs) as pathogenic. FoldX alone is inconclusive. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation because no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.626927 | Disordered | 0.413274 | Uncertain | 0.304 | 0.763 | 0.875 | -6.768 | Likely Benign | 0.100 | Likely Benign | Likely Benign | 1.96 | Ambiguous | 0.4 | 2.03 | Destabilizing | 2.00 | Destabilizing | 0.02 | Likely Benign | 0.402 | Likely Benign | -0.55 | Neutral | 0.143 | Benign | 0.028 | Benign | 1.33 | Pathogenic | 0.05 | Affected | 0.4130 | 0.5053 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||
| c.1321G>C | V441L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V441L is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome, while Rosetta and AlphaMissense‑Default are uncertain. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. No prediction or folding result is missing or inconclusive. Overall, the variant is most likely benign based on the preponderance of evidence, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.161087 | Structured | 0.259875 | Uncertain | 0.918 | 0.249 | 0.000 | -9.546 | Likely Pathogenic | 0.395 | Ambiguous | Likely Benign | -0.43 | Likely Benign | 0.0 | 0.59 | Ambiguous | 0.08 | Likely Benign | 0.45 | Likely Benign | 0.135 | Likely Benign | -2.27 | Neutral | 0.165 | Benign | 0.028 | Benign | 3.43 | Benign | 0.11 | Tolerated | 0.0961 | 0.3881 | 2 | 1 | -0.4 | 14.03 | ||||||||||||||||||||||||||||||
| c.2116G>A | E706K 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant E706K is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM all classify the change as benign. In contrast, ESM1b and AlphaMissense‑Default predict a pathogenic impact. Tools that return uncertain results—FoldX, Rosetta, Foldetta, and AlphaMissense‑Optimized—do not provide decisive evidence. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive (two pathogenic versus two benign calls). High‑accuracy assessments are likewise ambiguous: AlphaMissense‑Optimized is uncertain, Foldetta is uncertain, and the SGM Consensus remains inconclusive. Overall, the preponderance of evidence points to a benign effect, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.200174 | Structured | 0.377033 | Uncertain | 0.929 | 0.363 | 0.000 | Uncertain | 1 | -10.519 | Likely Pathogenic | 0.833 | Likely Pathogenic | Ambiguous | 1.17 | Ambiguous | 0.1 | 0.51 | Ambiguous | 0.84 | Ambiguous | 0.08 | Likely Benign | 0.080 | Likely Benign | -1.51 | Neutral | 0.345 | Benign | 0.028 | Benign | 4.15 | Benign | 0.52 | Tolerated | 3.47 | 10 | 0.2065 | 0.4511 | 0 | 1 | -0.4 | -0.94 | 187.1 | 49.2 | 0.0 | 0.0 | 0.4 | 0.1 | X | Uncertain | The carboxylate side chain of Glu706, located at the end and outer surface of an α-helix (res. Thr704-Gly712), forms a salt bridge with Lys710 and a hydrogen bond with its own backbone amino group at the helix end in the WT simulations. Although Lys706 is unable to make these transient interactions in the variant simulations, there is no apparent negative effect on the protein structure due to the residue swap. However, because the model ends abruptly at the C-terminus, no definite conclusions can be drawn based on the simulations. | |||||||||||||||||
| c.2165G>A | S722N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S722N is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that reach consensus classify the change as benign: REVEL, FoldX, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized all predict a neutral effect. No tool predicts pathogenicity; the only inconclusive results come from premPS and AlphaMissense‑Default, which are treated as unavailable evidence. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized reports a benign outcome, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) also predicts a benign impact. Taken together, the evidence overwhelmingly supports a benign classification, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.468512 | Structured | 0.457186 | Uncertain | 0.950 | 0.431 | 0.375 | -4.399 | Likely Benign | 0.379 | Ambiguous | Likely Benign | 0.18 | Likely Benign | 0.1 | 0.15 | Likely Benign | 0.17 | Likely Benign | 0.79 | Ambiguous | 0.051 | Likely Benign | -0.91 | Neutral | 0.072 | Benign | 0.028 | Benign | 2.58 | Benign | 0.27 | Tolerated | 0.1258 | 0.3452 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||||
| c.232C>G | R78G 2D  AIThe SynGAP1 R78G missense variant has no ClinVar entry and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments further support a benign status: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Taken together, the preponderance of evidence from both general and high‑accuracy predictors indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.505461 | Disordered | 0.448183 | Uncertain | 0.304 | 0.866 | 0.500 | -3.972 | Likely Benign | 0.480 | Ambiguous | Likely Benign | 0.072 | Likely Benign | -1.94 | Neutral | 0.385 | Benign | 0.028 | Benign | 3.83 | Benign | 0.00 | Affected | 0.3285 | 0.2986 | -3 | -2 | 4.1 | -99.14 | |||||||||||||||||||||||||||||||||||||||
| c.2918G>C | G973A 2D  AIThe SynGAP1 missense variant G973A is reported in gnomAD (ID 6‑33443470‑G‑C) but has no ClinVar entry. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. The high‑accuracy consensus (SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and this assessment is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.887230 | Disordered | 0.959498 | Binding | 0.390 | 0.897 | 0.625 | 6-33443470-G-C | 1 | 6.20e-7 | -3.847 | Likely Benign | 0.075 | Likely Benign | Likely Benign | 0.091 | Likely Benign | 0.14 | Neutral | 0.112 | Benign | 0.028 | Benign | 4.30 | Benign | 0.62 | Tolerated | 4.32 | 2 | 0.3405 | 0.5133 | 0 | 1 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||||||
| c.2957A>T | E986V 2D  AIThe SynGAP1 E986V missense variant is not reported in ClinVar and has no gnomAD entry. Functional prediction tools cluster into two groups: benign predictions come from REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and ESM1b, whereas pathogenic predictions arise from PROVEAN, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments reinforce the pathogenic signal: AlphaMissense‑Optimized predicts pathogenic, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default (pathogenic), ESM1b (benign), FATHMM (pathogenic), and PROVEAN (pathogenic)—also indicates pathogenic. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of the majority of prediction tools and the high‑accuracy methods points to a pathogenic effect for E986V, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.750527 | Disordered | 0.929726 | Binding | 0.349 | 0.902 | 0.750 | -4.511 | Likely Benign | 0.965 | Likely Pathogenic | Likely Pathogenic | 0.220 | Likely Benign | -3.48 | Deleterious | 0.018 | Benign | 0.028 | Benign | 2.10 | Pathogenic | 0.00 | Affected | 0.1146 | 0.7960 | -2 | -2 | 7.7 | -29.98 | |||||||||||||||||||||||||||||||||||||||
| c.3048C>A | D1016E 2D  AIThe SynGAP1 missense variant D1016E is reported in ClinVar (ID 3803472.0) as benign and is present in gnomAD (variant ID 6‑33443600‑C‑A). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, aligning with the ClinVar benign classification and showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.801317 | Disordered | 0.944705 | Binding | 0.323 | 0.811 | 0.625 | Likely Benign | 1 | 6-33443600-C-A | 2 | 1.24e-6 | -3.422 | Likely Benign | 0.216 | Likely Benign | Likely Benign | 0.017 | Likely Benign | -0.37 | Neutral | 0.008 | Benign | 0.028 | Benign | 2.64 | Benign | 0.65 | Tolerated | 3.77 | 5 | 0.2237 | 0.6898 | 2 | 3 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||
| c.3048C>G | D1016E 2D  AIThe SynGAP1 missense variant D1016E is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign. Foldetta results are unavailable, so they do not influence the overall assessment. **Thus, the variant is most likely benign, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists.** Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.801317 | Disordered | 0.944705 | Binding | 0.323 | 0.811 | 0.625 | -3.422 | Likely Benign | 0.216 | Likely Benign | Likely Benign | 0.017 | Likely Benign | -0.37 | Neutral | 0.008 | Benign | 0.028 | Benign | 2.64 | Benign | 0.65 | Tolerated | 3.77 | 5 | 0.2237 | 0.6898 | 2 | 3 | 0.0 | 14.03 | |||||||||||||||||||||||||||||||||||||
| c.3185G>C | G1062A 2D  AIThe SynGAP1 missense variant G1062A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this conclusion does not contradict any ClinVar annotation (none present). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.976962 | Disordered | 0.936972 | Binding | 0.368 | 0.917 | 0.875 | -6.124 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.350 | Likely Benign | 0.20 | Neutral | 0.059 | Benign | 0.028 | Benign | 4.20 | Benign | 0.51 | Tolerated | 0.3305 | 0.5144 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3305C>A | A1102D 2D  AIThe SynGAP1 missense variant A1102D is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also favors benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which currently contains no entry for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.915074 | Disordered | 0.962659 | Binding | 0.388 | 0.859 | 0.875 | -4.647 | Likely Benign | 0.388 | Ambiguous | Likely Benign | 0.081 | Likely Benign | -1.38 | Neutral | 0.033 | Benign | 0.028 | Benign | 2.27 | Pathogenic | 0.12 | Tolerated | 0.2045 | 0.2984 | 0 | -2 | -5.3 | 44.01 | ||||||||||||||||||||||||||||||||||||||||
| c.3305C>T | A1102V 2D  AIThe SynGAP1 missense variant A1102V is listed in ClinVar (ID 2846719.0) as Benign and is present in gnomAD (variant ID 6‑33443857‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote). Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion aligns with the ClinVar benign classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.915074 | Disordered | 0.962659 | Binding | 0.388 | 0.859 | 0.875 | Benign | 1 | 6-33443857-C-T | -2.440 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.081 | Likely Benign | -1.27 | Neutral | 0.017 | Benign | 0.028 | Benign | 2.29 | Pathogenic | 0.12 | Tolerated | 3.77 | 5 | 0.1333 | 0.6264 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||
| c.3579T>A | D1193E 2D  AIThe SynGAP1 missense variant D1193E is predicted to be benign by the majority of in silico tools. Benign predictions come from REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT classifies the change as pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign outcome. High‑accuracy assessments further support a benign effect: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. ClinVar contains no entry for this variant, and it is absent from gnomAD, so there is no external evidence to contradict the computational predictions. Therefore, the variant is most likely benign, with no conflict with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.585406 | Disordered | 0.433390 | Uncertain | 0.807 | 0.600 | 0.375 | -3.788 | Likely Benign | 0.237 | Likely Benign | Likely Benign | 0.307 | Likely Benign | -1.09 | Neutral | 0.008 | Benign | 0.028 | Benign | 5.47 | Benign | 0.00 | Affected | 0.1242 | 0.3957 | 3 | 2 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||||||||
| c.3579T>G | D1193E 2D  AIThe SynGAP1 missense variant D1193E is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the only pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates a benign outcome. Foldetta results are not available, so they do not influence the assessment. Overall, the preponderance of evidence supports a benign classification for D1193E, and this conclusion is consistent with the lack of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.585406 | Disordered | 0.433390 | Uncertain | 0.807 | 0.600 | 0.375 | -3.788 | Likely Benign | 0.237 | Likely Benign | Likely Benign | 0.307 | Likely Benign | -1.09 | Neutral | 0.008 | Benign | 0.028 | Benign | 5.47 | Benign | 0.00 | Affected | 0.1242 | 0.3957 | 3 | 2 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||||||||
| c.3611C>T | S1204L 2D  AIThe SynGAP1 missense variant S1204L is reported in gnomAD (variant ID 6‑33446603‑C‑T) and has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign; Foldetta results are not available. Based on the collective predictions, the variant is most likely benign, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.595080 | Disordered | 0.541098 | Binding | 0.887 | 0.587 | 0.375 | 6-33446603-C-T | 1 | 6.20e-7 | -4.672 | Likely Benign | 0.325 | Likely Benign | Likely Benign | 0.375 | Likely Benign | -0.86 | Neutral | 0.035 | Benign | 0.028 | Benign | 5.35 | Benign | 0.32 | Tolerated | 3.77 | 5 | 0.1000 | 0.4082 | -2 | -3 | 4.6 | 26.08 | |||||||||||||||||||||||||||||||||
| c.3637A>C | N1213H 2D  AIThe SynGAP1 missense variant N1213H is predicted to be benign by the majority of in‑silico tools. Benign predictions come from REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT classifies the change as pathogenic, while the consensus score from the SGM framework (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized reports benign, and the SGM Consensus also reports likely benign. Foldetta, a protein‑folding stability predictor, has no available result for this variant. The variant is not listed in ClinVar and has no entry in gnomAD, so there is no external evidence to contradict the computational predictions. Based on the collective predictions, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.580690 | Disordered | 0.521638 | Binding | 0.888 | 0.561 | 0.500 | -5.358 | Likely Benign | 0.147 | Likely Benign | Likely Benign | 0.036 | Likely Benign | -1.23 | Neutral | 0.015 | Benign | 0.028 | Benign | 2.69 | Benign | 0.02 | Affected | 0.0883 | 0.4516 | 2 | 1 | 0.3 | 23.04 | ||||||||||||||||||||||||||||||||||||||
| c.3902C>T | P1301L 2D  AIThe SynGAP1 missense variant P1301L is listed in ClinVar (ID 4749342) with an uncertain significance annotation and is present in gnomAD (variant ID 6‑33451776‑C‑T). Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign effects. No tool in the dataset predicts pathogenicity. High‑accuracy assessments corroborate this benign profile: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. A protein‑folding stability analysis with Foldetta is unavailable, so it does not influence the overall assessment. Overall, the computational evidence strongly supports a benign classification, which is consistent with the ClinVar uncertain status and does not contradict it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.728858 | Disordered | 0.885064 | Binding | 0.447 | 0.841 | 0.875 | Uncertain | 1 | 6-33451776-C-T | 3 | 1.86e-6 | -4.152 | Likely Benign | 0.099 | Likely Benign | Likely Benign | 0.072 | Likely Benign | -1.63 | Neutral | 0.017 | Benign | 0.028 | Benign | 2.83 | Benign | 0.11 | Tolerated | 3.77 | 5 | 0.1886 | 0.4655 | -3 | -3 | 5.4 | 16.04 | ||||||||||||||||||||||||||||||||
| c.3937C>G | P1313A 2D  AIThe SynGAP1 missense variant P1313A is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this benign classification: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions strongly suggests that P1313A is most likely benign, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.862302 | Disordered | 0.970301 | Binding | 0.452 | 0.902 | 0.750 | -3.855 | Likely Benign | 0.054 | Likely Benign | Likely Benign | 0.069 | Likely Benign | -0.37 | Neutral | 0.021 | Benign | 0.028 | Benign | 4.27 | Benign | 0.25 | Tolerated | 0.3529 | 0.5842 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||||||
| c.3952C>G | L1318V 2D  AIThe SynGAP1 missense variant L1318V is catalogued in gnomAD (ID 6‑33451826‑C‑G) but has no ClinVar entry. Across a broad panel of in silico predictors, every tool reports a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool in the set predicts pathogenicity, so the pathogenic group is empty. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized returns a benign prediction, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, and this is consistent with the absence of a ClinVar pathogenic classification. Thus, the variant is most likely benign, and this assessment does not contradict the ClinVar record, which contains no pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.887230 | Disordered | 0.968271 | Binding | 0.399 | 0.865 | 0.750 | 6-33451826-C-G | 1 | 6.31e-7 | -3.938 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.038 | Likely Benign | -0.58 | Neutral | 0.113 | Benign | 0.028 | Benign | 4.08 | Benign | 0.75 | Tolerated | 3.77 | 5 | 0.1861 | 0.3027 | 1 | 2 | 0.4 | -14.03 | ||||||||||||||||||||||||||||||||||
| c.707C>G | A236G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A236G is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are REVEL, premPS, and PROVEAN. Four tools (FoldX, Rosetta, Foldetta, and ESM1b) return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—also favors benign, while Foldetta remains uncertain. Overall, the majority of available predictions support a benign impact, and there is no ClinVar annotation to contradict this assessment. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.185198 | Structured | 0.329926 | Uncertain | 0.775 | 0.330 | 0.000 | -7.412 | In-Between | 0.162 | Likely Benign | Likely Benign | 0.92 | Ambiguous | 0.1 | 1.15 | Ambiguous | 1.04 | Ambiguous | 1.09 | Destabilizing | 0.615 | Likely Pathogenic | -3.35 | Deleterious | 0.067 | Benign | 0.028 | Benign | 5.74 | Benign | 0.07 | Tolerated | 0.2200 | 0.3899 | 1 | 0 | -2.2 | -14.03 | ||||||||||||||||||||||||||||||
| c.191T>A | I64K 2D  AIThe SynGAP1 missense variant I64K is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus remains benign; Foldetta results are unavailable. Overall, the majority of predictions lean toward a benign impact, and this conclusion does not contradict the ClinVar status, which contains no report for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.422041 | Structured | 0.475481 | Uncertain | 0.478 | 0.747 | 0.125 | -3.206 | Likely Benign | 0.964 | Likely Pathogenic | Likely Pathogenic | 0.159 | Likely Benign | -0.47 | Neutral | 0.334 | Benign | 0.029 | Benign | 4.07 | Benign | 0.00 | Affected | 0.0878 | 0.0740 | -2 | -3 | -8.4 | 15.01 | |||||||||||||||||||||||||||||||||||||||
| c.19T>C | S7P 2D  AIThe SynGAP1 missense variant S7P is reported in gnomAD (ID 6‑33420283‑T‑C) but has no ClinVar entry. In silico prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign status. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority‑vote) is benign; Foldetta results are unavailable. Overall, the consensus of the majority of tools, including the high‑accuracy methods, points to a benign effect. This prediction is consistent with the absence of a ClinVar pathogenic classification, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.590140 | Disordered | 0.548467 | Binding | 0.386 | 0.922 | 0.750 | 6-33420283-T-C | -3.798 | Likely Benign | 0.111 | Likely Benign | Likely Benign | 0.210 | Likely Benign | -0.23 | Neutral | 0.267 | Benign | 0.029 | Benign | 4.07 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2474 | 0.4925 | -1 | 1 | -0.8 | 10.04 | ||||||||||||||||||||||||||||||||||||
| c.2299A>G | I767V 2D  AIThe SynGAP1 missense variant I767V is listed in ClinVar (ID 1402700.0) with an “Uncertain” clinical significance and is not reported in gnomAD. All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a “Likely Benign” outcome. No tool predicts pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, while Foldetta (combining FoldX‑MD and Rosetta stability predictions) has no available result for this variant. Overall, the computational evidence strongly supports a benign effect, and this conclusion does not contradict the current ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.321458 | Structured | 0.927771 | Binding | 0.369 | 0.872 | 0.125 | Uncertain | 1 | -2.791 | Likely Benign | 0.064 | Likely Benign | Likely Benign | 0.096 | Likely Benign | 0.10 | Neutral | 0.072 | Benign | 0.029 | Benign | 4.21 | Benign | 1.00 | Tolerated | 3.64 | 6 | 0.1418 | 0.4227 | 4 | 3 | -0.3 | -14.03 | |||||||||||||||||||||||||||||||||||
| c.230G>T | S77I 2D  AIThe SynGAP1 missense variant S77I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. There is no ClinVar entry to contradict this assessment. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.446124 | Uncertain | 0.310 | 0.855 | 0.375 | -3.918 | Likely Benign | 0.140 | Likely Benign | Likely Benign | 0.038 | Likely Benign | -1.41 | Neutral | 0.604 | Possibly Damaging | 0.029 | Benign | 4.07 | Benign | 0.00 | Affected | 0.0758 | 0.5165 | -1 | -2 | 5.3 | 26.08 | |||||||||||||||||||||||||||||||||||||||
| c.2852A>G | H951R 2D  AIThe SynGAP1 missense variant H951R is listed in ClinVar as Pathogenic (ClinVar ID 1003739.0) and is not reported in gnomAD. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign. The Foldetta protein‑folding stability analysis is unavailable for this variant. Based on the collective predictions, the variant is most likely benign, which contradicts its ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988291 | Disordered | 0.901477 | Binding | 0.415 | 0.925 | 0.750 | Likely Pathogenic | 1 | -4.964 | Likely Benign | 0.125 | Likely Benign | Likely Benign | 0.185 | Likely Benign | -1.08 | Neutral | 0.048 | Benign | 0.029 | Benign | 5.46 | Benign | 0.24 | Tolerated | 3.77 | 5 | 0.2808 | 0.3220 | 2 | 0 | -1.3 | 19.05 | |||||||||||||||||||||||||||||||||||
| c.287G>T | G96V 2D  AIThe SynGAP1 missense variant G96V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.657645 | Disordered | 0.599491 | Binding | 0.335 | 0.871 | 0.625 | -4.266 | Likely Benign | 0.078 | Likely Benign | Likely Benign | 0.109 | Likely Benign | -1.43 | Neutral | 0.334 | Benign | 0.029 | Benign | 4.18 | Benign | 0.00 | Affected | 0.1575 | 0.3833 | -1 | -3 | 4.6 | 42.08 | |||||||||||||||||||||||||||||||||||||||
| c.3332A>T | K1111M 2D  AIThe SynGAP1 missense variant K1111M is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable. Taken together, the majority of evidence points to a benign impact for K1111M. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.910643 | Disordered | 0.921455 | Binding | 0.300 | 0.902 | 0.875 | -5.579 | Likely Benign | 0.759 | Likely Pathogenic | Likely Benign | 0.071 | Likely Benign | -1.75 | Neutral | 0.072 | Benign | 0.029 | Benign | 2.59 | Benign | 0.12 | Tolerated | 0.1388 | 0.4701 | 0 | -1 | 5.8 | 3.02 | |||||||||||||||||||||||||||||||||||||||
| c.3854C>T | P1285L 2D  AIThe SynGAP1 missense variant P1285L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the collective evidence strongly supports a benign classification for P1285L, and this conclusion is consistent with the lack of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.834292 | Disordered | 0.821643 | Binding | 0.557 | 0.759 | 0.750 | -3.663 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.072 | Likely Benign | 0.06 | Neutral | 0.072 | Benign | 0.029 | Benign | 4.31 | Benign | 1.00 | Tolerated | 0.2159 | 0.4539 | -3 | -3 | 5.4 | 16.04 | |||||||||||||||||||||||||||||||||||||||
| c.3974C>G | P1325R 2D  AIThe SynGAP1 missense variant P1325R is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and there is no conflict with ClinVar status, which contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.876521 | Disordered | 0.893621 | Binding | 0.439 | 0.791 | 0.875 | -5.427 | Likely Benign | 0.173 | Likely Benign | Likely Benign | 0.051 | Likely Benign | -0.33 | Neutral | 0.182 | Benign | 0.029 | Benign | 4.06 | Benign | 0.00 | Affected | 0.1668 | 0.3081 | 0 | -2 | -2.9 | 59.07 | |||||||||||||||||||||||||||||||||||||||
| c.1042G>C | V348L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V348L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign; and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. No other high‑confidence tools provide conflicting evidence. Based on the preponderance of predictions, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.170161 | Structured | 0.346556 | Uncertain | 0.951 | 0.414 | 0.000 | -4.842 | Likely Benign | 0.270 | Likely Benign | Likely Benign | -0.58 | Ambiguous | 0.0 | 0.04 | Likely Benign | -0.27 | Likely Benign | 0.24 | Likely Benign | 0.072 | Likely Benign | -0.75 | Neutral | 0.264 | Benign | 0.030 | Benign | 1.89 | Pathogenic | 0.62 | Tolerated | 0.1088 | 0.4953 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||
| c.1042G>T | V348L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V348L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign; and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. No other high‑confidence tools provide conflicting evidence. Based on the preponderance of predictions, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.170161 | Structured | 0.346556 | Uncertain | 0.951 | 0.414 | 0.000 | -4.842 | Likely Benign | 0.270 | Likely Benign | Likely Benign | -0.58 | Ambiguous | 0.0 | 0.04 | Likely Benign | -0.27 | Likely Benign | 0.24 | Likely Benign | 0.072 | Likely Benign | -0.75 | Neutral | 0.264 | Benign | 0.030 | Benign | 1.89 | Pathogenic | 0.62 | Tolerated | 0.1088 | 0.4953 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||
| c.1981C>A | Q661K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant Q661K is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Only ESM1b predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign; the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign majority (2 benign vs. 1 pathogenic, with one uncertain), and Foldetta also predicts benign stability. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.048328 | Structured | 0.117089 | Uncertain | 0.924 | 0.309 | 0.000 | -10.581 | Likely Pathogenic | 0.400 | Ambiguous | Likely Benign | -0.01 | Likely Benign | 0.0 | -0.18 | Likely Benign | -0.10 | Likely Benign | 0.04 | Likely Benign | 0.108 | Likely Benign | -1.89 | Neutral | 0.098 | Benign | 0.030 | Benign | 3.59 | Benign | 0.42 | Tolerated | 0.1756 | 0.3592 | 1 | 1 | -0.4 | 0.04 | ||||||||||||||||||||||||||||||
| c.1984C>A | Q662K 2D  AIThe SynGAP1 missense variant Q662K is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign, while only ESM1b predicts pathogenicity. When high‑accuracy methods are considered separately, AlphaMissense‑Optimized remains benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a Likely Benign verdict, and Foldetta (combining FoldX‑MD and Rosetta outputs) also reports a benign effect. No conflicting evidence is present. Therefore, the variant is most likely benign, and this assessment does not contradict any ClinVar annotation (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.046336 | Structured | 0.103446 | Uncertain | 0.932 | 0.323 | 0.000 | -8.892 | Likely Pathogenic | 0.309 | Likely Benign | Likely Benign | -0.02 | Likely Benign | 0.2 | 0.03 | Likely Benign | 0.01 | Likely Benign | -0.04 | Likely Benign | 0.108 | Likely Benign | -0.80 | Neutral | 0.321 | Benign | 0.030 | Benign | 3.49 | Benign | 0.37 | Tolerated | 0.2391 | 0.3198 | 1 | 1 | -0.4 | 0.04 | |||||||||||||||||||||||||||||
| c.2168C>T | T723I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T723I is listed in ClinVar as Benign (ClinVar ID 436924.0) and is observed in gnomAD (variant ID 6‑33441633‑C‑T). Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only SIFT classifies the change as pathogenic. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports “Likely Benign.” High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, the SGM Consensus is likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also indicates a benign impact. No prediction or stability result is missing or inconclusive. Based on the collective evidence, the variant is most likely benign, and this conclusion is consistent with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.476583 | Structured | 0.458243 | Uncertain | 0.945 | 0.447 | 0.375 | Likely Benign | 1 | 6-33441633-C-T | 2 | 1.24e-6 | -2.591 | Likely Benign | 0.120 | Likely Benign | Likely Benign | -0.39 | Likely Benign | 0.0 | -0.20 | Likely Benign | -0.30 | Likely Benign | 0.26 | Likely Benign | 0.045 | Likely Benign | -2.09 | Neutral | 0.088 | Benign | 0.030 | Benign | 3.39 | Benign | 0.03 | Affected | 3.50 | 8 | 0.0708 | 0.5803 | 0 | -1 | 5.2 | 12.05 | 252.3 | -31.6 | 0.0 | 0.0 | -0.2 | 0.2 | X | Uncertain | The hydroxyl group of Thr723, located on the outer surface of an α-helix (res. Leu714-Arg726), continuously forms hydrogen bonds with the backbone carbonyl of Asn719 in the WT simulations, potentially lowering the stability of the α-helix. In the variant simulations, the sec-butyl side chain of Ile723 cannot form any hydrogen bonds, which, in theory, could increase the helix stability. However, because the model ends abruptly at the C-terminus, no definite conclusions can be drawn based on the simulations. | |||||||||||||
| c.2309A>T | Q770L 2D  AIThe SynGAP1 missense variant Q770L is not reported in ClinVar and is absent from gnomAD, indicating no known population frequency data. Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign. No Foldetta stability prediction is available, so it does not influence the assessment. Overall, the majority of evidence points to a benign effect for Q770L, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.436924 | Structured | 0.923732 | Binding | 0.328 | 0.887 | 0.250 | -5.524 | Likely Benign | 0.521 | Ambiguous | Likely Benign | 0.197 | Likely Benign | -2.17 | Neutral | 0.095 | Benign | 0.030 | Benign | 4.14 | Benign | 0.01 | Affected | 0.0776 | 0.6230 | -2 | -2 | 7.3 | -14.97 | |||||||||||||||||||||||||||||||||||||||
| c.3119G>C | G1040A 2D  AIThe SynGAP1 missense variant G1040A is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are REVEL and FATHMM, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote among AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign prediction (2 benign vs. 1 pathogenic). Foldetta, a protein‑folding stability method, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation, as the variant is not yet classified in that database. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.964893 | Disordered | 0.973805 | Binding | 0.332 | 0.816 | 0.625 | -2.625 | Likely Benign | 0.409 | Ambiguous | Likely Benign | 0.606 | Likely Pathogenic | -1.65 | Neutral | 0.114 | Benign | 0.030 | Benign | -0.74 | Pathogenic | 0.30 | Tolerated | 0.3246 | 0.5331 | 1 | 0 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.3143G>A | G1048E 2D  AIThe SynGAP1 missense variant G1048E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only REVEL predicts a pathogenic outcome, while ESM1b is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar claim exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.977651 | Disordered | 0.923876 | Binding | 0.346 | 0.916 | 0.750 | -7.028 | In-Between | 0.331 | Likely Benign | Likely Benign | 0.529 | Likely Pathogenic | -0.62 | Neutral | 0.018 | Benign | 0.030 | Benign | 2.54 | Benign | 0.10 | Tolerated | 0.1444 | 0.4063 | 0 | -2 | -3.1 | 72.06 | |||||||||||||||||||||||||||||||||||||||
| c.3299G>A | G1100D 2D  AIThe SynGAP1 missense variant G1100D is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized classifying the variant as benign, while the SGM Consensus (a majority vote among AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.882776 | Disordered | 0.972009 | Binding | 0.360 | 0.865 | 0.875 | -5.235 | Likely Benign | 0.577 | Likely Pathogenic | Likely Benign | 0.141 | Likely Benign | -1.67 | Neutral | 0.049 | Benign | 0.030 | Benign | 1.93 | Pathogenic | 0.01 | Affected | 0.1924 | 0.3043 | 1 | -1 | -3.1 | 58.04 | ||||||||||||||||||||||||||||||||||||||||
| c.3398T>C | I1133T 2D  AIThe SynGAP1 missense variant I1133T has no ClinVar entry and is present in gnomAD (ID 6‑33443950‑T‑C). Consensus among most tools is benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized all predict a benign effect, while AlphaMissense‑Default remains uncertain and no tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized reports benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates “Likely Benign,” and Foldetta’s protein‑folding stability analysis is unavailable. Overall, the evidence strongly supports a benign classification, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.832785 | Binding | 0.316 | 0.892 | 0.750 | 6-33443950-T-C | -4.522 | Likely Benign | 0.563 | Ambiguous | Likely Benign | 0.275 | Likely Benign | -0.30 | Neutral | 0.026 | Benign | 0.030 | Benign | 5.50 | Benign | 0.18 | Tolerated | 4.32 | 3 | 0.1007 | 0.1975 | -1 | 0 | -5.2 | -12.05 | ||||||||||||||||||||||||||||||||||||
| c.826C>G | P276A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant P276A is reported in gnomAD (variant ID 6‑33437731‑C‑G) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The high‑accuracy consensus methods give a benign signal: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign,” while Foldetta (combining FoldX‑MD and Rosetta outputs) is inconclusive. Overall, the majority of evidence points to a benign effect, and this assessment does not contradict any ClinVar status (none is available). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.037156 | Structured | 0.338937 | Uncertain | 0.724 | 0.230 | 0.250 | 6-33437731-C-G | 5 | 3.10e-6 | -3.414 | Likely Benign | 0.058 | Likely Benign | Likely Benign | 1.42 | Ambiguous | 0.1 | 1.01 | Ambiguous | 1.22 | Ambiguous | 0.50 | Likely Benign | 0.187 | Likely Benign | -2.31 | Neutral | 0.044 | Benign | 0.030 | Benign | 1.98 | Pathogenic | 0.57 | Tolerated | 3.38 | 19 | 0.3149 | 0.3669 | -1 | 1 | 3.4 | -26.04 | ||||||||||||||||||||||||
| c.990C>A | D330E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 D330E missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are Rosetta and FATHMM. The remaining tools—FoldX, AlphaMissense‑Default, and Foldetta—return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact. This conclusion is not contradicted by any ClinVar annotation, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.380708 | Structured | 0.360008 | Uncertain | 0.805 | 0.488 | 0.250 | -3.427 | Likely Benign | 0.407 | Ambiguous | Likely Benign | 0.98 | Ambiguous | 0.2 | 2.31 | Destabilizing | 1.65 | Ambiguous | 0.39 | Likely Benign | 0.073 | Likely Benign | -1.38 | Neutral | 0.122 | Benign | 0.030 | Benign | 0.97 | Pathogenic | 0.67 | Tolerated | 0.1464 | 0.4416 | 3 | 2 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||
| c.990C>G | D330E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 D330E missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are Rosetta and FATHMM. The remaining tools—FoldX, AlphaMissense‑Default, and Foldetta—return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact. This conclusion is not contradicted by any ClinVar annotation, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.380708 | Structured | 0.360008 | Uncertain | 0.805 | 0.488 | 0.250 | -3.427 | Likely Benign | 0.407 | Ambiguous | Likely Benign | 0.98 | Ambiguous | 0.2 | 2.31 | Destabilizing | 1.65 | Ambiguous | 0.39 | Likely Benign | 0.075 | Likely Benign | -1.38 | Neutral | 0.122 | Benign | 0.030 | Benign | 0.97 | Pathogenic | 0.67 | Tolerated | 0.1464 | 0.4416 | 3 | 2 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||
| c.2272T>C | Y758H 2D  AIThe SynGAP1 missense variant Y758H is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this assessment does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.517562 | Disordered | 0.856063 | Binding | 0.289 | 0.871 | 0.375 | -3.194 | Likely Benign | 0.299 | Likely Benign | Likely Benign | 0.097 | Likely Benign | -0.92 | Neutral | 0.064 | Benign | 0.031 | Benign | 2.71 | Benign | 0.02 | Affected | 0.2519 | 0.0331 | 0 | 2 | -1.9 | -26.03 | |||||||||||||||||||||||||||||||||||||||
| c.3241G>T | A1081S 2D  AIThe SynGAP1 missense variant A1081S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta results are unavailable. Consequently, the variant is most likely benign, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.874069 | Disordered | 0.979759 | Binding | 0.288 | 0.895 | 0.750 | -3.536 | Likely Benign | 0.109 | Likely Benign | Likely Benign | 0.078 | Likely Benign | -0.29 | Neutral | 0.021 | Benign | 0.031 | Benign | 4.02 | Benign | 0.23 | Tolerated | 0.2268 | 0.4915 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.3301C>A | P1101T 2D  AIThe SynGAP1 missense variant P1101T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.874069 | Disordered | 0.968967 | Binding | 0.457 | 0.861 | 0.875 | -4.161 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.126 | Likely Benign | -1.60 | Neutral | 0.115 | Benign | 0.031 | Benign | 4.22 | Benign | 0.04 | Affected | 0.1729 | 0.5860 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.401G>T | S134I 2D  AIThe SynGAP1 missense variant S134I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. The SGM‑Consensus, which is a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Pathogenic. High‑accuracy assessments show AlphaMissense‑Optimized as Pathogenic and the SGM‑Consensus as Likely Pathogenic; Foldetta results are unavailable. Based on the collective predictions, the variant is most likely pathogenic, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.447574 | Structured | 0.695837 | Binding | 0.333 | 0.898 | 0.250 | -8.860 | Likely Pathogenic | 0.983 | Likely Pathogenic | Likely Pathogenic | 0.230 | Likely Benign | -3.27 | Deleterious | 0.041 | Benign | 0.031 | Benign | 3.81 | Benign | 0.00 | Affected | 0.0683 | 0.5191 | -1 | -2 | 5.3 | 26.08 | |||||||||||||||||||||||||||||||||||||||
| c.494G>T | S165I 2D  AISynGAP1 missense variant S165I has no ClinVar record and is absent from gnomAD. Individual prediction tools cluster into two groups: benign predictions come from REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized; pathogenic predictions come from PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Pathogenic. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM‑Consensus remains Likely Pathogenic; Foldetta stability analysis is unavailable. Overall, the majority of single‑tool predictions favor a benign effect, whereas the consensus score suggests pathogenicity. Consequently, the variant is most likely benign according to the bulk of evidence, and this assessment does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.429200 | Structured | 0.509123 | Binding | 0.324 | 0.644 | 0.250 | -11.304 | Likely Pathogenic | 0.774 | Likely Pathogenic | Likely Benign | 0.259 | Likely Benign | -2.53 | Deleterious | 0.084 | Benign | 0.031 | Benign | 3.96 | Benign | 0.00 | Affected | 0.0943 | 0.5027 | -1 | -2 | 5.3 | 26.08 | |||||||||||||||||||||||||||||||||||||||
| c.70G>A | V24I 2D  AIThe SynGAP1 missense variant V24I is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6-33423479-G-A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Taken together, the overwhelming majority of computational evidence supports a benign impact for V24I, and this conclusion does not contradict the ClinVar designation, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.438970 | Uncertain | 0.382 | 0.890 | 0.500 | Uncertain | 1 | 6-33423479-G-A | 9 | 5.58e-6 | -3.701 | Likely Benign | 0.137 | Likely Benign | Likely Benign | 0.069 | Likely Benign | -0.25 | Neutral | 0.043 | Benign | 0.031 | Benign | 3.96 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0844 | 0.4564 | 3 | 4 | 0.3 | 14.03 | ||||||||||||||||||||||||||||||||
| c.118G>A | D40N 2D  AIThe SynGAP1 missense variant D40N is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus, SGM‑Consensus, classifies the variant as Likely Benign, and AlphaMissense‑Optimized also reports a benign prediction. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The predictions do not contradict ClinVar status, as ClinVar contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.384043 | Structured | 0.432002 | Uncertain | 0.319 | 0.769 | 0.375 | -3.841 | Likely Benign | 0.210 | Likely Benign | Likely Benign | 0.103 | Likely Benign | -0.81 | Neutral | 0.028 | Benign | 0.032 | Benign | 4.05 | Benign | 0.00 | Affected | 0.2676 | 0.8761 | 2 | 1 | 0.0 | -0.98 | |||||||||||||||||||||||||||||||||||||||
| c.119A>G | D40G 2D  AIThe SynGAP1 D40G missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.384043 | Structured | 0.432002 | Uncertain | 0.319 | 0.769 | 0.375 | -2.777 | Likely Benign | 0.287 | Likely Benign | Likely Benign | 0.113 | Likely Benign | -1.43 | Neutral | 0.012 | Benign | 0.032 | Benign | 4.01 | Benign | 0.00 | Affected | 0.4110 | 0.7949 | 1 | -1 | 3.1 | -58.04 | |||||||||||||||||||||||||||||||||||||||
| c.1604G>A | S535N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S535N is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Across the broad panel of in‑silico predictors, 13 tools (REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus “Likely Benign”) uniformly predict a benign effect, whereas only FATHMM assigns a pathogenic label. High‑accuracy assessments corroborate the benign consensus: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign stability. Thus, the variant is most likely benign based on the available predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.236433 | Structured | 0.041365 | Uncertain | 0.918 | 0.343 | 0.000 | -4.957 | Likely Benign | 0.196 | Likely Benign | Likely Benign | -0.46 | Likely Benign | 0.2 | -0.26 | Likely Benign | -0.36 | Likely Benign | 0.18 | Likely Benign | 0.176 | Likely Benign | -0.05 | Neutral | 0.070 | Benign | 0.032 | Benign | -1.25 | Pathogenic | 0.29 | Tolerated | 0.1390 | 0.4784 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||||
| c.184G>A | D62N 2D  AIThe SynGAP1 missense variant D62N is reported in gnomAD (variant ID 6‑33423593‑G‑A) but has no ClinVar entry. In silico prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign status. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of tools indicates that D62N is most likely benign, and this assessment does not contradict any ClinVar classification because none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.425610 | Structured | 0.476010 | Uncertain | 0.575 | 0.720 | 0.125 | 6-33423593-G-A | 1 | 6.20e-7 | -4.607 | Likely Benign | 0.207 | Likely Benign | Likely Benign | 0.075 | Likely Benign | -1.08 | Neutral | 0.028 | Benign | 0.032 | Benign | 4.11 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1670 | 0.6154 | 1 | 2 | 0.0 | -0.98 | ||||||||||||||||||||||||||||||||||
| c.185A>G | D62G 2D  AIThe SynGAP1 D62G missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.425610 | Structured | 0.476010 | Uncertain | 0.575 | 0.720 | 0.125 | -4.047 | Likely Benign | 0.316 | Likely Benign | Likely Benign | 0.097 | Likely Benign | -1.76 | Neutral | 0.012 | Benign | 0.032 | Benign | 4.07 | Benign | 0.00 | Affected | 0.4016 | 0.6081 | 1 | -1 | 3.1 | -58.04 | |||||||||||||||||||||||||||||||||||||||
| c.1931A>C | D644A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 D644A missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that classify the variant as benign include REVEL, FoldX, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, Foldetta, and premPS. Only PROVEAN predicts it as pathogenic, while Rosetta and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) resolves to benign; and Foldetta also predicts benign stability. Based on the aggregate evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.066181 | Structured | 0.248888 | Uncertain | 0.883 | 0.320 | 0.000 | -3.358 | Likely Benign | 0.502 | Ambiguous | Likely Benign | -0.14 | Likely Benign | 0.1 | -0.83 | Ambiguous | -0.49 | Likely Benign | -0.18 | Likely Benign | 0.274 | Likely Benign | -3.90 | Deleterious | 0.311 | Benign | 0.032 | Benign | 3.51 | Benign | 0.39 | Tolerated | 0.3883 | 0.5481 | 0 | -2 | 5.3 | -44.01 | ||||||||||||||||||||||||||||||
| c.20C>T | S7F 2D  AIThe SynGAP1 missense variant S7F is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus, SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. AlphaMissense‑Optimized independently predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, was not available for this variant. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.590140 | Disordered | 0.548467 | Binding | 0.386 | 0.922 | 0.750 | -4.346 | Likely Benign | 0.250 | Likely Benign | Likely Benign | 0.171 | Likely Benign | -0.24 | Neutral | 0.296 | Benign | 0.032 | Benign | 4.06 | Benign | 0.00 | Affected | 0.0622 | 0.5177 | -3 | -2 | 3.6 | 60.10 | |||||||||||||||||||||||||||||||||||||||
| c.23T>A | I8N 2D  AIThe SynGAP1 missense variant I8N is not reported in ClinVar and has no entry in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only two tools (polyPhen‑2 HumDiv and SIFT) predict pathogenicity, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the consensus of the majority of predictors, including the high‑accuracy methods, points to a benign impact. This conclusion is consistent with the absence of any ClinVar classification, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.543080 | Binding | 0.341 | 0.916 | 0.625 | -3.979 | Likely Benign | 0.163 | Likely Benign | Likely Benign | 0.120 | Likely Benign | 0.05 | Neutral | 0.561 | Possibly Damaging | 0.032 | Benign | 3.99 | Benign | 0.00 | Affected | 0.0803 | 0.0540 | -2 | -3 | -8.0 | 0.94 | |||||||||||||||||||||||||||||||||||||||
| c.2671C>G | L891V 2D  AIThe SynGAP1 missense variant L891V is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta results are unavailable, so they do not influence the assessment. Overall, the variant is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.712013 | Disordered | 0.505861 | Binding | 0.305 | 0.923 | 0.750 | -4.992 | Likely Benign | 0.094 | Likely Benign | Likely Benign | 0.044 | Likely Benign | -0.65 | Neutral | 0.057 | Benign | 0.032 | Benign | 2.72 | Benign | 0.70 | Tolerated | 0.1614 | 0.2945 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3304G>T | A1102S 2D  AIThe SynGAP1 missense variant A1102S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the collective evidence strongly supports a benign classification, and this conclusion does not contradict the ClinVar status, which currently contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.915074 | Disordered | 0.962659 | Binding | 0.388 | 0.859 | 0.875 | -2.900 | Likely Benign | 0.067 | Likely Benign | Likely Benign | 0.041 | Likely Benign | 0.05 | Neutral | 0.019 | Benign | 0.032 | Benign | 2.57 | Benign | 0.59 | Tolerated | 0.2666 | 0.6501 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.41C>G | P14R 2D  AIThe SynGAP1 missense variant P14R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. The high‑accuracy consensus methods give a benign verdict: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” No result is available from Foldetta, so its folding‑stability assessment is not considered. Overall, the majority of evidence points to a benign impact, and this is consistent with the lack of any ClinVar classification. Thus, the variant is most likely benign and does not contradict ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.471596 | Uncertain | 0.399 | 0.909 | 0.375 | -2.794 | Likely Benign | 0.253 | Likely Benign | Likely Benign | 0.183 | Likely Benign | 0.13 | Neutral | 0.486 | Possibly Damaging | 0.032 | Benign | 4.20 | Benign | 0.00 | Affected | 0.1597 | 0.3536 | 0 | -2 | -2.9 | 59.07 | |||||||||||||||||||||||||||||||||||||||
| c.50C>T | S17F 2D  AIThe SynGAP1 missense variant S17F is listed in ClinVar (ID 3451958) with an “Uncertain” status and is present in gnomAD (variant ID 6‑33420314‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, SIFT, and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.483068 | Structured | 0.452228 | Uncertain | 0.341 | 0.910 | 0.375 | Conflicting | 2 | 6-33420314-C-T | 10 | 6.49e-6 | -3.888 | Likely Benign | 0.637 | Likely Pathogenic | Likely Benign | 0.048 | Likely Benign | -0.99 | Neutral | 0.486 | Possibly Damaging | 0.032 | Benign | 3.99 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0729 | 0.6468 | -2 | -3 | 3.6 | 60.10 | ||||||||||||||||||||||||||||||||
| c.106C>G | H36D 2D  AIThe SynGAP1 missense variant H36D is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta results are not available for this variant. Overall, the majority of computational evidence indicates that H36D is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -2.859 | Likely Benign | 0.225 | Likely Benign | Likely Benign | 0.117 | Likely Benign | -1.06 | Neutral | 0.043 | Benign | 0.033 | Benign | 4.21 | Benign | 0.00 | Affected | 0.2805 | 0.3496 | 1 | -1 | -0.3 | -22.05 | |||||||||||||||||||||||||||||||||||||||
| c.107A>G | H36R 2D  AIThe SynGAP1 missense variant H36R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -2.513 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.034 | Likely Benign | -0.70 | Neutral | 0.084 | Benign | 0.033 | Benign | 4.25 | Benign | 0.00 | Affected | 0.2417 | 0.3590 | 2 | 0 | -1.3 | 19.05 | |||||||||||||||||||||||||||||||||||||||
| c.1211C>A | A404E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A404E is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. In contrast, the majority of tools predict a pathogenic impact: FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate pathogenicity, and the SGM‑Consensus score is “Likely Pathogenic.” High‑accuracy assessments further support a deleterious effect: AlphaMissense‑Optimized predicts pathogenic; the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Pathogenic”; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, also predicts pathogenicity. Based on the preponderance of pathogenic predictions, the variant is most likely pathogenic, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | C2 | 0.232838 | Structured | 0.415505 | Uncertain | 0.965 | 0.355 | 0.000 | -13.639 | Likely Pathogenic | 0.992 | Likely Pathogenic | Likely Pathogenic | 2.16 | Destabilizing | 0.1 | 3.04 | Destabilizing | 2.60 | Destabilizing | 1.51 | Destabilizing | 0.163 | Likely Benign | -2.77 | Deleterious | 0.179 | Benign | 0.033 | Benign | 4.02 | Benign | 0.02 | Affected | 0.1423 | 0.2383 | 0 | -1 | -5.3 | 58.04 | |||||||||||||||||||||||||||||
| c.1280A>T | H427L 2D  3DClick to see structure in 3D Viewer AISynGAP1 H427L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools cluster into two groups: nine tools (REVEL, FoldX, Rosetta, Foldetta, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Optimized) predict a benign effect, while five tools (SGM‑Consensus, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default) predict pathogenicity. High‑accuracy methods provide a more focused view: AlphaMissense‑Optimized indicates a benign outcome; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, remains pathogenic; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also predicts benign. Taken together, the majority of evidence supports a benign impact for H427L, and this assessment does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.081712 | Structured | 0.394261 | Uncertain | 0.962 | 0.287 | 0.000 | -9.691 | Likely Pathogenic | 0.755 | Likely Pathogenic | Likely Benign | -0.17 | Likely Benign | 0.0 | 0.05 | Likely Benign | -0.06 | Likely Benign | 0.31 | Likely Benign | 0.272 | Likely Benign | -5.38 | Deleterious | 0.299 | Benign | 0.033 | Benign | 3.42 | Benign | 0.01 | Affected | 0.0942 | 0.4953 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||
| c.1289T>A | M430K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M430K is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are PROVEAN and ESM1b. The remaining tools—FoldX, Foldetta, premPS, and AlphaMissense‑Default—return uncertain or inconclusive results. High‑accuracy methods give a mixed picture: AlphaMissense‑Optimized classifies the variant as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) favors a pathogenic outcome; Foldetta remains uncertain. Overall, the majority of individual predictors lean toward a benign interpretation, whereas the SGM Consensus suggests pathogenicity. Given the lack of ClinVar evidence, there is no contradiction with existing clinical annotations. Based on the aggregate predictions, the variant is most likely benign, and this assessment does not conflict with the current ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.071867 | Structured | 0.385298 | Uncertain | 0.952 | 0.306 | 0.000 | -10.816 | Likely Pathogenic | 0.494 | Ambiguous | Likely Benign | 0.78 | Ambiguous | 0.1 | 0.44 | Likely Benign | 0.61 | Ambiguous | 0.86 | Ambiguous | 0.149 | Likely Benign | -2.66 | Deleterious | 0.134 | Benign | 0.033 | Benign | 3.47 | Benign | 0.32 | Tolerated | 0.1660 | 0.1271 | 0 | -1 | -5.8 | -3.02 | ||||||||||||||||||||||||||||||
| c.1595C>A | T532K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 T532K missense variant is not reported in ClinVar and has no entries in gnomAD. Prediction tools that classify the change as benign include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and AlphaMissense‑Optimized. Those that predict pathogenicity are ESM1b, FATHMM, AlphaMissense‑Default, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM Consensus as likely pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) as benign. With ten benign versus four pathogenic predictions and two of the three high‑accuracy tools supporting a benign outcome, the variant is most likely benign. This assessment does not contradict the ClinVar status, which currently has no classification for T532K. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.275179 | Structured | 0.021478 | Uncertain | 0.889 | 0.385 | 0.000 | -8.460 | Likely Pathogenic | 0.644 | Likely Pathogenic | Likely Benign | -0.44 | Likely Benign | 0.2 | 0.24 | Likely Benign | -0.10 | Likely Benign | 0.34 | Likely Benign | 0.376 | Likely Benign | -1.97 | Neutral | 0.259 | Benign | 0.033 | Benign | -1.21 | Pathogenic | 0.29 | Tolerated | 0.0943 | 0.1992 | 0 | -1 | -3.2 | 27.07 | |||||||||||||||||||||||||||||
| c.2020A>T | T674S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T674S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Functional prediction tools uniformly indicate a benign effect: SIFT, PolyPhen‑2 (HumDiv and HumVar), REVEL, PROVEAN, premPS, FoldX, Rosetta, and AlphaMissense‑Default all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also reports a benign effect. With all available evidence pointing to a benign outcome and no conflicting ClinVar annotation, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.129801 | Structured | 0.109297 | Uncertain | 0.521 | 0.349 | 0.000 | -5.320 | Likely Benign | 0.082 | Likely Benign | Likely Benign | -0.23 | Likely Benign | 0.0 | 0.44 | Likely Benign | 0.11 | Likely Benign | -0.14 | Likely Benign | 0.084 | Likely Benign | 0.47 | Neutral | 0.107 | Benign | 0.033 | Benign | 3.47 | Benign | 1.00 | Tolerated | 0.3329 | 0.4938 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||
| c.2021C>G | T674S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T674S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Functional prediction tools uniformly indicate a benign effect: SIFT, PolyPhen‑2 (HumDiv and HumVar), REVEL, PROVEAN, premPS, FoldX, Rosetta, and AlphaMissense‑Default all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also reports a benign effect. With all available evidence pointing to a benign outcome and no conflicting ClinVar annotation, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.129801 | Structured | 0.109297 | Uncertain | 0.521 | 0.349 | 0.000 | -5.320 | Likely Benign | 0.082 | Likely Benign | Likely Benign | -0.23 | Likely Benign | 0.0 | 0.44 | Likely Benign | 0.11 | Likely Benign | -0.14 | Likely Benign | 0.088 | Likely Benign | 0.47 | Neutral | 0.107 | Benign | 0.033 | Benign | 3.47 | Benign | 1.00 | Tolerated | 0.3329 | 0.4938 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||
| c.2187C>A | N729K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N729K has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus call (Likely Benign). Only AlphaMissense‑Default predicts a pathogenic outcome. Tools with uncertain or mixed results are Foldetta (protein‑folding stability) and Rosetta. High‑accuracy assessments: AlphaMissense‑Optimized reports a benign effect; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also indicates likely benign; Foldetta’s stability prediction is inconclusive. Overall, the majority of evidence points to a benign impact for the variant, and this conclusion does not contradict the current ClinVar status, which contains no report for this change. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.750527 | Disordered | 0.426547 | Uncertain | 0.651 | 0.583 | 0.625 | -5.101 | Likely Benign | 0.648 | Likely Pathogenic | Likely Benign | -0.03 | Likely Benign | 0.1 | 1.92 | Ambiguous | 0.95 | Ambiguous | 0.12 | Likely Benign | 0.036 | Likely Benign | -1.39 | Neutral | 0.109 | Benign | 0.033 | Benign | 3.51 | Benign | 0.47 | Tolerated | 0.1948 | 0.3612 | 1 | 0 | -0.4 | 14.07 | ||||||||||||||||||||||||||||||
| c.2187C>G | N729K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N729K has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus call (Likely Benign). Only AlphaMissense‑Default predicts a pathogenic outcome. Tools with uncertain or mixed results are Foldetta (protein‑folding stability) and Rosetta. High‑accuracy assessments: AlphaMissense‑Optimized reports a benign effect; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also indicates likely benign; Foldetta’s stability prediction is inconclusive. Overall, the majority of evidence points to a benign impact for the variant, and this conclusion does not contradict the current ClinVar status, which contains no report for this change. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.750527 | Disordered | 0.426547 | Uncertain | 0.651 | 0.583 | 0.625 | -5.101 | Likely Benign | 0.648 | Likely Pathogenic | Likely Benign | -0.03 | Likely Benign | 0.1 | 1.92 | Ambiguous | 0.95 | Ambiguous | 0.12 | Likely Benign | 0.036 | Likely Benign | -1.39 | Neutral | 0.109 | Benign | 0.033 | Benign | 3.51 | Benign | 0.47 | Tolerated | 0.1948 | 0.3612 | 1 | 0 | -0.4 | 14.07 | ||||||||||||||||||||||||||||||
| c.2188A>G | I730V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I730V is reported as “Likely Benign” in ClinVar and is not present in gnomAD. All available in‑silico predictors classify the change as benign: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.733139 | Disordered | 0.420109 | Uncertain | 0.591 | 0.619 | 0.750 | -3.960 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.25 | Likely Benign | 0.1 | 0.04 | Likely Benign | 0.15 | Likely Benign | 0.03 | Likely Benign | 0.028 | Likely Benign | -0.14 | Neutral | 0.112 | Benign | 0.033 | Benign | 3.48 | Benign | 0.39 | Tolerated | 0.1010 | 0.2303 | 4 | 3 | -0.3 | -14.03 | ||||||||||||||||||||||||||||||
| c.2239G>C | V747L 2D  AIThe SynGAP1 missense variant V747L (ClinVar ID 1985039.0) is listed as ClinVar status Uncertain and is present in gnomAD (6‑33441704‑G‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta stability analysis is unavailable. Overall, the majority of computational evidence supports a benign classification, which is consistent with the ClinVar Uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.816150 | Disordered | 0.594069 | Binding | 0.343 | 0.873 | 0.750 | Uncertain | 1 | 6-33441704-G-C | 2 | 1.24e-6 | -2.790 | Likely Benign | 0.096 | Likely Benign | Likely Benign | 0.047 | Likely Benign | -0.52 | Neutral | 0.065 | Benign | 0.033 | Benign | 2.67 | Benign | 0.00 | Affected | 4.32 | 2 | 0.0699 | 0.3674 | 2 | 1 | -0.4 | 14.03 | ||||||||||||||||||||||||||||||||
| c.2239G>T | V747L 2D  AIThe SynGAP1 missense variant V747L is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Only SIFT predicts a pathogenic outcome. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.816150 | Disordered | 0.594069 | Binding | 0.343 | 0.873 | 0.750 | -2.790 | Likely Benign | 0.096 | Likely Benign | Likely Benign | 0.046 | Likely Benign | -0.52 | Neutral | 0.065 | Benign | 0.033 | Benign | 2.67 | Benign | 0.00 | Affected | 4.32 | 2 | 0.0699 | 0.3674 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||
| c.2278A>C | M760L 2D  AIThe SynGAP1 missense variant M760L is not reported in ClinVar and is absent from gnomAD. Prediction tools that assess pathogenicity uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts a pathogenic outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” verdict. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.893402 | Binding | 0.346 | 0.865 | 0.375 | -2.260 | Likely Benign | 0.134 | Likely Benign | Likely Benign | 0.145 | Likely Benign | -0.68 | Neutral | 0.065 | Benign | 0.033 | Benign | 2.69 | Benign | 0.15 | Tolerated | 0.1802 | 0.4805 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2278A>T | M760L 2D  AIThe SynGAP1 missense variant M760L is not reported in ClinVar and is absent from gnomAD. Prediction tools that assess pathogenicity uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts a pathogenic outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” verdict. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.893402 | Binding | 0.346 | 0.865 | 0.375 | -2.260 | Likely Benign | 0.134 | Likely Benign | Likely Benign | 0.145 | Likely Benign | -0.68 | Neutral | 0.065 | Benign | 0.033 | Benign | 2.69 | Benign | 0.15 | Tolerated | 0.1802 | 0.4805 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2280G>A | M760I 2D  AIThe SynGAP1 missense variant M760I is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) also indicates benign. Foldetta results are unavailable, so they do not influence the assessment. Overall, the collective evidence strongly suggests that M760I is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.893402 | Binding | 0.346 | 0.865 | 0.375 | -3.696 | Likely Benign | 0.486 | Ambiguous | Likely Benign | 0.065 | Likely Benign | -1.03 | Neutral | 0.029 | Benign | 0.033 | Benign | 2.67 | Benign | 0.09 | Tolerated | 0.1595 | 0.4054 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2280G>C | M760I 2D  AIThe SynGAP1 missense variant M760I is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) also indicates benign. Foldetta results are unavailable, so they do not influence the assessment. Overall, the collective evidence strongly suggests that M760I is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.893402 | Binding | 0.346 | 0.865 | 0.375 | -3.696 | Likely Benign | 0.486 | Ambiguous | Likely Benign | 0.065 | Likely Benign | -1.03 | Neutral | 0.029 | Benign | 0.033 | Benign | 2.67 | Benign | 0.09 | Tolerated | 0.1595 | 0.4054 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2280G>T | M760I 2D  AIThe SynGAP1 missense variant M760I is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) also indicates benign. Foldetta results are unavailable, so they do not influence the assessment. Overall, the collective evidence strongly suggests that M760I is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.893402 | Binding | 0.346 | 0.865 | 0.375 | -3.696 | Likely Benign | 0.486 | Ambiguous | Likely Benign | 0.065 | Likely Benign | -1.03 | Neutral | 0.029 | Benign | 0.033 | Benign | 2.67 | Benign | 0.09 | Tolerated | 0.1595 | 0.4054 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.2413C>A | L805M 2D  AIThe SynGAP1 missense variant L805M is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, and no Foldetta stability data are available. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical databases. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.775545 | Disordered | 0.827669 | Binding | 0.341 | 0.903 | 0.625 | -4.229 | Likely Benign | 0.133 | Likely Benign | Likely Benign | 0.034 | Likely Benign | -0.67 | Neutral | 0.029 | Benign | 0.033 | Benign | 2.39 | Pathogenic | 0.00 | Affected | 0.0911 | 0.3867 | 4 | 2 | -1.9 | 18.03 | ||||||||||||||||||||||||||||||||||||||
| c.2806G>A | A936T 2D  AIThe SynGAP1 missense variant A936T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443358‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; no tool predicts pathogenicity. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence overwhelmingly indicates that the variant is most likely benign, which does not contradict the ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.812494 | Disordered | 0.973218 | Binding | 0.319 | 0.874 | 0.625 | Uncertain | 1 | 6-33443358-G-A | 1 | 6.20e-7 | -4.540 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.047 | Likely Benign | -0.50 | Neutral | 0.051 | Benign | 0.033 | Benign | 2.67 | Benign | 0.09 | Tolerated | 3.77 | 5 | 0.1693 | 0.7003 | 0 | 1 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||||
| c.3128G>A | R1043K 2D  AIThe SynGAP1 missense variant R1043K is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this is consistent with the lack of ClinVar or gnomAD entries—there is no contradiction with existing database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.978672 | Disordered | 0.954069 | Binding | 0.299 | 0.853 | 0.625 | -4.038 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.376 | Likely Benign | -1.41 | Neutral | 0.069 | Benign | 0.033 | Benign | 5.39 | Benign | 0.00 | Affected | 0.5141 | 0.4070 | Weaken | 3 | 2 | 0.6 | -28.01 | ||||||||||||||||||||||||||||||||||||||
| c.3790G>A | G1264S 2D  AIThe SynGAP1 missense variant G1264S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: benign predictions are made by REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized classifies the variant as benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also reports it as likely benign. Foldetta results are unavailable. Overall, the evidence strongly supports a benign classification, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.429200 | Structured | 0.762385 | Binding | 0.897 | 0.579 | 0.000 | -0.797 | Likely Benign | 0.096 | Likely Benign | Likely Benign | 0.147 | Likely Benign | 0.82 | Neutral | 0.062 | Benign | 0.033 | Benign | 2.94 | Benign | 1.00 | Tolerated | 0.2376 | 0.4256 | 1 | 0 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||||||||
| c.865A>C | M289L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M289L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only FATHMM predicts a pathogenic outcome. Grouping by consensus, the benign‑predicting tools outnumber the single pathogenic prediction. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a “Likely Benign” verdict; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also reports a benign effect. No prediction or stability result is missing or inconclusive. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.127496 | Structured | 0.403499 | Uncertain | 0.886 | 0.276 | 0.000 | -5.778 | Likely Benign | 0.157 | Likely Benign | Likely Benign | 0.13 | Likely Benign | 0.0 | -0.10 | Likely Benign | 0.02 | Likely Benign | 0.39 | Likely Benign | 0.070 | Likely Benign | -0.95 | Neutral | 0.136 | Benign | 0.033 | Benign | 1.79 | Pathogenic | 0.27 | Tolerated | 0.1092 | 0.3634 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||
| c.865A>T | M289L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M289L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is benign. No prediction or folding stability result is missing or inconclusive. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because the variant is not currently listed in ClinVar. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.127496 | Structured | 0.403499 | Uncertain | 0.886 | 0.276 | 0.000 | -5.778 | Likely Benign | 0.157 | Likely Benign | Likely Benign | 0.13 | Likely Benign | 0.0 | -0.10 | Likely Benign | 0.02 | Likely Benign | 0.39 | Likely Benign | 0.070 | Likely Benign | -0.95 | Neutral | 0.136 | Benign | 0.033 | Benign | 1.79 | Pathogenic | 0.27 | Tolerated | 0.1092 | 0.3634 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||
| c.98A>T | Q33L 2D  AIThe SynGAP1 missense variant Q33L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.436712 | Uncertain | 0.342 | 0.860 | 0.375 | 0.253 | Likely Benign | 0.174 | Likely Benign | Likely Benign | 0.075 | Likely Benign | -1.24 | Neutral | 0.084 | Benign | 0.033 | Benign | 4.18 | Benign | 0.00 | Affected | 0.1127 | 0.6214 | -2 | -2 | 7.3 | -14.97 | |||||||||||||||||||||||||||||||||||||||
| c.2402G>C | G801A 2D  AIThe SynGAP1 missense variant G801A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. The high‑accuracy consensus methods likewise support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta results are not available, so they do not influence the assessment. **Overall, the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which currently has no entry for this variant.** Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.874069 | Disordered | 0.636323 | Binding | 0.320 | 0.892 | 0.625 | -3.747 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.025 | Likely Benign | -0.61 | Neutral | 0.025 | Benign | 0.034 | Benign | 2.83 | Benign | 0.50 | Tolerated | 0.3458 | 0.4681 | 1 | 0 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||||||||||
| c.359G>A | G120D 2D  AIThe SynGAP1 missense variant G120D is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The high‑accuracy consensus, SGM‑Consensus, classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence overwhelmingly supports a benign classification, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.659993 | Binding | 0.359 | 0.887 | 0.750 | -3.483 | Likely Benign | 0.465 | Ambiguous | Likely Benign | 0.037 | Likely Benign | -0.57 | Neutral | 0.165 | Benign | 0.034 | Benign | 4.25 | Benign | 0.21 | Tolerated | 0.1847 | 0.2024 | 1 | -1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||||||||||||
| c.394T>G | F132V 2D  AIThe SynGAP1 missense variant F132V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect are PROVEAN, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized; ESM1b is uncertain. High‑accuracy assessments show AlphaMissense‑Optimized predicts pathogenicity, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also predicts pathogenic. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of high‑confidence predictions indicate a pathogenic effect, and this conclusion does not contradict the ClinVar status, which is currently unreported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.450668 | Structured | 0.727897 | Binding | 0.345 | 0.892 | 0.250 | -7.637 | In-Between | 0.979 | Likely Pathogenic | Likely Pathogenic | 0.328 | Likely Benign | -3.86 | Deleterious | 0.084 | Benign | 0.034 | Benign | 3.38 | Benign | 0.00 | Affected | 0.2349 | 0.1725 | -1 | -1 | 1.4 | -48.04 | ||||||||||||||||||||||||||||||||||||||||
| c.3962C>A | P1321Q 2D  AIThe SynGAP1 missense variant P1321Q is listed in ClinVar (ID 833687.0) as benign and is present in gnomAD (variant ID 6‑33451836‑C‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only polyPhen‑2 HumDiv predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification, and AlphaMissense‑Optimized also reports benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence—including the high‑confidence SGM consensus and AlphaMissense‑Optimized—supports a benign impact. This conclusion aligns with the ClinVar benign status, with no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.933505 | Binding | 0.463 | 0.828 | 0.875 | Benign | 1 | 6-33451836-C-A | 1 | 6.58e-7 | -5.594 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.055 | Likely Benign | -0.74 | Neutral | 0.659 | Possibly Damaging | 0.034 | Benign | 4.24 | Benign | 0.09 | Tolerated | 3.77 | 5 | 0.1839 | 0.4884 | 0 | -1 | -1.9 | 31.01 | ||||||||||||||||||||||||||||||||
| c.1312G>T | A438S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A438S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree that the substitution is benign: REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify it as benign. The only inconclusive results come from Rosetta and premPS, which are listed as uncertain. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also reports benign. Overall, the evidence strongly supports a benign effect, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.147574 | Structured | 0.290154 | Uncertain | 0.929 | 0.293 | 0.000 | -6.085 | Likely Benign | 0.105 | Likely Benign | Likely Benign | 0.30 | Likely Benign | 0.0 | 0.62 | Ambiguous | 0.46 | Likely Benign | 0.68 | Ambiguous | 0.012 | Likely Benign | -1.27 | Neutral | 0.042 | Benign | 0.035 | Benign | 4.14 | Benign | 0.15 | Tolerated | 0.2143 | 0.3995 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.2122C>G | L708V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L708V is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results are from FoldX (uncertain) and Foldetta (uncertain). High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also predicts benign, while Foldetta remains uncertain. Overall, the evidence strongly indicates that the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.250310 | Structured | 0.365875 | Uncertain | 0.931 | 0.378 | 0.000 | -4.361 | Likely Benign | 0.097 | Likely Benign | Likely Benign | 1.59 | Ambiguous | 0.0 | 0.12 | Likely Benign | 0.86 | Ambiguous | -0.02 | Likely Benign | 0.122 | Likely Benign | -0.23 | Neutral | 0.158 | Benign | 0.035 | Benign | 3.35 | Benign | 0.94 | Tolerated | 0.1094 | 0.2460 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||
| c.400A>G | S134G 2D  AIThe SynGAP1 missense variant S134G is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The high‑accuracy consensus (SGM‑Consensus) is derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yielding a “Likely Benign” classification. AlphaMissense‑Optimized also reports a benign prediction. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence from multiple independent predictors and the high‑accuracy consensus indicates that the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.447574 | Structured | 0.695837 | Binding | 0.333 | 0.898 | 0.250 | -6.722 | Likely Benign | 0.457 | Ambiguous | Likely Benign | 0.120 | Likely Benign | -2.19 | Neutral | 0.034 | Benign | 0.035 | Benign | 3.84 | Benign | 0.00 | Affected | 0.2268 | 0.4008 | 1 | 0 | 0.4 | -30.03 | |||||||||||||||||||||||||||||||||||||||
| c.1994A>T | Y665F 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant Y665F is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only ESM1b predicts a pathogenic outcome, while Rosetta and Foldetta are uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” status. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, and Foldetta as uncertain. Overall, the preponderance of evidence points to a benign impact. This conclusion is consistent with the absence of a ClinVar claim; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.098513 | Structured | 0.086641 | Uncertain | 0.922 | 0.361 | 0.000 | -8.460 | Likely Pathogenic | 0.100 | Likely Benign | Likely Benign | 0.07 | Likely Benign | 0.1 | 1.03 | Ambiguous | 0.55 | Ambiguous | 0.38 | Likely Benign | 0.106 | Likely Benign | -1.23 | Neutral | 0.159 | Benign | 0.036 | Benign | 3.45 | Benign | 0.47 | Tolerated | 0.2277 | 0.3076 | 7 | 3 | 4.1 | -16.00 | |||||||||||||||||||||||||||||
| c.2027G>A | S676N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S676N is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, Rosetta, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote). Only SIFT predicts a pathogenic outcome. Predictions that are inconclusive (FoldX, premPS, ESM1b) are treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus, derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates benign. Foldetta, which integrates FoldX‑MD and Rosetta outputs, is unavailable because one component (FoldX) is uncertain. Overall, the majority of evidence points to a benign effect, and this is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.209395 | Structured | 0.113632 | Uncertain | 0.551 | 0.338 | 0.125 | -7.486 | In-Between | 0.282 | Likely Benign | Likely Benign | 0.60 | Ambiguous | 0.3 | -0.21 | Likely Benign | 0.20 | Likely Benign | 0.55 | Ambiguous | 0.099 | Likely Benign | -1.63 | Neutral | 0.438 | Benign | 0.036 | Benign | 3.42 | Benign | 0.05 | Affected | 0.1454 | 0.4599 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||||
| c.239A>C | K80T 2D  AIThe SynGAP1 missense variant K80T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), which classifies the variant as Likely Benign. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, SIFT, and AlphaMissense‑Default. The high‑accuracy AlphaMissense‑Optimized prediction is uncertain, and the Foldetta protein‑folding stability assessment is unavailable. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of a ClinVar entry, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.477530 | Uncertain | 0.331 | 0.873 | 0.500 | -4.987 | Likely Benign | 0.830 | Likely Pathogenic | Ambiguous | 0.072 | Likely Benign | -1.60 | Neutral | 0.588 | Possibly Damaging | 0.036 | Benign | 3.89 | Benign | 0.00 | Affected | 0.2139 | 0.2321 | 0 | -1 | 3.2 | -27.07 | |||||||||||||||||||||||||||||||||||||||
| c.254C>A | T85K 2D  AIThe SynGAP1 missense variant T85K is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect include polyPhen‑2 HumDiv, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments are mixed: AlphaMissense‑Optimized predicts pathogenic, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields an equal split (2 pathogenic, 2 benign) and is therefore considered unavailable; Foldetta results are not provided and are likewise unavailable. Overall, the majority of available predictions (5 pathogenic vs. 4 benign) indicate that the variant is most likely pathogenic. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.680603 | Disordered | 0.542004 | Binding | 0.288 | 0.888 | 0.500 | -9.278 | Likely Pathogenic | 0.992 | Likely Pathogenic | Likely Pathogenic | 0.127 | Likely Benign | -2.32 | Neutral | 0.588 | Possibly Damaging | 0.036 | Benign | 3.88 | Benign | 0.00 | Affected | 0.0823 | 0.2796 | 0 | -1 | -3.2 | 27.07 | ||||||||||||||||||||||||||||||||||||||||
| c.265C>A | P89T 2D  AIThe SynGAP1 missense variant P89T has no ClinVar entry and is not reported in gnomAD. Functional prediction tools show a split: benign calls come from REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, and FATHMM, while pathogenic calls come from polyPhen‑2 HumDiv, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments further highlight this discordance: AlphaMissense‑Optimized predicts pathogenic, whereas the SGM‑Consensus (majority vote) predicts benign. Foldetta stability analysis is unavailable. Overall, the preponderance of evidence leans toward a benign effect, but the presence of a pathogenic prediction from AlphaMissense‑Optimized introduces uncertainty. This assessment does not contradict ClinVar status, as no ClinVar classification exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.703578 | Disordered | 0.545797 | Binding | 0.316 | 0.865 | 0.500 | -5.429 | Likely Benign | 0.979 | Likely Pathogenic | Likely Pathogenic | 0.105 | Likely Benign | -2.49 | Neutral | 0.588 | Possibly Damaging | 0.036 | Benign | 3.74 | Benign | 0.00 | Affected | 0.1786 | 0.4702 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.2663C>G | A888G 2D  AIThe SynGAP1 missense variant A888G is reported in gnomAD (ID 6‑33443215‑C‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic, representing a single dissenting opinion. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence points to a benign impact, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.784345 | Disordered | 0.575860 | Binding | 0.352 | 0.928 | 0.625 | 6-33443215-C-G | 5 | 3.10e-6 | -2.523 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.047 | Likely Benign | -0.32 | Neutral | 0.033 | Benign | 0.036 | Benign | 2.69 | Benign | 0.00 | Affected | 4.32 | 4 | 0.2312 | 0.5207 | 0 | 1 | -2.2 | -14.03 | 10.1016/j.ajhg.2020.11.011 | |||||||||||||||||||||||||||||||||
| c.2881C>A | H961N 2D  AIThe SynGAP1 missense variant H961N is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only ESM1b predicts a pathogenic outcome, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports the variant as likely benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence indicates that H961N is most likely benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.989835 | Disordered | 0.984562 | Binding | 0.323 | 0.893 | 0.750 | -8.561 | Likely Pathogenic | 0.096 | Likely Benign | Likely Benign | 0.084 | Likely Benign | -0.32 | Neutral | 0.069 | Benign | 0.036 | Benign | 4.17 | Benign | 0.81 | Tolerated | 0.2074 | 0.3695 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.2881C>G | H961D 2D  AIThe SynGAP1 missense variant H961D is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only ESM1b predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, and Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.989835 | Disordered | 0.984562 | Binding | 0.323 | 0.893 | 0.750 | -12.522 | Likely Pathogenic | 0.224 | Likely Benign | Likely Benign | 0.115 | Likely Benign | -0.98 | Neutral | 0.069 | Benign | 0.036 | Benign | 4.19 | Benign | 0.09 | Tolerated | 0.2487 | 0.2723 | 1 | -1 | -0.3 | -22.05 | |||||||||||||||||||||||||||||||||||||||
| c.3846G>C | E1282D 2D  AIThe SynGAP1 missense variant E1282D is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6-33447894-G-C). All available in silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic prediction. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant, so its status is unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, which is consistent with the ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.817364 | Binding | 0.465 | 0.725 | 0.875 | Uncertain | 1 | 6-33447894-G-C | 1 | 6.44e-7 | -3.879 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -1.26 | Neutral | 0.112 | Benign | 0.036 | Benign | 2.70 | Benign | 0.39 | Tolerated | 3.77 | 5 | 0.1826 | 0.3464 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||
| c.3846G>T | E1282D 2D  AIThe SynGAP1 missense variant E1282D is reported in gnomAD (ID 6‑33447894‑G‑T) but has no ClinVar entry. All in‑silico predictors reviewed—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—classify the change as benign. No tool predicts pathogenicity, so the benign group includes every listed predictor, while the pathogenic group is empty. High‑accuracy assessments further support a benign effect: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta results are unavailable. Overall, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.817364 | Binding | 0.465 | 0.725 | 0.875 | 6-33447894-G-T | -3.879 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -1.26 | Neutral | 0.112 | Benign | 0.036 | Benign | 2.70 | Benign | 0.39 | Tolerated | 3.77 | 5 | 0.1826 | 0.3464 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||||||||
| c.53A>C | Y18S 2D  AIThe SynGAP1 missense variant Y18S is reported in gnomAD (ID 6‑33420317‑A‑C) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it as pathogenic. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of prediction tools and the high‑accuracy methods points to a benign impact for Y18S. This conclusion is consistent with the absence of a pathogenic ClinVar classification, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.483068 | Structured | 0.446314 | Uncertain | 0.345 | 0.908 | 0.375 | 6-33420317-A-C | -1.061 | Likely Benign | 0.280 | Likely Benign | Likely Benign | 0.124 | Likely Benign | -0.50 | Neutral | 0.389 | Benign | 0.036 | Benign | 4.09 | Benign | 0.00 | Affected | 4.32 | 1 | 0.4961 | 0.2640 | -2 | -3 | 0.5 | -76.10 | ||||||||||||||||||||||||||||||||||||
| c.55G>A | A19T 2D  AIThe SynGAP1 missense variant A19T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact. The variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.529623 | Disordered | 0.443393 | Uncertain | 0.378 | 0.906 | 0.500 | -3.422 | Likely Benign | 0.131 | Likely Benign | Likely Benign | 0.030 | Likely Benign | -0.02 | Neutral | 0.371 | Benign | 0.036 | Benign | 4.14 | Benign | 0.00 | Affected | 0.2357 | 0.7248 | 1 | 0 | -2.5 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.56C>G | A19G 2D  AIThe SynGAP1 missense variant A19G is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The prediction is consistent with the lack of ClinVar annotation, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.529623 | Disordered | 0.443393 | Uncertain | 0.378 | 0.906 | 0.500 | -3.525 | Likely Benign | 0.194 | Likely Benign | Likely Benign | 0.037 | Likely Benign | -0.45 | Neutral | 0.371 | Benign | 0.036 | Benign | 4.10 | Benign | 0.00 | Affected | 0.2324 | 0.5447 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.56C>T | A19V 2D  AIThe SynGAP1 missense variant A19V is reported in gnomAD (ID 6‑33420320‑C‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it as pathogenic. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign; Foldetta results are unavailable. Overall, the consensus of available predictions points to a benign impact for A19V, and this conclusion is not contradicted by ClinVar status, which currently lacks an entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.529623 | Disordered | 0.443393 | Uncertain | 0.378 | 0.906 | 0.500 | 6-33420320-C-T | -3.157 | Likely Benign | 0.095 | Likely Benign | Likely Benign | 0.063 | Likely Benign | 0.42 | Neutral | 0.371 | Benign | 0.036 | Benign | 4.27 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1804 | 0.6942 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||||
| c.1148G>C | G383A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G383A is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are FoldX, Rosetta, and Foldetta. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, and Foldetta as pathogenic. Thus, the majority of evidence points to a benign impact, with only a minority of high‑accuracy tools suggesting pathogenicity. The variant is most likely benign based on the overall predictions, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.728858 | Disordered | 0.429104 | Uncertain | 0.296 | 0.949 | 0.750 | -6.205 | Likely Benign | 0.105 | Likely Benign | Likely Benign | 3.17 | Destabilizing | 1.2 | 3.47 | Destabilizing | 3.32 | Destabilizing | 0.04 | Likely Benign | 0.178 | Likely Benign | -0.64 | Neutral | 0.055 | Benign | 0.037 | Benign | 4.20 | Benign | 0.11 | Tolerated | 0.3880 | 0.4361 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||
| c.1313C>G | A438G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A438G is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote). Only PROVEAN predicts a pathogenic outcome. Predictions that are inconclusive or unavailable are FoldX, Rosetta, Foldetta, and premPS. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as Likely Benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact for A438G, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.147574 | Structured | 0.290154 | Uncertain | 0.929 | 0.293 | 0.000 | -5.790 | Likely Benign | 0.182 | Likely Benign | Likely Benign | 1.08 | Ambiguous | 0.1 | 1.78 | Ambiguous | 1.43 | Ambiguous | 0.80 | Ambiguous | 0.034 | Likely Benign | -2.51 | Deleterious | 0.247 | Benign | 0.037 | Benign | 4.12 | Benign | 0.11 | Tolerated | 0.1788 | 0.2719 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||
| c.1411T>G | S471A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S471A is not reported in ClinVar and is absent from gnomAD. Across the spectrum of in‑silico predictors, the majority (REVEL, FoldX, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus) classify the change as benign, whereas only FATHMM predicts it as pathogenic; Rosetta is inconclusive. High‑accuracy assessments reinforce the benign interpretation: AlphaMissense‑Optimized scores benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign stability. No evidence suggests pathogenicity, and the predictions do not contradict the absence of ClinVar annotation. Therefore, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.305330 | Structured | 0.355411 | Uncertain | 0.888 | 0.261 | 0.000 | -5.516 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.11 | Likely Benign | 0.1 | -0.61 | Ambiguous | -0.25 | Likely Benign | 0.23 | Likely Benign | 0.252 | Likely Benign | -1.93 | Neutral | 0.010 | Benign | 0.037 | Benign | -1.22 | Pathogenic | 0.29 | Tolerated | 0.4439 | 0.3691 | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||||||
| c.142T>G | F48V 2D  AIThe SynGAP1 missense variant F48V is not reported in ClinVar and is absent from gnomAD. Consensus from routine in silico predictors shows six tools (REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Optimized) classifying the change as benign, while two (SIFT, AlphaMissense‑Default) predict pathogenicity; ESM1b remains uncertain. High‑accuracy assessment further supports a benign outcome: AlphaMissense‑Optimized is benign, the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign majority vote, and Foldetta data are unavailable. Consequently, the overall evidence points to a benign effect for F48V, and this conclusion does not conflict with the absence of a ClinVar assertion. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.298791 | Structured | 0.440452 | Uncertain | 0.558 | 0.707 | 0.125 | -7.591 | In-Between | 0.756 | Likely Pathogenic | Likely Benign | 0.144 | Likely Benign | -1.87 | Neutral | 0.092 | Benign | 0.037 | Benign | 4.00 | Benign | 0.00 | Affected | 0.2533 | 0.2300 | -1 | -1 | 1.4 | -48.04 | ||||||||||||||||||||||||||||||||||||||||
| c.149T>C | I50T 2D  AIThe SynGAP1 missense variant I50T is not reported in ClinVar and has no entries in gnomAD. Functional prediction tools largely agree that the change is benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM all classify it as benign. In contrast, SIFT and AlphaMissense‑Default predict a pathogenic effect. AlphaMissense‑Optimized returns an uncertain result, and Foldetta (a protein‑folding stability method combining FoldX‑MD and Rosetta outputs) has no available prediction for this variant. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a “Likely Benign” verdict. High‑accuracy assessments therefore point to a benign outcome: AlphaMissense‑Optimized is inconclusive, SGM‑Consensus is benign, and Foldetta data are missing. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.295083 | Structured | 0.449965 | Uncertain | 0.545 | 0.708 | 0.000 | -5.121 | Likely Benign | 0.949 | Likely Pathogenic | Ambiguous | 0.128 | Likely Benign | -1.61 | Neutral | 0.092 | Benign | 0.037 | Benign | 3.76 | Benign | 0.00 | Affected | 0.0950 | 0.0708 | 0 | -1 | -5.2 | -12.05 | |||||||||||||||||||||||||||||||||||||||
| c.1963C>A | L655M 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L655M is not reported in ClinVar and is absent from gnomAD. All available in‑silico predictors classify the change as benign: REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). No tool predicts pathogenicity. High‑accuracy assessments are consistent: AlphaMissense‑Optimized predicts benign; the SGM Consensus indicates “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. Based on the unanimous benign predictions and the absence of any pathogenic calls, the variant is most likely benign, and this conclusion does not contradict the ClinVar status (which has no entry). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.015344 | Structured | 0.268808 | Uncertain | 0.961 | 0.274 | 0.000 | -3.077 | Likely Benign | 0.083 | Likely Benign | Likely Benign | 0.21 | Likely Benign | 0.0 | 0.25 | Likely Benign | 0.23 | Likely Benign | -0.22 | Likely Benign | 0.076 | Likely Benign | 0.58 | Neutral | 0.048 | Benign | 0.037 | Benign | 3.43 | Benign | 0.18 | Tolerated | 0.0979 | 0.3252 | 4 | 2 | -1.9 | 18.03 | |||||||||||||||||||||||||||||
| c.2029A>C | S677R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S677R is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Those that predict a pathogenic effect are FoldX, ESM1b, and AlphaMissense‑Default. Uncertain results come from Rosetta, Foldetta, and premPS. High‑accuracy assessments show AlphaMissense‑Optimized predicts a benign outcome, while the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta is also inconclusive. Overall, the majority of tools and the high‑accuracy benign prediction suggest the variant is most likely benign, and this assessment does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.194234 | Structured | 0.115685 | Uncertain | 0.555 | 0.338 | 0.125 | -9.388 | Likely Pathogenic | 0.714 | Likely Pathogenic | Likely Benign | 2.59 | Destabilizing | 0.1 | 0.73 | Ambiguous | 1.66 | Ambiguous | 0.71 | Ambiguous | 0.104 | Likely Benign | -2.49 | Neutral | 0.385 | Benign | 0.037 | Benign | 3.31 | Benign | 0.12 | Tolerated | 0.1120 | 0.4195 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||
| c.2031T>A | S677R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S677R is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and FATHMM. Those that predict a pathogenic effect are FoldX, ESM1b, and AlphaMissense‑Default. Tools with uncertain or inconclusive results are Rosetta, Foldetta, and premPS. High‑accuracy methods give mixed signals: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta is uncertain. Overall, the majority of consensus tools (six benign vs. three pathogenic) lean toward a benign interpretation. This prediction does not contradict any ClinVar status, as none is available. Thus, the variant is most likely benign based on current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.194234 | Structured | 0.115685 | Uncertain | 0.555 | 0.338 | 0.125 | -9.388 | Likely Pathogenic | 0.714 | Likely Pathogenic | Likely Benign | 2.59 | Destabilizing | 0.1 | 0.73 | Ambiguous | 1.66 | Ambiguous | 0.71 | Ambiguous | 0.150 | Likely Benign | -2.49 | Neutral | 0.385 | Benign | 0.037 | Benign | 3.31 | Benign | 0.12 | Tolerated | 0.1120 | 0.4195 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||
| c.2031T>G | S677R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S677R is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, and FATHMM. Those that predict a pathogenic effect are FoldX, ESM1b, and AlphaMissense‑Default. Tools with uncertain or inconclusive results are Rosetta, Foldetta, and premPS. High‑accuracy methods give mixed signals: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta is uncertain. Overall, the majority of consensus tools (six benign vs. three pathogenic) lean toward a benign interpretation. This prediction does not contradict any ClinVar status, as none is available. Thus, the variant is most likely benign based on current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.194234 | Structured | 0.115685 | Uncertain | 0.555 | 0.338 | 0.125 | -9.388 | Likely Pathogenic | 0.714 | Likely Pathogenic | Likely Benign | 2.59 | Destabilizing | 0.1 | 0.73 | Ambiguous | 1.66 | Ambiguous | 0.71 | Ambiguous | 0.150 | Likely Benign | -2.49 | Neutral | 0.385 | Benign | 0.037 | Benign | 3.31 | Benign | 0.12 | Tolerated | 0.1120 | 0.4195 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||
| c.2236G>A | V746M 2D  AIThe SynGAP1 missense variant V746M is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, was not available for this variant. Overall, the consensus of available predictions strongly suggests that V746M is most likely benign, and this conclusion is consistent with the lack of ClinVar evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.808535 | Disordered | 0.576597 | Binding | 0.336 | 0.867 | 0.875 | -4.194 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.010 | Likely Benign | -0.58 | Neutral | 0.065 | Benign | 0.037 | Benign | 2.79 | Benign | 0.60 | Tolerated | 0.0756 | 0.4576 | 2 | 1 | -2.3 | 32.06 | |||||||||||||||||||||||||||||||||||||||
| c.2342T>C | M781T 2D  AIThe SynGAP1 missense variant M781T is catalogued in gnomAD (ID 6‑33442894‑T‑C) and has no ClinVar entry. Across the evaluated in‑silico tools, every predictor classifies the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool reports a pathogenic or likely pathogenic outcome. The high‑accuracy consensus methods reinforce this view: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability predictor, has no available result for this variant. Based on the unanimous benign predictions and the lack of any ClinVar pathogenic annotation, the variant is most likely benign and does not contradict ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.580690 | Disordered | 0.792850 | Binding | 0.342 | 0.889 | 0.625 | 6-33442894-T-C | 1 | 6.21e-7 | -3.774 | Likely Benign | 0.310 | Likely Benign | Likely Benign | 0.175 | Likely Benign | -0.86 | Neutral | 0.015 | Benign | 0.037 | Benign | 2.75 | Benign | 0.59 | Tolerated | 3.64 | 6 | 0.2267 | 0.1599 | -1 | -1 | -2.6 | -30.09 | ||||||||||||||||||||||||||||||||||
| c.2563C>G | L855V 2D  AIThe SynGAP1 missense variant L855V is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact. This conclusion is consistent with the lack of a ClinVar entry, so there is no contradiction with existing clinical annotations. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.724957 | Disordered | 0.485558 | Uncertain | 0.285 | 0.823 | 0.625 | -3.866 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.086 | Likely Benign | -0.71 | Neutral | 0.059 | Benign | 0.037 | Benign | 4.10 | Benign | 0.56 | Tolerated | 0.1621 | 0.3731 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2578G>C | V860L 2D  AIThe SynGAP1 missense variant V860L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.545602 | Disordered | 0.518121 | Binding | 0.269 | 0.803 | 0.250 | -3.053 | Likely Benign | 0.123 | Likely Benign | Likely Benign | 0.028 | Likely Benign | -0.89 | Neutral | 0.124 | Benign | 0.037 | Benign | 4.17 | Benign | 0.00 | Affected | 0.1052 | 0.5446 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2749C>G | P917A 2D  AIThe SynGAP1 missense variant P917A is not reported in ClinVar and is present in gnomAD (ID 6‑33443301‑C‑G). Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote) all classify the change as benign or likely benign. Only SIFT predicts a pathogenic effect. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized reports benign, and the SGM‑Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the collective predictions point to a benign impact, and this conclusion does not contradict the ClinVar status, which currently has no entry for P917A. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.599170 | Disordered | 0.863949 | Binding | 0.314 | 0.862 | 0.375 | 6-33443301-C-G | 1 | 6.20e-7 | -3.681 | Likely Benign | 0.062 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -1.33 | Neutral | 0.065 | Benign | 0.037 | Benign | 2.72 | Benign | 0.00 | Affected | 3.77 | 5 | 0.3458 | 0.4772 | -1 | 1 | 3.4 | -26.04 | ||||||||||||||||||||||||||||||||||
| c.321G>C | R107S 2D  AIThe SynGAP1 missense variant R107S is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized classifying the variant as pathogenic, while the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome; Foldetta results are unavailable. Overall, the majority of predictions lean toward a benign impact, and this is not contradicted by any ClinVar annotation. Thus, the variant is most likely benign, although high‑accuracy tools provide conflicting evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.666105 | Disordered | 0.663448 | Binding | 0.331 | 0.863 | 0.875 | -1.162 | Likely Benign | 0.995 | Likely Pathogenic | Likely Pathogenic | 0.147 | Likely Benign | -2.37 | Neutral | 0.231 | Benign | 0.037 | Benign | 2.99 | Benign | 0.00 | Affected | 0.2771 | 0.4148 | 0 | -1 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||||
| c.321G>T | R107S 2D  AIThe SynGAP1 missense variant R107S is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome; Foldetta results are unavailable. Overall, the majority of predictions lean toward a benign impact, but the high‑accuracy tools provide conflicting evidence. Thus, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.666105 | Disordered | 0.663448 | Binding | 0.331 | 0.863 | 0.875 | -1.162 | Likely Benign | 0.995 | Likely Pathogenic | Likely Pathogenic | 0.147 | Likely Benign | -2.37 | Neutral | 0.231 | Benign | 0.037 | Benign | 2.99 | Benign | 0.00 | Affected | 0.2771 | 0.4148 | 0 | -1 | 3.7 | -69.11 | |||||||||||||||||||||||||||||||||||||||
| c.3274T>G | L1092V 2D  AIThe SynGAP1 missense variant L1092V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The high‑accuracy consensus (SGM‑Consensus) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.924947 | Disordered | 0.985431 | Binding | 0.385 | 0.890 | 1.000 | -4.906 | Likely Benign | 0.451 | Ambiguous | Likely Benign | 0.034 | Likely Benign | -0.36 | Neutral | 0.051 | Benign | 0.037 | Benign | 2.79 | Benign | 0.37 | Tolerated | 0.1658 | 0.3912 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.407G>T | R136L 2D  AIThe SynGAP1 missense variant R136L is catalogued in gnomAD (ID 6‑33432704‑G‑T) but has no ClinVar entry. Functional prediction tools cluster into two groups: benign predictions come from REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM; pathogenic predictions arise from PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Pathogenic. High‑accuracy assessments further support a deleterious effect: AlphaMissense‑Optimized predicts Pathogenic, and the SGM‑Consensus (majority vote) also indicates Likely Pathogenic. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence points to a pathogenic impact, and this assessment does not conflict with ClinVar, which currently has no classification for R136L. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.433034 | Structured | 0.657394 | Binding | 0.351 | 0.894 | 0.250 | 6-33432704-G-T | 1 | 7.05e-7 | -11.512 | Likely Pathogenic | 0.987 | Likely Pathogenic | Likely Pathogenic | 0.347 | Likely Benign | -4.19 | Deleterious | 0.190 | Benign | 0.037 | Benign | 3.48 | Benign | 0.01 | Affected | 3.61 | 5 | 0.1718 | 0.4475 | -2 | -3 | 8.3 | -43.03 | ||||||||||||||||||||||||||||||||||
| c.440A>T | Q147L 2D  AIThe SynGAP1 missense variant Q147L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default; the SGM‑Consensus score is “Likely Pathogenic.” High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates likely pathogenic. Foldetta results are unavailable. Overall, the majority of evidence points toward a pathogenic interpretation, and this is not contradicted by any ClinVar classification because none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.541878 | Disordered | 0.503877 | Binding | 0.349 | 0.840 | 0.625 | -9.879 | Likely Pathogenic | 0.740 | Likely Pathogenic | Likely Benign | 0.185 | Likely Benign | -3.51 | Deleterious | 0.079 | Benign | 0.037 | Benign | 3.92 | Benign | 0.03 | Affected | 0.0861 | 0.4927 | -2 | -2 | 7.3 | -14.97 | |||||||||||||||||||||||||||||||||||||||
| c.512C>A | A171D 2D  AIThe SynGAP1 missense variant A171D is not reported in ClinVar and has no entries in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain, and no Foldetta stability assessment is available. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments therefore lean toward a benign interpretation: AlphaMissense‑Optimized is inconclusive, SGM‑Consensus is benign, and Foldetta data are missing. Based on the collective predictions, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.562014 | Disordered | 0.492272 | Uncertain | 0.358 | 0.652 | 0.375 | -6.977 | Likely Benign | 0.908 | Likely Pathogenic | Ambiguous | 0.144 | Likely Benign | -1.45 | Neutral | 0.244 | Benign | 0.037 | Benign | 4.15 | Benign | 0.01 | Affected | 0.1634 | 0.2462 | 0 | -2 | -5.3 | 44.01 | |||||||||||||||||||||||||||||||||||||||
| c.589G>A | E197K 2D  AIThe SynGAP1 missense variant E197K is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show a split: benign calls come from REVEL, polyPhen‑2 (HumDiv and HumVar) and FATHMM, whereas pathogenic calls arise from PROVEAN, SIFT, ESM1b, AlphaMissense‑Default and AlphaMissense‑Optimized. Grouping by consensus, the majority of high‑confidence predictors (AlphaMissense‑Optimized, SGM‑Consensus, PROVEAN, SIFT, ESM1b) indicate pathogenicity, while a minority (REVEL, polyPhen‑2, FATHMM) suggest benign impact. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM and PROVEAN, is classified as Likely Pathogenic. AlphaMissense‑Optimized also predicts Pathogenic. Foldetta results are unavailable. Overall, the preponderance of evidence points to a pathogenic effect for E197K, and this assessment does not conflict with ClinVar, which currently has no entry for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.349426 | Structured | 0.431896 | Uncertain | 0.452 | 0.492 | 0.125 | -11.045 | Likely Pathogenic | 0.962 | Likely Pathogenic | Likely Pathogenic | 0.198 | Likely Benign | -2.50 | Deleterious | 0.118 | Benign | 0.037 | Benign | 4.09 | Benign | 0.02 | Affected | 0.1824 | 0.5890 | 0 | 1 | -0.4 | -0.94 | |||||||||||||||||||||||||||||||||||||||
| c.590A>G | E197G 2D  AIThe SynGAP1 missense variant E197G is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on benign impact include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict pathogenicity are PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. The high‑accuracy consensus, SGM‑Consensus, classifies the variant as Likely Pathogenic, while AlphaMissense‑Optimized remains benign; Foldetta results are unavailable. Overall, the majority of individual predictors lean toward pathogenicity, and the SGM‑Consensus supports this view. Therefore, the variant is most likely pathogenic, and this assessment does not contradict any ClinVar annotation because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.349426 | Structured | 0.431896 | Uncertain | 0.452 | 0.492 | 0.125 | -8.480 | Likely Pathogenic | 0.749 | Likely Pathogenic | Likely Benign | 0.165 | Likely Benign | -3.13 | Deleterious | 0.118 | Benign | 0.037 | Benign | 4.03 | Benign | 0.01 | Affected | 0.2638 | 0.4615 | 0 | -2 | 3.1 | -72.06 | |||||||||||||||||||||||||||||||||||||||
| c.1061C>G | A354G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A354G is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. Predictions marked uncertain (Rosetta, Foldetta, premPS) are treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as likely benign, while Foldetta’s stability analysis is inconclusive. Overall, the majority of evidence points to a benign impact for A354G, and this conclusion does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.203355 | Structured | 0.381329 | Uncertain | 0.882 | 0.335 | 0.125 | -4.285 | Likely Benign | 0.113 | Likely Benign | Likely Benign | 0.41 | Likely Benign | 0.0 | 1.24 | Ambiguous | 0.83 | Ambiguous | 0.65 | Ambiguous | 0.037 | Likely Benign | -1.87 | Neutral | 0.146 | Benign | 0.038 | Benign | 1.75 | Pathogenic | 0.05 | Affected | 0.2563 | 0.4777 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||
| c.1061C>T | A354V 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant A354V is not reported in ClinVar and is present in the gnomAD database (variant ID 6‑33437966‑C‑T). Functional prediction tools that agree on a benign effect include REVEL, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The high‑accuracy consensus methods support a benign interpretation: AlphaMissense‑Optimized is benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is also benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, predicts a benign outcome. Predictions from FoldX and Rosetta are inconclusive and are treated as unavailable. Overall, the majority of evidence indicates that A354V is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.203355 | Structured | 0.381329 | Uncertain | 0.882 | 0.335 | 0.125 | 6-33437966-C-T | 1 | 6.26e-7 | -6.223 | Likely Benign | 0.122 | Likely Benign | Likely Benign | 0.65 | Ambiguous | 0.1 | -1.02 | Ambiguous | -0.19 | Likely Benign | 0.18 | Likely Benign | 0.027 | Likely Benign | -1.11 | Neutral | 0.146 | Benign | 0.038 | Benign | 1.81 | Pathogenic | 0.05 | Affected | 3.38 | 24 | 0.1429 | 0.6388 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||
| c.1097C>A | T366N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense change T366N is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote) all indicate a benign or likely benign outcome. Only two tools—polyPhen‑2 HumDiv and FATHMM—suggest a pathogenic effect, while premPS remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also reports likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) classifies the variant as benign. Taken together, the majority of evidence supports a benign impact and is consistent with the absence of any ClinVar pathogenic annotation. Therefore, the variant is most likely benign and does not contradict ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.414856 | Structured | 0.441902 | Uncertain | 0.897 | 0.642 | 0.250 | -5.694 | Likely Benign | 0.176 | Likely Benign | Likely Benign | 0.06 | Likely Benign | 0.0 | 0.36 | Likely Benign | 0.21 | Likely Benign | 0.92 | Ambiguous | 0.038 | Likely Benign | -1.72 | Neutral | 0.454 | Possibly Damaging | 0.038 | Benign | 1.72 | Pathogenic | 0.34 | Tolerated | 0.1595 | 0.4934 | 0 | 0 | -2.8 | 13.00 | |||||||||||||||||||||||||||||
| c.1180A>G | K394E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant K394E is not listed in ClinVar (ClinVar ID None) but is present in gnomAD (ID 6‑33438085‑A‑G). Prediction tools that agree on a benign effect include REVEL, FoldX, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM. Those that predict a pathogenic effect are premPS, PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments are inconclusive: AlphaMissense‑Optimized is uncertain; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is a 2‑vs‑2 tie, and Foldetta is uncertain. No prediction or folding‑stability result is available that decisively supports either outcome. Overall, the majority of tools (six benign vs four pathogenic) lean toward a benign interpretation, and this assessment does not contradict the absence of a ClinVar classification. Thus, the variant is most likely benign based on current predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.505461 | Disordered | 0.399336 | Uncertain | 0.387 | 0.634 | 0.625 | 6-33438085-A-G | 1 | 6.20e-7 | -6.903 | Likely Benign | 0.896 | Likely Pathogenic | Ambiguous | 0.07 | Likely Benign | 0.1 | 3.71 | Destabilizing | 1.89 | Ambiguous | 1.20 | Destabilizing | 0.446 | Likely Benign | -2.54 | Deleterious | 0.063 | Benign | 0.038 | Benign | 4.61 | Benign | 0.04 | Affected | 3.44 | 14 | 0.4556 | 0.1916 | 1 | 0 | 0.4 | 0.94 | |||||||||||||||||||||||||
| c.1321G>A | V441I 2D  AIThe SynGAP1 missense variant V441I is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign, while only ESM1b predicts it as pathogenic. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized reports benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) also indicates benign stability. No contradictory evidence is present. Based on the collective predictions, the variant is most likely benign, and this conclusion does not conflict with the absence of a ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.161087 | Structured | 0.259875 | Uncertain | 0.918 | 0.249 | 0.000 | -8.773 | Likely Pathogenic | 0.122 | Likely Benign | Likely Benign | -0.24 | Likely Benign | 0.3 | 0.11 | Likely Benign | -0.07 | Likely Benign | 0.25 | Likely Benign | 0.135 | Likely Benign | -0.82 | Neutral | 0.287 | Benign | 0.038 | Benign | 3.41 | Benign | 0.16 | Tolerated | 0.0694 | 0.3412 | 4 | 3 | 0.3 | 14.03 | |||||||||||||||||||||||||||||
| c.1832T>C | M611T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M611T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33440884‑T‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. Four tools (FoldX, Rosetta, Foldetta, premPS) return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact, and this does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.236433 | Structured | 0.210791 | Uncertain | 0.870 | 0.253 | 0.000 | Uncertain | 1 | 6-33440884-T-C | 1 | 6.19e-7 | -5.696 | Likely Benign | 0.101 | Likely Benign | Likely Benign | 1.98 | Ambiguous | 0.2 | 0.94 | Ambiguous | 1.46 | Ambiguous | 0.87 | Ambiguous | 0.240 | Likely Benign | -2.40 | Neutral | 0.034 | Benign | 0.038 | Benign | -1.19 | Pathogenic | 0.29 | Tolerated | 3.37 | 35 | 0.1635 | 0.1415 | -1 | -1 | -2.6 | -30.09 | ||||||||||||||||||||||
| c.1927G>C | E643Q 2D  AIThe SynGAP1 missense variant E643Q is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SGM‑Consensus (Likely Pathogenic), PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized predicts a benign outcome, while premPS is uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta as benign. Overall, the majority of predictions (8 benign vs. 5 pathogenic) and the two of three high‑accuracy tools favor a benign classification. Thus, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.033407 | Structured | 0.215915 | Uncertain | 0.871 | 0.315 | 0.000 | -12.404 | Likely Pathogenic | 0.688 | Likely Pathogenic | Likely Benign | 0.49 | Likely Benign | 0.6 | 0.15 | Likely Benign | 0.32 | Likely Benign | 0.83 | Ambiguous | 0.341 | Likely Benign | -2.86 | Deleterious | 0.446 | Benign | 0.038 | Benign | 2.94 | Benign | 0.01 | Affected | 0.1603 | 0.6308 | 2 | 2 | 0.0 | -0.98 | |||||||||||||||||||||||||||||
| c.2230C>G | Q744E 2D  AIThe SynGAP1 missense variant Q744E is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” while Foldetta results are unavailable. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.852992 | Disordered | 0.540428 | Binding | 0.316 | 0.866 | 0.875 | -4.053 | Likely Benign | 0.100 | Likely Benign | Likely Benign | 0.060 | Likely Benign | -0.57 | Neutral | 0.065 | Benign | 0.038 | Benign | 2.77 | Benign | 0.08 | Tolerated | 0.1453 | 0.1892 | 2 | 2 | 0.0 | 0.98 | |||||||||||||||||||||||||||||||||||||||
| c.2408A>G | K803R 2D  AIThe SynGAP1 missense variant K803R is listed in ClinVar (ID 834618.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: SGM‑Consensus, REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only two tools—SIFT and FATHMM—predict pathogenicity. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates the variant is most likely benign, which does not contradict the current ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.827927 | Disordered | 0.733908 | Binding | 0.349 | 0.900 | 0.625 | Uncertain | 1 | -2.281 | Likely Benign | 0.097 | Likely Benign | Likely Benign | 0.018 | Likely Benign | -1.52 | Neutral | 0.103 | Benign | 0.038 | Benign | 2.38 | Pathogenic | 0.00 | Affected | 3.77 | 5 | 0.4857 | 0.1715 | 3 | 2 | -0.6 | 28.01 | ||||||||||||||||||||||||||||||||||
| c.2496G>C | Q832H 2D  AIThe SynGAP1 missense variant Q832H is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.720929 | Disordered | 0.619913 | Binding | 0.290 | 0.877 | 0.375 | -3.322 | Likely Benign | 0.197 | Likely Benign | Likely Benign | 0.071 | Likely Benign | -1.24 | Neutral | 0.064 | Benign | 0.038 | Benign | 2.76 | Benign | 0.03 | Affected | 0.1263 | 0.3230 | 3 | 0 | 0.3 | 9.01 | |||||||||||||||||||||||||||||||||||||||
| c.2496G>T | Q832H 2D  AIThe SynGAP1 missense variant Q832H is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.720929 | Disordered | 0.619913 | Binding | 0.290 | 0.877 | 0.375 | -3.322 | Likely Benign | 0.197 | Likely Benign | Likely Benign | 0.071 | Likely Benign | -1.24 | Neutral | 0.064 | Benign | 0.038 | Benign | 2.76 | Benign | 0.03 | Affected | 0.1263 | 0.3230 | 3 | 0 | 0.3 | 9.01 | |||||||||||||||||||||||||||||||||||||||
| c.266C>A | P89Q 2D  AIThe SynGAP1 missense variant P89Q is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that agree on a pathogenic effect are PROVEAN, polyPhen‑2 HumDiv, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized predicts pathogenic. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive (2 pathogenic vs. 2 benign). Foldetta results are unavailable. Overall, the balance of evidence (five pathogenic vs. four benign predictions) indicates the variant is most likely pathogenic, and this assessment does not contradict any ClinVar status because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.703578 | Disordered | 0.545797 | Binding | 0.316 | 0.865 | 0.500 | -4.779 | Likely Benign | 0.987 | Likely Pathogenic | Likely Pathogenic | 0.107 | Likely Benign | -2.63 | Deleterious | 0.642 | Possibly Damaging | 0.038 | Benign | 3.74 | Benign | 0.00 | Affected | 0.1633 | 0.4310 | 0 | -1 | -1.9 | 31.01 | ||||||||||||||||||||||||||||||||||||||||
| c.278G>C | R93P 2D  AIThe SynGAP1 missense variant R93P is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, SGM‑Consensus indicates Likely Benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.549151 | Binding | 0.290 | 0.874 | 0.625 | -3.164 | Likely Benign | 0.473 | Ambiguous | Likely Benign | 0.121 | Likely Benign | -0.32 | Neutral | 0.361 | Benign | 0.038 | Benign | 3.99 | Benign | 0.00 | Affected | 0.2200 | 0.4844 | 0 | -2 | 2.9 | -59.07 | |||||||||||||||||||||||||||||||||||||||
| c.2974G>C | V992L 2D  AIThe SynGAP1 missense variant V992L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the evidence strongly supports a benign classification, and this conclusion is consistent with the lack of a ClinVar entry, so there is no contradiction with existing clinical annotations. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.921728 | Binding | 0.331 | 0.917 | 0.750 | -2.333 | Likely Benign | 0.086 | Likely Benign | Likely Benign | 0.060 | Likely Benign | -0.50 | Neutral | 0.086 | Benign | 0.038 | Benign | 4.24 | Benign | 0.16 | Tolerated | 0.1211 | 0.5202 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3703A>G | M1235V 2D  AIThe SynGAP1 missense variant M1235V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as tolerated or benign. Only SIFT predicts a damaging effect. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this residue. Taken together, the preponderance of evidence indicates that M1235V is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.690604 | Disordered | 0.577958 | Binding | 0.872 | 0.532 | 0.125 | -3.870 | Likely Benign | 0.099 | Likely Benign | Likely Benign | 0.055 | Likely Benign | -1.54 | Neutral | 0.139 | Benign | 0.038 | Benign | 2.69 | Benign | 0.02 | Affected | 0.3048 | 0.3120 | 2 | 1 | 2.3 | -32.06 | ||||||||||||||||||||||||||||||||||||||
| c.379C>G | R127G 2D  AIThe SynGAP1 R127G missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The high‑accuracy consensus, SGM‑Consensus, is derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN; it yields a “Likely Benign” classification (3 benign vs. 1 pathogenic). AlphaMissense‑Optimized also predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this assessment does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.497853 | Structured | 0.711716 | Binding | 0.333 | 0.870 | 0.625 | -1.062 | Likely Benign | 0.700 | Likely Pathogenic | Likely Benign | 0.107 | Likely Benign | -2.17 | Neutral | 0.287 | Benign | 0.038 | Benign | 3.91 | Benign | 0.01 | Affected | 0.3561 | 0.3037 | -3 | -2 | 4.1 | -99.14 | |||||||||||||||||||||||||||||||||||||||
| c.3874C>G | L1292V 2D  AIThe SynGAP1 missense variant L1292V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this conclusion does not contradict any ClinVar annotation (none present). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.779859 | Disordered | 0.882643 | Binding | 0.586 | 0.800 | 0.625 | -4.943 | Likely Benign | 0.121 | Likely Benign | Likely Benign | 0.049 | Likely Benign | -0.14 | Neutral | 0.058 | Benign | 0.038 | Benign | 2.67 | Benign | 0.07 | Tolerated | 0.1769 | 0.2605 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.1178G>C | G393A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G393A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only FATHMM predicts a pathogenic outcome. Stability‑based methods (FoldX, Rosetta, Foldetta) are inconclusive, so they provide no evidence for or against pathogenicity. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as likely benign, and Foldetta as uncertain. Overall, the majority of reliable predictions indicate a benign effect, and there is no ClinVar annotation to contradict this assessment. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.538167 | Disordered | 0.402365 | Uncertain | 0.333 | 0.670 | 0.625 | -4.507 | Likely Benign | 0.129 | Likely Benign | Likely Benign | 1.93 | Ambiguous | 0.5 | -0.68 | Ambiguous | 0.63 | Ambiguous | 0.22 | Likely Benign | 0.381 | Likely Benign | -1.89 | Neutral | 0.176 | Benign | 0.039 | Benign | 1.33 | Pathogenic | 0.08 | Tolerated | 0.4143 | 0.5193 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||
| c.1195G>A | A399T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A399T is listed in ClinVar (ID 1990638.0) as Benign and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from FoldX, Rosetta, and Foldetta, which are treated as unavailable evidence. High‑accuracy assessments show AlphaMissense‑Optimized as Benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign, and Foldetta as Uncertain. Overall, the variant is most likely benign, and this conclusion aligns with its ClinVar benign classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.394753 | Structured | 0.407674 | Uncertain | 0.939 | 0.490 | 0.125 | Benign | 1 | -5.236 | Likely Benign | 0.114 | Likely Benign | Likely Benign | 1.24 | Ambiguous | 0.1 | 0.91 | Ambiguous | 1.08 | Ambiguous | 0.49 | Likely Benign | 0.272 | Likely Benign | -0.40 | Neutral | 0.131 | Benign | 0.039 | Benign | 5.41 | Benign | 0.69 | Tolerated | 3.38 | 26 | 0.1335 | 0.6477 | 1 | 0 | -2.5 | 30.03 | 211.4 | -41.4 | 0.0 | 0.0 | 0.6 | 0.4 | X | Potentially Pathogenic | The methyl group of Ala399, located in an anti-parallel β sheet strand (res. Ala399-Ile411), is swapped for a hydroxyl-containing threonine. In the variant simulations, the hydroxyl group of Thr399 can form H-bonds with the backbone atoms of the residues in the membrane-facing loops (e.g., Gly382) in the C2 domain. Consequently, the ability of the Thr399 side chain to form H-bonds with the membrane-facing loops could adversely affect the dynamics and stability of the SynGAP-membrane association. However, since the effects on the dynamics of the membrane-facing loops can only be studied through the SynGAP-membrane complex, no definite conclusions can be drawn. | ||||||||||||||||
| c.151A>T | I51F 2D  AIThe SynGAP1 missense variant I51F is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, SGM‑Consensus indicates Likely Benign, and Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign effect for I51F, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.291804 | Structured | 0.454181 | Uncertain | 0.606 | 0.710 | 0.000 | -5.687 | Likely Benign | 0.526 | Ambiguous | Likely Benign | 0.114 | Likely Benign | -0.87 | Neutral | 0.099 | Benign | 0.039 | Benign | 4.13 | Benign | 0.00 | Affected | 0.0565 | 0.3070 | 1 | 0 | -1.7 | 34.02 | |||||||||||||||||||||||||||||||||||||||
| c.1597G>T | A533S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A533S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. Only FATHMM predicts a pathogenic outcome. When predictions are grouped by consensus, the benign group contains all tools except FATHMM, which stands alone in the pathogenic group. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized reports benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) also indicates a benign effect. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.179055 | Structured | 0.026324 | Uncertain | 0.843 | 0.393 | 0.000 | -3.740 | Likely Benign | 0.075 | Likely Benign | Likely Benign | 0.24 | Likely Benign | 0.0 | -0.05 | Likely Benign | 0.10 | Likely Benign | -0.14 | Likely Benign | 0.193 | Likely Benign | 0.33 | Neutral | 0.009 | Benign | 0.039 | Benign | -1.14 | Pathogenic | 0.45 | Tolerated | 0.2612 | 0.5334 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.172A>C | M58L 2D  AIThe SynGAP1 missense variant M58L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as tolerated or benign. Only SIFT predicts a deleterious impact. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized scores benign, and the SGM‑Consensus result is benign; Foldetta data are not available. Consequently, the aggregate evidence points to a benign effect for M58L, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.188120 | Structured | 0.484415 | Uncertain | 0.515 | 0.665 | 0.000 | -0.661 | Likely Benign | 0.240 | Likely Benign | Likely Benign | 0.208 | Likely Benign | -0.11 | Neutral | 0.006 | Benign | 0.039 | Benign | 4.77 | Benign | 0.00 | Affected | 0.1514 | 0.4621 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.172A>T | M58L 2D  AIThe SynGAP1 missense variant M58L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this is consistent with the lack of ClinVar annotation—there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.188120 | Structured | 0.484415 | Uncertain | 0.515 | 0.665 | 0.000 | -0.661 | Likely Benign | 0.240 | Likely Benign | Likely Benign | 0.208 | Likely Benign | -0.11 | Neutral | 0.006 | Benign | 0.039 | Benign | 4.77 | Benign | 0.00 | Affected | 0.1514 | 0.4621 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.1957C>G | L653V 2D  AIThe SynGAP1 missense variant L653V is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that classify the variant as benign include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are FoldX, Rosetta, and premPS, while ESM1b is inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) as Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as pathogenic. Overall, the majority of evidence points to a benign impact, and this does not contradict the ClinVar “Uncertain” classification. Thus, based on current predictions, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.049374 | Structured | 0.335213 | Uncertain | 0.963 | 0.332 | 0.000 | Uncertain | 1 | -7.050 | In-Between | 0.301 | Likely Benign | Likely Benign | 3.28 | Destabilizing | 0.3 | 2.18 | Destabilizing | 2.73 | Destabilizing | 1.32 | Destabilizing | 0.146 | Likely Benign | -2.25 | Neutral | 0.227 | Benign | 0.039 | Benign | 3.28 | Benign | 0.08 | Tolerated | 0.1436 | 0.3557 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||
| c.2590G>T | A864S 2D  AIThe SynGAP1 missense variant A864S is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.549308 | Disordered | 0.611966 | Binding | 0.269 | 0.788 | 0.250 | -3.169 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.035 | Likely Benign | 0.08 | Neutral | 0.112 | Benign | 0.039 | Benign | 2.50 | Benign | 0.16 | Tolerated | 0.2792 | 0.6196 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.2821C>T | P941S 2D  AIThe SynGAP1 missense variant P941S is reported in gnomAD (ID 6‑33443373‑C‑T) but has no ClinVar entry. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, which evaluates protein‑folding stability via FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of all available predictions indicates that P941S is most likely benign, and this assessment does not contradict any ClinVar status (none). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.874069 | Disordered | 0.900790 | Binding | 0.403 | 0.906 | 0.625 | 6-33443373-C-T | 1 | 6.20e-7 | -5.099 | Likely Benign | 0.062 | Likely Benign | Likely Benign | 0.039 | Likely Benign | 0.58 | Neutral | 0.036 | Benign | 0.039 | Benign | 3.05 | Benign | 0.95 | Tolerated | 4.32 | 4 | 0.2759 | 0.5576 | -1 | 1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||||
| c.2822C>T | P941L 2D  AIThe SynGAP1 missense variant P941L is listed in ClinVar (ID 3451960.0) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). The only tool that predicts a pathogenic outcome is SIFT. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates “Likely Benign.” No Foldetta (FoldX‑MD/Rosetta) stability result is available, so it does not influence the assessment. Overall, the majority of predictions, including the high‑accuracy tools, point to a benign effect, which is consistent with the ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.874069 | Disordered | 0.900790 | Binding | 0.403 | 0.906 | 0.625 | Uncertain | 2 | -5.692 | Likely Benign | 0.066 | Likely Benign | Likely Benign | 0.054 | Likely Benign | -0.44 | Neutral | 0.144 | Benign | 0.039 | Benign | 2.76 | Benign | 0.01 | Affected | 0.2196 | 0.5931 | -3 | -3 | 5.4 | 16.04 | |||||||||||||||||||||||||||||||||||||
| c.2827G>A | G943S 2D  AIThe SynGAP1 missense variant G943S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign. Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.973328 | Disordered | 0.860437 | Binding | 0.372 | 0.910 | 0.750 | -5.785 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.295 | Likely Benign | 0.34 | Neutral | 0.059 | Benign | 0.039 | Benign | 2.88 | Benign | 0.51 | Tolerated | 0.2482 | 0.5502 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.2866T>G | S956A 2D  AIThe SynGAP1 missense variant S956A is not reported in ClinVar or gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify it as benign, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports it as likely benign. Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta stability analysis is unavailable. Overall, the collective evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.984871 | Disordered | 0.957345 | Binding | 0.364 | 0.917 | 0.750 | -4.468 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.111 | Likely Benign | -0.47 | Neutral | 0.112 | Benign | 0.039 | Benign | 2.18 | Pathogenic | 0.13 | Tolerated | 0.4239 | 0.4838 | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||||||||||||||||
| c.2980A>G | K994E 2D  AIThe SynGAP1 missense variant K994E is reported in gnomAD (variant ID 6‑33443532‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of tools, including the high‑accuracy methods, points to a benign impact. This prediction does not contradict any ClinVar status, as none is assigned. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.862302 | Disordered | 0.930054 | Binding | 0.289 | 0.912 | 0.750 | 6-33443532-A-G | 1 | 6.20e-7 | -2.587 | Likely Benign | 0.339 | Likely Benign | Likely Benign | 0.057 | Likely Benign | -0.58 | Neutral | 0.036 | Benign | 0.039 | Benign | 4.14 | Benign | 0.02 | Affected | 4.32 | 2 | 0.4148 | 0.1699 | 1 | 0 | 0.4 | 0.94 | ||||||||||||||||||||||||||||||||||
| c.2986C>A | P996T 2D  AIThe SynGAP1 missense variant P996T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.775545 | Disordered | 0.942262 | Binding | 0.312 | 0.900 | 0.750 | -5.138 | Likely Benign | 0.058 | Likely Benign | Likely Benign | 0.062 | Likely Benign | -0.71 | Neutral | 0.036 | Benign | 0.039 | Benign | 4.26 | Benign | 0.03 | Affected | 0.1418 | 0.6163 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.3040G>A | G1014S 2D  AIThe SynGAP1 missense variant G1014S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Overall, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.771762 | Disordered | 0.914808 | Binding | 0.293 | 0.835 | 0.625 | -3.219 | Likely Benign | 0.085 | Likely Benign | Likely Benign | 0.026 | Likely Benign | -0.60 | Neutral | 0.068 | Benign | 0.039 | Benign | 2.95 | Benign | 0.50 | Tolerated | 0.2677 | 0.5408 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.3080A>G | N1027S 2D  AIThe SynGAP1 missense variant N1027S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the collective predictions strongly suggest that the variant is most likely benign, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.831250 | Disordered | 0.994357 | Binding | 0.347 | 0.745 | 0.500 | -2.443 | Likely Benign | 0.093 | Likely Benign | Likely Benign | 0.088 | Likely Benign | -0.35 | Neutral | 0.068 | Benign | 0.039 | Benign | 2.77 | Benign | 0.50 | Tolerated | 0.3540 | 0.6874 | 1 | 1 | 2.7 | -27.03 | |||||||||||||||||||||||||||||||||||||||
| c.326G>T | S109I 2D  AIThe SynGAP1 missense variant S109I is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments are inconclusive: AlphaMissense‑Optimized remains uncertain; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is a 2‑vs‑2 tie, and Foldetta data are unavailable. Overall, the majority of evidence (five benign vs. three pathogenic predictions) points toward a benign impact. This conclusion does not contradict ClinVar, as the variant has no ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.622677 | Disordered | 0.669335 | Binding | 0.328 | 0.864 | 0.750 | -5.195 | Likely Benign | 0.927 | Likely Pathogenic | Ambiguous | 0.200 | Likely Benign | -2.56 | Deleterious | 0.267 | Benign | 0.039 | Benign | 3.47 | Benign | 0.00 | Affected | 0.0910 | 0.4930 | -1 | -2 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||||||||||||
| c.338G>C | G113A 2D  AIThe SynGAP1 missense variant G113A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign. Foldetta results are not available for this variant. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status, as none is assigned. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.784345 | Disordered | 0.639486 | Binding | 0.350 | 0.870 | 0.750 | -3.552 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.045 | Likely Benign | -0.61 | Neutral | 0.131 | Benign | 0.039 | Benign | 4.20 | Benign | 0.22 | Tolerated | 0.4059 | 0.4684 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.367G>T | A123S 2D  AIThe SynGAP1 missense variant A123S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions strongly supports a benign classification, and this conclusion does not contradict the ClinVar status, which currently contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.521092 | Disordered | 0.689505 | Binding | 0.324 | 0.886 | 0.750 | -3.309 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.081 | Likely Benign | -0.03 | Neutral | 0.131 | Benign | 0.039 | Benign | 4.21 | Benign | 0.28 | Tolerated | 0.2979 | 0.6011 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.3928A>G | T1310A 2D  AIThe SynGAP1 missense variant T1310A is not reported in ClinVar and is absent from gnomAD. All evaluated in‑silico predictors classify it as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. The high‑accuracy consensus methods likewise support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.812494 | Disordered | 0.959076 | Binding | 0.398 | 0.904 | 0.750 | -2.883 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.084 | Likely Benign | -0.21 | Neutral | 0.041 | Benign | 0.039 | Benign | 2.83 | Benign | 0.13 | Tolerated | 0.3238 | 0.3177 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||||||||||||
| c.457A>T | T153S 2D  AIThe SynGAP1 missense variant T153S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.501700 | Disordered | 0.502105 | Binding | 0.297 | 0.818 | 0.625 | -1.890 | Likely Benign | 0.118 | Likely Benign | Likely Benign | 0.118 | Likely Benign | -0.50 | Neutral | 0.235 | Benign | 0.039 | Benign | 4.19 | Benign | 0.14 | Tolerated | 0.3756 | 0.2886 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.458C>G | T153S 2D  AIThe SynGAP1 missense variant T153S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.501700 | Disordered | 0.502105 | Binding | 0.297 | 0.818 | 0.625 | -1.890 | Likely Benign | 0.118 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -0.50 | Neutral | 0.235 | Benign | 0.039 | Benign | 4.19 | Benign | 0.14 | Tolerated | 0.3756 | 0.2886 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.521T>G | M174R 2D  AIThe SynGAP1 missense variant M174R is not reported in ClinVar and is absent from gnomAD. Computational assessment shows a split in predictions: benign calls come from REVEL, polyPhen‑2 (HumDiv and HumVar), and FATHMM, whereas pathogenic calls arise from PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Pathogenic. High‑accuracy tools further support a deleterious effect: AlphaMissense‑Optimized predicts pathogenicity, and the SGM‑Consensus (majority vote) also indicates Likely Pathogenic. Foldetta results are unavailable, so no additional stability evidence is provided. Overall, the preponderance of evidence points to a pathogenic effect; this conclusion is not contradicted by any ClinVar annotation, as none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.661982 | Disordered | 0.485854 | Uncertain | 0.373 | 0.620 | 0.375 | -9.114 | Likely Pathogenic | 0.970 | Likely Pathogenic | Likely Pathogenic | 0.308 | Likely Benign | -3.15 | Deleterious | 0.139 | Benign | 0.039 | Benign | 4.05 | Benign | 0.01 | Affected | 0.1545 | 0.1037 | 0 | -1 | -6.4 | 24.99 | |||||||||||||||||||||||||||||||||||||||
| c.523C>A | Q175K 2D  AIThe SynGAP1 missense variant Q175K is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM. Only AlphaMissense‑Default predicts a pathogenic outcome, while AlphaMissense‑Optimized is uncertain. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support this: AlphaMissense‑Optimized remains uncertain, SGM‑Consensus is benign, and Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign effect for Q175K, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.653063 | Disordered | 0.474689 | Uncertain | 0.367 | 0.618 | 0.375 | -5.908 | Likely Benign | 0.826 | Likely Pathogenic | Ambiguous | 0.202 | Likely Benign | -0.89 | Neutral | 0.118 | Benign | 0.039 | Benign | 4.18 | Benign | 0.37 | Tolerated | 0.1655 | 0.3723 | 1 | 1 | -0.4 | 0.04 | |||||||||||||||||||||||||||||||||||||||
| c.523C>G | Q175E 2D  AIThe SynGAP1 missense variant Q175E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Consensus from multiple in‑silico predictors shows a benign bias: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized all classify the change as benign, whereas ESM1b predicts it to be pathogenic. AlphaMissense‑Default remains uncertain. High‑accuracy assessment further supports a benign outcome: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) resolves to benign, and Foldetta stability analysis is unavailable. Consequently, the overall evidence points to a benign effect for Q175E. This conclusion is consistent with the absence of a ClinVar assertion, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.653063 | Disordered | 0.474689 | Uncertain | 0.367 | 0.618 | 0.375 | -11.035 | Likely Pathogenic | 0.502 | Ambiguous | Likely Benign | 0.149 | Likely Benign | -0.68 | Neutral | 0.118 | Benign | 0.039 | Benign | 4.17 | Benign | 0.40 | Tolerated | 0.1351 | 0.2028 | 2 | 2 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||||||||||||
| c.524A>T | Q175L 2D  AIThe SynGAP1 missense variant Q175L is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are ESM1b and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 vs 2), and Foldetta results are unavailable. Overall, seven tools favor benign while two favor pathogenic, with no ClinVar evidence to contradict this. Thus, the variant is most likely benign based on the available predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.653063 | Disordered | 0.474689 | Uncertain | 0.367 | 0.618 | 0.375 | -8.699 | Likely Pathogenic | 0.579 | Likely Pathogenic | Likely Benign | 0.188 | Likely Benign | -2.46 | Neutral | 0.118 | Benign | 0.039 | Benign | 4.10 | Benign | 0.14 | Tolerated | 0.0647 | 0.5109 | -2 | -2 | 7.3 | -14.97 | ||||||||||||||||||||||||||||||||||||||||
| c.526A>G | S176G 2D  AIThe SynGAP1 missense variant S176G is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33435168‑A‑G). Consensus among most in silico predictors is benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized all report a benign effect. No tool predicts pathogenicity. Two predictors are inconclusive: ESM1b and AlphaMissense‑Default, which are grouped under uncertain. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, while the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) remains uncertain, and Foldetta stability analysis is unavailable. Overall, the computational evidence overwhelmingly favors a benign impact, which does not contradict the ClinVar uncertain classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.562014 | Disordered | 0.466016 | Uncertain | 0.380 | 0.597 | 0.375 | Uncertain | 1 | 6-33435168-A-G | 1 | 6.20e-7 | -7.541 | In-Between | 0.360 | Ambiguous | Likely Benign | 0.066 | Likely Benign | -1.08 | Neutral | 0.131 | Benign | 0.039 | Benign | 4.08 | Benign | 0.22 | Tolerated | 3.54 | 6 | 0.2361 | 0.3414 | 0 | 1 | 0.4 | -30.03 | |||||||||||||||||||||||||||||||||
| c.542A>T | H181L 2D  AIThe SynGAP1 H181L variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and ESM1b; AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) leans pathogenic (2 pathogenic vs 1 benign). Foldetta, which would provide a protein‑folding stability estimate, has no available result for this variant. Overall, the majority of standard predictors classify the variant as benign, and there is no ClinVar entry to contradict this assessment. Thus, the variant is most likely benign based on current predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.505461 | Disordered | 0.439530 | Uncertain | 0.294 | 0.616 | 0.500 | -11.014 | Likely Pathogenic | 0.561 | Ambiguous | Likely Benign | 0.212 | Likely Benign | -3.51 | Deleterious | 0.267 | Benign | 0.039 | Benign | 4.17 | Benign | 0.04 | Affected | 0.0734 | 0.4395 | -2 | -3 | 7.0 | -23.98 | ||||||||||||||||||||||||||||||||||||||||
| c.727A>C | I243L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 I243L missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from AlphaMissense‑Default and ESM1b, which are treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields benign; and Foldetta also predicts benign. Overall, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.363090 | Structured | 0.344471 | Uncertain | 0.842 | 0.347 | 0.000 | -7.890 | In-Between | 0.524 | Ambiguous | Likely Benign | -0.03 | Likely Benign | 0.2 | -0.35 | Likely Benign | -0.19 | Likely Benign | 0.32 | Likely Benign | 0.380 | Likely Benign | -0.33 | Neutral | 0.048 | Benign | 0.039 | Benign | 5.74 | Benign | 0.19 | Tolerated | 0.0626 | 0.2891 | 2 | 2 | -0.7 | 0.00 | ||||||||||||||||||||||||||||||
| c.739C>G | Q247E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant Q247E is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), and FATHMM. Tools that predict a pathogenic effect are premPS, SIFT, and ESM1b. AlphaMissense‑Default is uncertain, while AlphaMissense‑Optimized predicts benign. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a benign consensus (2 benign vs. 1 pathogenic, with the uncertain result treated as unavailable). High‑accuracy assessments confirm benignity: AlphaMissense‑Optimized is benign, Foldetta (combining FoldX‑MD and Rosetta outputs) is benign, and the SGM Consensus is benign. Overall, the collective evidence indicates that the variant is most likely benign, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.490133 | Structured | 0.283012 | Uncertain | 0.822 | 0.339 | 0.250 | -13.564 | Likely Pathogenic | 0.373 | Ambiguous | Likely Benign | 0.42 | Likely Benign | 0.1 | -0.26 | Likely Benign | 0.08 | Likely Benign | 1.04 | Destabilizing | 0.474 | Likely Benign | -1.46 | Neutral | 0.128 | Benign | 0.039 | Benign | 5.80 | Benign | 0.03 | Affected | 0.1055 | 0.1476 | 2 | 2 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||
| c.745G>C | A249P 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A249P is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. All other evaluated algorithms—SGM‑Consensus, REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized—predict a pathogenic impact. The SGM‑Consensus, which is a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is therefore pathogenic (3 pathogenic vs. 1 benign). High‑accuracy assessments are consistent: AlphaMissense‑Optimized indicates pathogenicity; the SGM‑Consensus (majority vote) is pathogenic; and Foldetta, integrating FoldX‑MD and Rosetta stability outputs, also predicts pathogenic. **Based on the collective predictions, the variant is most likely pathogenic, and this conclusion is not contradicted by any ClinVar status.** Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.505461 | Disordered | 0.255452 | Uncertain | 0.810 | 0.336 | 0.125 | -10.727 | Likely Pathogenic | 1.000 | Likely Pathogenic | Likely Pathogenic | 2.76 | Destabilizing | 0.4 | 8.40 | Destabilizing | 5.58 | Destabilizing | 1.04 | Destabilizing | 0.756 | Likely Pathogenic | -3.32 | Deleterious | 0.176 | Benign | 0.039 | Benign | 5.57 | Benign | 0.03 | Affected | 0.1429 | 0.3537 | 1 | -1 | -3.4 | 26.04 | |||||||||||||||||||||||||||||
| c.85A>C | M29L 2D  AIThe SynGAP1 missense variant M29L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.438540 | Uncertain | 0.341 | 0.883 | 0.250 | -1.633 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.200 | Likely Benign | -0.49 | Neutral | 0.006 | Benign | 0.039 | Benign | 4.27 | Benign | 0.00 | Affected | 0.1984 | 0.5336 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.85A>T | M29L 2D  AIThe SynGAP1 missense variant M29L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect; there is no conflict with ClinVar status because the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.541878 | Disordered | 0.438540 | Uncertain | 0.341 | 0.883 | 0.250 | -1.633 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.200 | Likely Benign | -0.49 | Neutral | 0.006 | Benign | 0.039 | Benign | 4.27 | Benign | 0.00 | Affected | 0.1984 | 0.5336 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.1016A>G | K339R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant K339R is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is benign. No predictions or stability results are missing or inconclusive. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.447574 | Structured | 0.384153 | Uncertain | 0.535 | 0.465 | 0.250 | -5.773 | Likely Benign | 0.120 | Likely Benign | Likely Benign | -0.15 | Likely Benign | 0.1 | 0.46 | Likely Benign | 0.16 | Likely Benign | 0.08 | Likely Benign | 0.134 | Likely Benign | -2.05 | Neutral | 0.046 | Benign | 0.040 | Benign | 1.94 | Pathogenic | 0.48 | Tolerated | 0.4197 | 0.0976 | 3 | 2 | -0.6 | 28.01 | |||||||||||||||||||||||||||||
| c.2219G>T | R740L 2D  AIThe SynGAP1 missense variant R740L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the preponderance of evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.771762 | Disordered | 0.475392 | Uncertain | 0.269 | 0.849 | 0.875 | -4.958 | Likely Benign | 0.126 | Likely Benign | Likely Benign | 0.043 | Likely Benign | -2.30 | Neutral | 0.064 | Benign | 0.040 | Benign | 2.57 | Benign | 0.03 | Affected | 0.2341 | 0.4243 | -3 | -2 | 8.3 | -43.03 | |||||||||||||||||||||||||||||||||||||||
| c.238A>C | K80Q 2D  AIThe SynGAP1 missense variant K80Q is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates Likely Benign. No Foldetta stability prediction is available for this variant. Overall, the majority of computational evidence points to a benign impact, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.477530 | Uncertain | 0.331 | 0.873 | 0.500 | -4.475 | Likely Benign | 0.662 | Likely Pathogenic | Likely Benign | 0.064 | Likely Benign | -0.81 | Neutral | 0.414 | Benign | 0.040 | Benign | 3.93 | Benign | 0.00 | Affected | 0.4373 | 0.1132 | 1 | 1 | 0.4 | -0.04 | |||||||||||||||||||||||||||||||||||||||
| c.3325C>A | L1109I 2D  AIThe SynGAP1 missense variant L1109I is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign, and this conclusion is consistent with the lack of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.856457 | Disordered | 0.948334 | Binding | 0.343 | 0.893 | 0.875 | -5.475 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.058 | Likely Benign | -0.40 | Neutral | 0.126 | Benign | 0.040 | Benign | 2.70 | Benign | 0.23 | Tolerated | 0.1087 | 0.4703 | 2 | 2 | 0.7 | 0.00 | |||||||||||||||||||||||||||||||||||||||
| c.44C>A | A15E 2D  AIThe SynGAP1 missense variant A15E is reported in gnomAD (ID 6‑33420308‑C‑A) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar status (none is provided). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.436924 | Structured | 0.466055 | Uncertain | 0.330 | 0.912 | 0.375 | 6-33420308-C-A | -3.423 | Likely Benign | 0.277 | Likely Benign | Likely Benign | 0.169 | Likely Benign | 0.46 | Neutral | 0.406 | Benign | 0.040 | Benign | 4.13 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1563 | 0.1908 | -1 | 0 | -5.3 | 58.04 | ||||||||||||||||||||||||||||||||||||
| c.914C>A | T305N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T305N is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as tolerated or benign, and the SGM‑Consensus score is “Likely Benign.” Only FATHMM predicts a pathogenic outcome. Stability‑based methods (FoldX, Rosetta, and the combined Foldetta) return uncertain or inconclusive results. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is also benign, and Foldetta remains uncertain. Overall, the majority of evidence supports a benign impact for T305N, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.359901 | Structured | 0.299706 | Uncertain | 0.872 | 0.274 | 0.125 | -3.261 | Likely Benign | 0.158 | Likely Benign | Likely Benign | 1.18 | Ambiguous | 0.2 | 1.65 | Ambiguous | 1.42 | Ambiguous | 0.09 | Likely Benign | 0.098 | Likely Benign | 0.71 | Neutral | 0.046 | Benign | 0.040 | Benign | 2.34 | Pathogenic | 0.67 | Tolerated | 0.1327 | 0.4352 | 0 | 0 | -2.8 | 13.00 | |||||||||||||||||||||||||||||
| c.1160G>A | G387D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G387D is reported in gnomAD (6‑33438065‑G‑A) but has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are FoldX, Rosetta, Foldetta, SIFT, ESM1b, FATHMM, and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Pathogenic. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) as pathogenic. With two of the three high‑accuracy methods indicating pathogenicity and a majority of general predictors leaning toward pathogenic, the variant is most likely pathogenic. This conclusion is not contradicted by ClinVar, which contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | C2 | 0.642678 | Disordered | 0.422910 | Uncertain | 0.293 | 0.861 | 0.750 | 6-33438065-G-A | 2 | 1.24e-6 | -8.625 | Likely Pathogenic | 0.612 | Likely Pathogenic | Likely Benign | 3.57 | Destabilizing | 2.3 | 3.22 | Destabilizing | 3.40 | Destabilizing | 0.39 | Likely Benign | 0.459 | Likely Benign | -0.37 | Neutral | 0.069 | Benign | 0.041 | Benign | 1.32 | Pathogenic | 0.02 | Affected | 4.32 | 3 | 0.2145 | 0.2035 | -1 | 1 | -3.1 | 58.04 | ||||||||||||||||||||||||
| c.3185G>A | G1062E 2D  AIThe SynGAP1 missense variant G1062E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and ESM1b. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.976962 | Disordered | 0.936972 | Binding | 0.368 | 0.917 | 0.875 | -8.185 | Likely Pathogenic | 0.272 | Likely Benign | Likely Benign | 0.383 | Likely Benign | -1.02 | Neutral | 0.126 | Benign | 0.041 | Benign | 4.10 | Benign | 0.01 | Affected | 0.1490 | 0.4069 | 0 | -2 | -3.1 | 72.06 | |||||||||||||||||||||||||||||||||||||||
| c.3277C>G | Q1093E 2D  AIThe SynGAP1 missense change Q1093E is not reported in ClinVar and is absent from gnomAD. In silico assessment shows unanimous benign predictions: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. No tool predicts pathogenicity. High‑accuracy consensus methods reinforce this view: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” The protein‑folding stability predictor Foldetta was not available for this variant. Overall, the evidence strongly supports a benign effect, and this conclusion is consistent with the lack of ClinVar annotation. Thus, the variant is most likely benign and does not contradict existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.983312 | Binding | 0.351 | 0.886 | 1.000 | -3.104 | Likely Benign | 0.265 | Likely Benign | Likely Benign | 0.047 | Likely Benign | -0.59 | Neutral | 0.112 | Benign | 0.041 | Benign | 2.78 | Benign | 0.07 | Tolerated | 0.1514 | 0.3717 | 2 | 2 | 0.0 | 0.98 | |||||||||||||||||||||||||||||||||||||||
| c.3371G>C | G1124A 2D  AIThe SynGAP1 missense variant G1124A is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized scores the variant as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign,” and the Foldetta stability analysis is unavailable. Taken together, the overwhelming majority of computational evidence classifies G1124A as benign, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.837511 | Disordered | 0.833401 | Binding | 0.341 | 0.931 | 0.875 | -6.608 | Likely Benign | 0.084 | Likely Benign | Likely Benign | 0.199 | Likely Benign | -0.39 | Neutral | 0.059 | Benign | 0.041 | Benign | 4.81 | Benign | 0.02 | Affected | 0.3494 | 0.5138 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3839T>A | M1280K 2D  AIThe SynGAP1 missense variant M1280K is not reported in ClinVar and has no entry in gnomAD, indicating it is not catalogued in these databases. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), AlphaMissense‑Default, AlphaMissense‑Optimized, and ESM1b. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) also indicates benign. The Foldetta protein‑folding stability analysis is unavailable for this variant. Overall, the preponderance of evidence from multiple prediction algorithms and consensus methods points to a benign impact for M1280K, and this conclusion does not contradict any ClinVar status, as none is assigned. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.882776 | Disordered | 0.822030 | Binding | 0.510 | 0.726 | 0.875 | -2.963 | Likely Benign | 0.223 | Likely Benign | Likely Benign | 0.213 | Likely Benign | -1.42 | Neutral | 0.126 | Benign | 0.041 | Benign | 2.37 | Pathogenic | 0.00 | Affected | 0.1096 | 0.0488 | 0 | -1 | -5.8 | -3.02 | |||||||||||||||||||||||||||||||||||||||
| c.3840G>A | M1280I 2D  AIThe SynGAP1 missense variant M1280I is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), AlphaMissense‑Default, AlphaMissense‑Optimized, and ESM1b. Tools that predict a pathogenic effect are PROVEAN, SIFT, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.882776 | Disordered | 0.822030 | Binding | 0.510 | 0.726 | 0.875 | -3.723 | Likely Benign | 0.251 | Likely Benign | Likely Benign | 0.166 | Likely Benign | -2.59 | Deleterious | 0.059 | Benign | 0.041 | Benign | 2.35 | Pathogenic | 0.00 | Affected | 0.0995 | 0.2468 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||||||||||||
| c.3840G>C | M1280I 2D  AIThe SynGAP1 missense variant M1280I is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), AlphaMissense‑Default, AlphaMissense‑Optimized, and ESM1b. Tools that predict a pathogenic effect are PROVEAN, SIFT, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.882776 | Disordered | 0.822030 | Binding | 0.510 | 0.726 | 0.875 | -3.723 | Likely Benign | 0.251 | Likely Benign | Likely Benign | 0.166 | Likely Benign | -2.59 | Deleterious | 0.059 | Benign | 0.041 | Benign | 2.35 | Pathogenic | 0.00 | Affected | 0.0995 | 0.2468 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||||||||||||
| c.3840G>T | M1280I 2D  AIThe SynGAP1 missense variant M1280I is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), AlphaMissense‑Default, AlphaMissense‑Optimized, and ESM1b. Tools that predict a pathogenic effect are PROVEAN, SIFT, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.882776 | Disordered | 0.822030 | Binding | 0.510 | 0.726 | 0.875 | -3.723 | Likely Benign | 0.251 | Likely Benign | Likely Benign | 0.166 | Likely Benign | -2.59 | Deleterious | 0.059 | Benign | 0.041 | Benign | 2.35 | Pathogenic | 0.00 | Affected | 0.0995 | 0.2468 | 2 | 1 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||||||||||||
| c.3859C>A | P1287T 2D  AIThe SynGAP1 missense variant P1287T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33447907‑C‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.813701 | Binding | 0.538 | 0.777 | 0.750 | Uncertain | 1 | 6-33447907-C-A | -3.940 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.044 | Likely Benign | -0.22 | Neutral | 0.126 | Benign | 0.041 | Benign | 2.78 | Benign | 0.04 | Affected | 3.77 | 5 | 0.1189 | 0.3995 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||
| c.3910C>T | P1304S 2D  AIThe SynGAP1 missense variant P1304S is reported in gnomAD (ID 6‑33451784‑C‑T) but has no ClinVar entry. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is provided). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.886417 | Binding | 0.475 | 0.866 | 0.875 | 6-33451784-C-T | 1 | 6.20e-7 | -4.103 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.039 | Likely Benign | 0.36 | Neutral | 0.059 | Benign | 0.041 | Benign | 3.03 | Benign | 0.58 | Tolerated | 3.77 | 5 | 0.2900 | 0.4442 | -1 | 1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||||
| c.1381G>A | A461T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 A461T missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are PROVEAN, polyPhen‑2 HumDiv, and ESM1b. Four tools (FoldX, Rosetta, Foldetta, premPS) returned uncertain results and are treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑vs‑2 split, and Foldetta is also inconclusive. Overall, the balance of evidence favors a benign classification, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.179055 | Structured | 0.292531 | Uncertain | 0.936 | 0.151 | 0.125 | -10.885 | Likely Pathogenic | 0.323 | Likely Benign | Likely Benign | 1.21 | Ambiguous | 0.2 | 0.51 | Ambiguous | 0.86 | Ambiguous | 0.65 | Ambiguous | 0.214 | Likely Benign | -3.27 | Deleterious | 0.508 | Possibly Damaging | 0.042 | Benign | 3.40 | Benign | 0.13 | Tolerated | 0.1083 | 0.5930 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||
| c.2186A>C | N729T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N729T is not reported in ClinVar (ClinVar status: not listed) but is present in gnomAD (gnomAD ID 6‑33441651‑A‑C). Consensus among the majority of in‑silico predictors is benign: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates “Likely Benign”; Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, yields an uncertain result. Overall, the evidence strongly supports a benign effect, and this conclusion does not contradict ClinVar status, which has no entry for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.750527 | Disordered | 0.426547 | Uncertain | 0.651 | 0.583 | 0.625 | 6-33441651-A-C | 1 | 6.20e-7 | -1.952 | Likely Benign | 0.103 | Likely Benign | Likely Benign | 0.52 | Ambiguous | 0.3 | 1.73 | Ambiguous | 1.13 | Ambiguous | -0.34 | Likely Benign | 0.052 | Likely Benign | -0.52 | Neutral | 0.123 | Benign | 0.042 | Benign | 3.33 | Benign | 1.00 | Tolerated | 3.59 | 7 | 0.1201 | 0.4805 | 0 | 0 | 2.8 | -13.00 | |||||||||||||||||||||||||
| c.2831G>C | G944A 2D  AIThe SynGAP1 missense variant G944A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.977651 | Disordered | 0.852408 | Binding | 0.360 | 0.923 | 0.750 | -6.397 | Likely Benign | 0.089 | Likely Benign | Likely Benign | 0.314 | Likely Benign | -0.61 | Neutral | 0.059 | Benign | 0.042 | Benign | 3.81 | Benign | 0.00 | Affected | 0.3352 | 0.4957 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2908G>A | E970K 2D  AIThe SynGAP1 missense variant E970K is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the collective evidence strongly supports a benign interpretation, and this conclusion does not contradict the ClinVar status, which currently contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.951925 | Disordered | 0.953422 | Binding | 0.342 | 0.902 | 0.750 | -3.344 | Likely Benign | 0.303 | Likely Benign | Likely Benign | 0.102 | Likely Benign | -0.24 | Neutral | 0.078 | Benign | 0.042 | Benign | 4.18 | Benign | 0.17 | Tolerated | 0.3372 | 0.7361 | 0 | 1 | -0.4 | -0.94 | |||||||||||||||||||||||||||||||||||||||
| c.2914C>A | P972T 2D  AIThe SynGAP1 missense variant P972T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.891961 | Disordered | 0.954150 | Binding | 0.472 | 0.904 | 0.625 | -5.144 | Likely Benign | 0.057 | Likely Benign | Likely Benign | 0.062 | Likely Benign | -1.01 | Neutral | 0.078 | Benign | 0.042 | Benign | 4.29 | Benign | 0.03 | Affected | 0.1802 | 0.5726 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.3173G>A | G1058D 2D  AIThe SynGAP1 missense variant G1058D is not reported in ClinVar (ClinVar status: none) but is present in gnomAD (ID 6‑33443725‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and ESM1b, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) resolves to benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict ClinVar, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.980739 | Disordered | 0.885724 | Binding | 0.407 | 0.929 | 0.875 | 6-33443725-G-A | 1 | 6.21e-7 | -10.344 | Likely Pathogenic | 0.391 | Ambiguous | Likely Benign | 0.177 | Likely Benign | -0.33 | Neutral | 0.077 | Benign | 0.042 | Benign | 5.20 | Benign | 0.01 | Affected | 3.77 | 5 | 0.1889 | 0.2435 | -1 | 1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||||||||
| c.3238G>A | A1080T 2D  AIThe SynGAP1 missense variant A1080T (ClinVar ID 1473274.0) is listed as “Uncertain” in ClinVar and is present in gnomAD (ID 6‑33443790‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the computational evidence strongly suggests the variant is most likely benign, which does not contradict the current ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.912647 | Disordered | 0.981457 | Binding | 0.303 | 0.900 | 0.750 | Conflicting | 2 | 6-33443790-G-A | 17 | 1.06e-5 | -3.928 | Likely Benign | 0.133 | Likely Benign | Likely Benign | 0.144 | Likely Benign | -0.19 | Neutral | 0.253 | Benign | 0.042 | Benign | 4.10 | Benign | 0.60 | Tolerated | 3.77 | 5 | 0.1564 | 0.7103 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||||
| c.3328A>C | S1110R 2D  AIThe SynGAP1 missense variant S1110R is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta results are unavailable. Overall, the balance of evidence (six benign versus three pathogenic predictions) indicates that the variant is most likely benign. This conclusion does not contradict ClinVar status, as the variant has no ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.912647 | Disordered | 0.934156 | Binding | 0.346 | 0.892 | 0.875 | -5.075 | Likely Benign | 0.773 | Likely Pathogenic | Likely Benign | 0.065 | Likely Benign | -2.46 | Neutral | 0.144 | Benign | 0.042 | Benign | 2.31 | Pathogenic | 0.01 | Affected | 0.1057 | 0.3571 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||||||||||
| c.3330C>A | S1110R 2D  AIThe SynGAP1 missense variant S1110R is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta results are unavailable. Overall, the majority of evidence (six benign vs. three pathogenic predictions) points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.912647 | Disordered | 0.934156 | Binding | 0.346 | 0.892 | 0.875 | -5.075 | Likely Benign | 0.773 | Likely Pathogenic | Likely Benign | 0.043 | Likely Benign | -2.46 | Neutral | 0.144 | Benign | 0.042 | Benign | 2.31 | Pathogenic | 0.01 | Affected | 0.1057 | 0.3571 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||||||||||
| c.3330C>G | S1110R 2D  AIThe SynGAP1 missense variant S1110R is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta results are unavailable. Overall, the majority of evidence (six benign vs. three pathogenic predictions) points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.912647 | Disordered | 0.934156 | Binding | 0.346 | 0.892 | 0.875 | -5.075 | Likely Benign | 0.773 | Likely Pathogenic | Likely Benign | 0.043 | Likely Benign | -2.46 | Neutral | 0.144 | Benign | 0.042 | Benign | 2.31 | Pathogenic | 0.01 | Affected | 0.1057 | 0.3571 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||||||||||
| c.347A>T | Y116F 2D  AIThe SynGAP1 missense variant Y116F is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only polyPhen‑2 HumDiv predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign likelihood. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.661982 | Disordered | 0.670235 | Binding | 0.381 | 0.878 | 0.625 | -2.911 | Likely Benign | 0.119 | Likely Benign | Likely Benign | 0.048 | Likely Benign | -0.71 | Neutral | 0.539 | Possibly Damaging | 0.042 | Benign | 4.21 | Benign | 0.33 | Tolerated | 0.2632 | 0.3069 | 7 | 3 | 4.1 | -16.00 | |||||||||||||||||||||||||||||||||||||||
| c.3914C>A | T1305K 2D  AIThe SynGAP1 missense variant T1305K is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores the variant as benign, and the SGM Consensus—derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—also indicates a likely benign effect. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact for T1305K, and this conclusion is not contradicted by any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.791621 | Disordered | 0.894658 | Binding | 0.390 | 0.873 | 0.875 | -4.289 | Likely Benign | 0.299 | Likely Benign | Likely Benign | 0.164 | Likely Benign | -1.33 | Neutral | 0.027 | Benign | 0.042 | Benign | 2.77 | Benign | 0.01 | Affected | 0.1290 | 0.3532 | 0 | -1 | -3.2 | 27.07 | |||||||||||||||||||||||||||||||||||||||
| c.436T>A | S146T 2D  AIThe SynGAP1 missense variant S146T is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments are inconclusive: AlphaMissense‑Optimized is uncertain; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is a tie (two pathogenic, two benign) and thus unavailable; Foldetta results are not provided. Overall, the majority of evidence (five benign vs. three pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as the variant is not yet catalogued there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.541878 | Disordered | 0.508612 | Binding | 0.349 | 0.837 | 0.625 | -9.795 | Likely Pathogenic | 0.786 | Likely Pathogenic | Ambiguous | 0.113 | Likely Benign | -2.09 | Neutral | 0.084 | Benign | 0.042 | Benign | 3.65 | Benign | 0.00 | Affected | 0.1267 | 0.5075 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.904T>A | S302T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S302T is not reported in ClinVar and is absent from gnomAD. All available in silico predictors classify the substitution as benign: REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). No tool predicts pathogenicity. High‑accuracy assessments concur: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields Likely Benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also indicates a benign effect. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.414856 | Structured | 0.263489 | Uncertain | 0.616 | 0.258 | 0.375 | -4.742 | Likely Benign | 0.072 | Likely Benign | Likely Benign | 0.20 | Likely Benign | 0.1 | 0.10 | Likely Benign | 0.15 | Likely Benign | -0.08 | Likely Benign | 0.043 | Likely Benign | -0.55 | Neutral | 0.037 | Benign | 0.042 | Benign | 4.16 | Benign | 0.15 | Tolerated | 0.1715 | 0.6886 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.2339C>G | S780C 2D  AIThe SynGAP1 missense variant S780C is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33442891‑C‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). No tool in the dataset predicts a pathogenic outcome; ESM1b is inconclusive and therefore treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign, while Foldetta results are not reported and thus unavailable. Based on the collective predictions, the variant is most likely benign, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.599170 | Disordered | 0.812415 | Binding | 0.283 | 0.883 | 0.500 | Uncertain | 4 | 6-33442891-C-G | 16 | 9.94e-6 | -7.603 | In-Between | 0.278 | Likely Benign | Likely Benign | 0.078 | Likely Benign | -1.41 | Neutral | 0.065 | Benign | 0.043 | Benign | 2.59 | Benign | 0.10 | Tolerated | 3.64 | 6 | 0.1063 | 0.6581 | -1 | 0 | 3.3 | 16.06 | ||||||||||||||||||||||||||||||||
| c.2525C>T | S842F 2D  AIThe SynGAP1 missense variant S842F is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show a split: benign calls from REVEL, polyPhen‑2 HumDiv and HumVar, whereas pathogenic calls come from PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default and AlphaMissense‑Optimized. The consensus predictor SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM and PROVEAN, reports the variant as Likely Pathogenic. High‑accuracy assessments further support a deleterious effect: AlphaMissense‑Optimized predicts pathogenicity, and the SGM‑Consensus result is consistent with this. Foldetta, a protein‑folding stability method that combines FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a pathogenic impact, and this conclusion does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.604312 | Disordered | 0.617281 | Binding | 0.274 | 0.861 | 0.250 | -14.590 | Likely Pathogenic | 0.988 | Likely Pathogenic | Likely Pathogenic | 0.188 | Likely Benign | -4.12 | Deleterious | 0.029 | Benign | 0.043 | Benign | 1.98 | Pathogenic | 0.00 | Affected | 0.0584 | 0.5692 | -3 | -2 | 3.6 | 60.10 | |||||||||||||||||||||||||||||||||||||||
| c.2879A>T | H960L 2D  AIThe SynGAP1 missense variant H960L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the only pathogenic prediction comes from ESM1b. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.987911 | Disordered | 0.983385 | Binding | 0.380 | 0.901 | 0.750 | -8.386 | Likely Pathogenic | 0.108 | Likely Benign | Likely Benign | 0.130 | Likely Benign | -1.30 | Neutral | 0.174 | Benign | 0.043 | Benign | 4.17 | Benign | 0.33 | Tolerated | 0.1465 | 0.5595 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.3841G>C | A1281P 2D  AIThe SynGAP1 missense variant A1281P is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign, and this conclusion is consistent with the lack of a ClinVar classification, so there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.821556 | Binding | 0.434 | 0.721 | 0.875 | -3.367 | Likely Benign | 0.071 | Likely Benign | Likely Benign | 0.067 | Likely Benign | -0.51 | Neutral | 0.149 | Benign | 0.043 | Benign | 2.64 | Benign | 0.14 | Tolerated | 0.2110 | 0.3522 | 1 | -1 | -3.4 | 26.04 | |||||||||||||||||||||||||||||||||||||||
| c.503A>C | H168P 2D  AIThe SynGAP1 missense variant H168P is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only SIFT predicts a pathogenic outcome, while ESM1b remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable for this variant. Taken together, the preponderance of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.502450 | Binding | 0.402 | 0.678 | 0.125 | -7.686 | In-Between | 0.124 | Likely Benign | Likely Benign | 0.279 | Likely Benign | -1.66 | Neutral | 0.072 | Benign | 0.043 | Benign | 4.19 | Benign | 0.02 | Affected | 0.2004 | 0.3960 | 0 | -2 | 1.6 | -40.02 | |||||||||||||||||||||||||||||||||||||||
| c.1346G>T | S449I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S449I is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, SIFT, AlphaMissense‑Default, AlphaMissense‑Optimized, and polyPhen2_HumVar. Those that predict a pathogenic effect are PROVEAN, polyPhen2_HumDiv, and ESM1b. Rosetta and Foldetta give uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs. 2 pathogenic). Foldetta also remains uncertain. Overall, the majority of evidence (7 benign vs. 3 pathogenic) points to a benign impact. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.254060 | Structured | 0.301437 | Uncertain | 0.958 | 0.251 | 0.000 | -9.549 | Likely Pathogenic | 0.310 | Likely Benign | Likely Benign | 0.04 | Likely Benign | 0.1 | -1.39 | Ambiguous | -0.68 | Ambiguous | 0.13 | Likely Benign | 0.105 | Likely Benign | -3.23 | Deleterious | 0.559 | Possibly Damaging | 0.044 | Benign | 3.40 | Benign | 0.14 | Tolerated | 0.0756 | 0.5006 | -1 | -2 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||
| c.1904A>G | N635S 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant N635S is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6-33440956-A-G). Functional prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and ESM1b. Predictions that are inconclusive or unavailable are FoldX, Rosetta, Foldetta, and premPS. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, and Foldetta is also inconclusive. Overall, the majority of available predictions lean toward a benign impact, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.039760 | Structured | 0.060246 | Uncertain | 0.900 | 0.252 | 0.000 | Conflicting | 4 | 6-33440956-A-G | 10 | 6.20e-6 | -9.002 | Likely Pathogenic | 0.101 | Likely Benign | Likely Benign | 0.80 | Ambiguous | 0.1 | 0.67 | Ambiguous | 0.74 | Ambiguous | 0.95 | Ambiguous | 0.104 | Likely Benign | -4.45 | Deleterious | 0.261 | Benign | 0.044 | Benign | 3.06 | Benign | 0.05 | Affected | 3.37 | 34 | 0.2816 | 0.4279 | 1 | 1 | 2.7 | -27.03 | 196.0 | 30.9 | 0.1 | 0.0 | -0.3 | 0.2 | X | Uncertain | In the WT simulations, the carboxamide side chain of Asn635, located on the outer surface of an α helix (res. Glu617-Asn635), forms hydrogen bonds with Gln631 on the same α helix and with the hydroxyl side chain of Ser590 on an opposing α helix (res. Glu582-Met603).In the variant simulations, the side chain of Ser635 is shorter than asparagine and thus prefers to hydrogen bond with the carbonyl group of Gln631 on the same helix and, to a lesser extent, with Ser590 compared to Asn635 in the WT. Ser635 forms hydrogen bonds with the backbone atoms of the same helix, which may destabilize the helix, although this is not clearly evident in the simulations. The weakening of the hydrogen bond between Ser635 and Ser590 in the variant may also weaken the tertiary structure assembly between the helices.Additionally, Asn635 is at the GTPase interface. However, the implication of the residue swap on the complex formation with the GTPase cannot be investigated using solvent-only simulations. | ||||||||||||||
| c.2961C>A | D987E 2D  AIThe SynGAP1 missense variant D987E is reported in gnomAD (6‑33443513‑C‑A) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus also indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of predictions points to a benign effect, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.919118 | Binding | 0.299 | 0.903 | 0.750 | 6-33443513-C-A | 1 | 6.20e-7 | -4.406 | Likely Benign | 0.425 | Ambiguous | Likely Benign | 0.124 | Likely Benign | -1.36 | Neutral | 0.049 | Benign | 0.044 | Benign | 2.64 | Benign | 0.04 | Affected | 4.32 | 2 | 0.1586 | 0.7239 | 2 | 3 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||||
| c.2961C>G | D987E 2D  AIThe SynGAP1 missense variant D987E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, SGM‑Consensus indicates Likely Benign, and Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign effect for D987E, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.919118 | Binding | 0.299 | 0.903 | 0.750 | -4.406 | Likely Benign | 0.425 | Ambiguous | Likely Benign | 0.124 | Likely Benign | -1.36 | Neutral | 0.049 | Benign | 0.044 | Benign | 2.64 | Benign | 0.04 | Affected | 4.32 | 2 | 0.1586 | 0.7239 | 2 | 3 | 0.0 | 14.03 | |||||||||||||||||||||||||||||||||||||
| c.3050T>A | F1017Y 2D  AIThe SynGAP1 missense variant F1017Y is reported in gnomAD (6‑33443602‑T‑A) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), AlphaMissense‑Default, AlphaMissense‑Optimized, ESM1b, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic outcome are SIFT and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.889439 | Disordered | 0.954171 | Binding | 0.322 | 0.801 | 0.625 | 6-33443602-T-A | -3.583 | Likely Benign | 0.186 | Likely Benign | Likely Benign | 0.042 | Likely Benign | -0.75 | Neutral | 0.012 | Benign | 0.044 | Benign | 2.47 | Pathogenic | 0.05 | Affected | 3.77 | 5 | 0.1432 | 0.1801 | 3 | 7 | -4.1 | 16.00 | ||||||||||||||||||||||||||||||||||||
| c.1076C>A | T359K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 T359K missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are ESM1b and FATHMM. The remaining tools—FoldX, Rosetta, premPS, and AlphaMissense‑Default—return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) as pathogenic, and Foldetta as benign. Overall, the majority of predictions (seven benign vs. two pathogenic) support a benign classification, and this conclusion does not contradict the lack of ClinVar annotation. Thus, the variant is most likely benign based on current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.281712 | Structured | 0.414952 | Uncertain | 0.939 | 0.480 | 0.250 | -10.822 | Likely Pathogenic | 0.517 | Ambiguous | Likely Benign | -0.65 | Ambiguous | 0.1 | 0.66 | Ambiguous | 0.01 | Likely Benign | 0.75 | Ambiguous | 0.163 | Likely Benign | -2.23 | Neutral | 0.255 | Benign | 0.045 | Benign | 1.79 | Pathogenic | 0.24 | Tolerated | 0.1618 | 0.3483 | 0 | -1 | -3.2 | 27.07 | ||||||||||||||||||||||||||||||
| c.1169G>A | G390E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G390E is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools that classify the variant as benign include premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Optimized. Those that predict pathogenicity are REVEL, FoldX, Rosetta, Foldetta, SIFT, FATHMM, and AlphaMissense‑Default. A high‑accuracy assessment shows AlphaMissense‑Optimized as benign, the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) as pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) as pathogenic. Overall, the majority of predictions support a pathogenic effect, and this aligns with the ClinVar designation of uncertain significance rather than contradicting it. Thus, the variant is most likely pathogenic based on current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.626927 | Disordered | 0.413274 | Uncertain | 0.304 | 0.763 | 0.875 | Uncertain | 1 | -7.913 | In-Between | 0.646 | Likely Pathogenic | Likely Benign | 2.61 | Destabilizing | 0.9 | 4.28 | Destabilizing | 3.45 | Destabilizing | 0.47 | Likely Benign | 0.575 | Likely Pathogenic | -0.87 | Neutral | 0.276 | Benign | 0.045 | Benign | 1.32 | Pathogenic | 0.05 | Affected | 4.32 | 8 | 0.1595 | 0.4309 | 0 | -2 | -3.1 | 72.06 | 241.5 | -108.4 | 0.6 | 0.5 | -0.1 | 0.1 | Uncertain | Gly390 is located in the Gly-rich Ω loop (res. Pro364-Pro398) between two anti-parallel β sheet strands (res. Thr359-Pro364 and res. Ala399-Ile411). The Ω loop is assumed to directly interact with the membrane, and it is observed to move arbitrarily throughout the WT solvent simulations. This loop potentially plays a crucial role in the SynGAP-membrane complex association, stability, and dynamics. However, this aspect cannot be fully addressed through solvent simulations alone.Ω loops are known to play significant roles in protein functions that require flexibility, and so they are rich in glycine residues, prolines, and to a lesser extent, small hydrophilic residues to ensure maximum flexibility. Thus, the variant’s Glu390 may not be as well tolerated in the Ω loop. Additionally, the carboxylate group of Glu390 occasionally forms H-bonds with other loop residues in the variant simulations. The interaction between the acidic carboxylate side chain and the acidic membrane lipids may further influence the SynGAP-membrane complex. However, since the effects on the Gly-rich Ω loop dynamics can only be well studied through the SynGAP-membrane complex, no definite conclusions can be drawn. | ||||||||||||||||||
| c.1175A>C | K392T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant K392T is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include Rosetta, Foldetta, premPS, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are REVEL, PROVEAN, and SIFT. Two tools—FoldX and AlphaMissense‑Default—return uncertain results and are treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) resolves to benign; and Foldetta, a protein‑folding stability method, also predicts benign. No ClinVar entry exists to contradict these predictions. Based on the collective evidence, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.541878 | Disordered | 0.405672 | Uncertain | 0.319 | 0.702 | 0.750 | -3.224 | Likely Benign | 0.481 | Ambiguous | Likely Benign | 0.52 | Ambiguous | 0.1 | -0.29 | Likely Benign | 0.12 | Likely Benign | 0.05 | Likely Benign | 0.512 | Likely Pathogenic | -3.14 | Deleterious | 0.276 | Benign | 0.045 | Benign | 4.60 | Benign | 0.02 | Affected | 0.2807 | 0.4053 | 0 | -1 | 3.2 | -27.07 | ||||||||||||||||||||||||||||||
| c.1318A>G | N440D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N440D is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome, while FoldX, Foldetta, and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.191378 | Structured | 0.267204 | Uncertain | 0.929 | 0.245 | 0.000 | -9.335 | Likely Pathogenic | 0.407 | Ambiguous | Likely Benign | -0.62 | Ambiguous | 0.0 | -0.41 | Likely Benign | -0.52 | Ambiguous | 0.47 | Likely Benign | 0.074 | Likely Benign | -1.71 | Neutral | 0.229 | Benign | 0.045 | Benign | 3.43 | Benign | 0.43 | Tolerated | 0.1544 | 0.2025 | 2 | 1 | 0.0 | 0.98 | ||||||||||||||||||||||||||||||
| c.2020A>G | T674A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T674A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly favor a benign effect: AlphaMissense‑Default, AlphaMissense‑Optimized, ESM1b, FATHMM, PROVEAN, SIFT, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, REVEL, FoldX, premPS, Foldetta, and the SGM‑Consensus score all indicate benign or likely benign. No tool predicts pathogenicity; the only inconclusive result comes from Rosetta, which is listed as uncertain. High‑accuracy methods corroborate the benign assessment: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. Based on the aggregate predictions, the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.129801 | Structured | 0.109297 | Uncertain | 0.521 | 0.349 | 0.000 | -5.915 | Likely Benign | 0.099 | Likely Benign | Likely Benign | -0.13 | Likely Benign | 0.0 | 0.89 | Ambiguous | 0.38 | Likely Benign | 0.29 | Likely Benign | 0.052 | Likely Benign | -1.48 | Neutral | 0.398 | Benign | 0.045 | Benign | 3.45 | Benign | 0.28 | Tolerated | 0.4042 | 0.4845 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||
| c.2167A>G | T723A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T723A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that assess sequence conservation, structural impact, or functional effect all converge on a benign outcome: SIFT, PolyPhen‑2 (HumDiv and HumVar), PROVEAN, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, REVEL, and the protein‑stability predictors FoldX, Rosetta, premPS, and Foldetta all indicate a benign effect. No tool predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, also predicts benign. Based on the uniform predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.476583 | Structured | 0.458243 | Uncertain | 0.945 | 0.447 | 0.375 | -4.572 | Likely Benign | 0.064 | Likely Benign | Likely Benign | -0.47 | Likely Benign | 0.0 | 0.16 | Likely Benign | -0.16 | Likely Benign | 0.25 | Likely Benign | 0.053 | Likely Benign | -0.91 | Neutral | 0.161 | Benign | 0.045 | Benign | 3.51 | Benign | 0.06 | Tolerated | 0.3902 | 0.3639 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||
| c.2884C>A | H962N 2D  AIThe SynGAP1 missense variant H962N is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only ESM1b predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, and Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.991070 | Disordered | 0.984483 | Binding | 0.369 | 0.886 | 0.750 | -8.481 | Likely Pathogenic | 0.098 | Likely Benign | Likely Benign | 0.058 | Likely Benign | -0.61 | Neutral | 0.174 | Benign | 0.045 | Benign | 4.18 | Benign | 0.24 | Tolerated | 0.1957 | 0.3216 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.2886C>A | H962Q 2D  AIThe SynGAP1 missense variant H962Q is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.991070 | Disordered | 0.984483 | Binding | 0.369 | 0.886 | 0.750 | -8.161 | Likely Pathogenic | 0.130 | Likely Benign | Likely Benign | 0.114 | Likely Benign | -1.04 | Neutral | 0.325 | Benign | 0.045 | Benign | 4.19 | Benign | 0.06 | Tolerated | 0.1943 | 0.3844 | 3 | 0 | -0.3 | -9.01 | |||||||||||||||||||||||||||||||||||||||
| c.2886C>G | H962Q 2D  AIThe SynGAP1 missense variant H962Q is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect. There is no ClinVar entry to contradict this conclusion. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.991070 | Disordered | 0.984483 | Binding | 0.369 | 0.886 | 0.750 | -8.161 | Likely Pathogenic | 0.130 | Likely Benign | Likely Benign | 0.114 | Likely Benign | -1.04 | Neutral | 0.325 | Benign | 0.045 | Benign | 4.19 | Benign | 0.06 | Tolerated | 0.1943 | 0.3844 | 3 | 0 | -0.3 | -9.01 | |||||||||||||||||||||||||||||||||||||||
| c.3107A>T | Q1036L 2D  AIThe SynGAP1 missense variant Q1036L is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and SIFT, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also favors a benign outcome, with two benign votes versus one pathogenic and one uncertain. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.948786 | Disordered | 0.987955 | Binding | 0.275 | 0.765 | 0.625 | -4.389 | Likely Benign | 0.435 | Ambiguous | Likely Benign | 0.092 | Likely Benign | -2.92 | Deleterious | 0.152 | Benign | 0.045 | Benign | 2.52 | Benign | 0.01 | Affected | 0.1069 | 0.5996 | -2 | -2 | 7.3 | -14.97 | ||||||||||||||||||||||||||||||||||||||||
| c.376T>A | F126I 2D  AIThe SynGAP1 missense variant F126I is not reported in ClinVar and has no entry in gnomAD. Functional prediction tools largely agree that the change is benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized all classify it as benign. Only two tools predict pathogenicity—SIFT and AlphaMissense‑Default. High‑accuracy consensus methods reinforce the benign assessment: the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and AlphaMissense‑Optimized itself predicts benign. The protein‑folding stability predictor Foldetta was not available for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.486429 | Structured | 0.712056 | Binding | 0.316 | 0.874 | 0.500 | -2.919 | Likely Benign | 0.735 | Likely Pathogenic | Likely Benign | 0.044 | Likely Benign | -2.20 | Neutral | 0.160 | Benign | 0.045 | Benign | 3.95 | Benign | 0.00 | Affected | 0.2569 | 0.2418 | 1 | 0 | 1.7 | -34.02 | |||||||||||||||||||||||||||||||||||||||
| c.376T>G | F126V 2D  AIThe SynGAP1 missense variant F126V is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact for F126V, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.486429 | Structured | 0.712056 | Binding | 0.316 | 0.874 | 0.500 | -1.584 | Likely Benign | 0.635 | Likely Pathogenic | Likely Benign | 0.086 | Likely Benign | -2.47 | Neutral | 0.160 | Benign | 0.045 | Benign | 3.98 | Benign | 0.00 | Affected | 0.2496 | 0.2646 | -1 | -1 | 1.4 | -48.04 | |||||||||||||||||||||||||||||||||||||||
| c.377T>C | F126S 2D  AIThe SynGAP1 missense variant F126S is not reported in ClinVar and has no entry in gnomAD. Functional prediction tools largely agree that the change is benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM all classify it as benign. In contrast, SIFT and AlphaMissense‑Default predict a pathogenic effect. AlphaMissense‑Optimized returns an uncertain result, and Foldetta (a protein‑folding stability method combining FoldX‑MD and Rosetta outputs) has no available prediction for this residue. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” Overall, the majority of high‑accuracy tools (REVEL, PROVEAN, polyPhen‑2, ESM1b, FATHMM, and SGM‑Consensus) support a benign interpretation, while only two tools (SIFT, AlphaMissense‑Default) suggest pathogenicity. Given the preponderance of benign predictions and the absence of ClinVar evidence, the variant is most likely benign and does not contradict any existing ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.486429 | Structured | 0.712056 | Binding | 0.316 | 0.874 | 0.500 | -0.234 | Likely Benign | 0.844 | Likely Pathogenic | Ambiguous | 0.086 | Likely Benign | -2.46 | Neutral | 0.160 | Benign | 0.045 | Benign | 3.96 | Benign | 0.00 | Affected | 0.4833 | 0.0000 | -3 | -2 | -3.6 | -60.10 | |||||||||||||||||||||||||||||||||||||||
| c.3940C>A | P1314T 2D  AIThe SynGAP1 missense variant P1314T is present in gnomAD (ID 6‑33451814‑C‑A) but has no ClinVar entry. Across a broad panel of in silico predictors, every tool reports a benign effect. Benign predictions come from REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; no tool in the set predicts pathogenicity, so the pathogenic group is empty. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized returns a benign prediction, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—labels the variant as Likely Benign. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status remains unavailable. Overall, the computational evidence overwhelmingly supports a benign impact, and this is consistent with the absence of a ClinVar pathogenic classification. Thus, the variant is most likely benign, and this is consistent with the lack of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.882776 | Disordered | 0.971592 | Binding | 0.467 | 0.903 | 0.750 | 6-33451814-C-A | -4.083 | Likely Benign | 0.096 | Likely Benign | Likely Benign | 0.066 | Likely Benign | 1.26 | Neutral | 0.061 | Benign | 0.045 | Benign | 4.32 | Benign | 0.97 | Tolerated | 3.77 | 5 | 0.1791 | 0.5626 | -1 | 0 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||||||||
| c.3947A>C | N1316T 2D  AIThe SynGAP1 missense variant N1316T is reported in gnomAD (ID 6‑33451821‑A‑C) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. In contrast, PROVEAN and SIFT predict a pathogenic impact. When predictions are grouped by consensus, the benign‑predicating tools outnumber the pathogenic ones. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta, a protein‑folding stability method, has no available result for this variant. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.899122 | Disordered | 0.971970 | Binding | 0.380 | 0.885 | 0.750 | 6-33451821-A-C | -3.080 | Likely Benign | 0.315 | Likely Benign | Likely Benign | 0.107 | Likely Benign | -3.18 | Deleterious | 0.127 | Benign | 0.045 | Benign | 3.96 | Benign | 0.00 | Affected | 3.77 | 5 | 0.1450 | 0.6621 | 0 | 0 | 2.8 | -13.00 | ||||||||||||||||||||||||||||||||||||
| c.3949G>A | G1317S 2D  AIThe SynGAP1 missense variant G1317S is listed in ClinVar with an uncertain significance and is present in the gnomAD database. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized returns a benign prediction, and the SGM‑Consensus (derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates benign. Foldetta, a protein‑folding stability method, has no available result for this variant. Overall, the computational evidence overwhelmingly points to a benign effect, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.908098 | Disordered | 0.971158 | Binding | 0.385 | 0.879 | 0.750 | Conflicting | 3 | 6-33451823-G-A | 1 | 6.26e-7 | -3.522 | Likely Benign | 0.145 | Likely Benign | Likely Benign | 0.092 | Likely Benign | -2.45 | Neutral | 0.127 | Benign | 0.045 | Benign | 4.08 | Benign | 0.00 | Affected | 3.77 | 5 | 0.2546 | 0.4935 | 1 | 0 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||
| c.4025A>G | D1342G 2D  AIThe SynGAP1 missense variant D1342G is reported in gnomAD (variant ID 6‑33451899‑A‑G) but has no ClinVar entry. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.921076 | Disordered | 0.981682 | Binding | 0.316 | 0.678 | 0.875 | 6-33451899-A-G | 1 | 7.29e-7 | -3.227 | Likely Benign | 0.129 | Likely Benign | Likely Benign | 0.021 | Likely Benign | -0.89 | Neutral | 0.225 | Benign | 0.045 | Benign | 4.05 | Benign | 0.09 | Tolerated | 4.32 | 4 | 0.3633 | 0.5601 | -1 | 1 | 3.1 | -58.04 | ||||||||||||||||||||||||||||||||||
| c.596A>G | N199S 2D  AIThe SynGAP1 missense variant N199S is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). All available in‑silico predictors classify the variant as benign: REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). No tool predicts pathogenicity. High‑accuracy assessments are consistent: AlphaMissense‑Optimized predicts benign; the SGM‑Consensus (majority vote) indicates likely benign; Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also predicts benign. Overall, the variant is most likely benign, and this prediction does not contradict the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.390993 | Structured | 0.431347 | Uncertain | 0.571 | 0.473 | 0.125 | -0.268 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.08 | Likely Benign | 0.4 | -0.08 | Likely Benign | 0.00 | Likely Benign | -0.44 | Likely Benign | 0.040 | Likely Benign | 0.09 | Neutral | 0.160 | Benign | 0.045 | Benign | 4.36 | Benign | 0.57 | Tolerated | 0.2601 | 0.6023 | 1 | 1 | 2.7 | -27.03 | |||||||||||||||||||||||||||||
| c.596A>T | N199I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N199I is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, polyPhen‑2 (HumDiv and HumVar), FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive because it yields an equal split of benign and pathogenic calls. Overall, the majority of evidence points to a benign impact. The variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.390993 | Structured | 0.431347 | Uncertain | 0.571 | 0.473 | 0.125 | -8.299 | Likely Pathogenic | 0.328 | Likely Benign | Likely Benign | 0.27 | Likely Benign | 0.1 | -0.11 | Likely Benign | 0.08 | Likely Benign | 0.20 | Likely Benign | 0.066 | Likely Benign | -3.27 | Deleterious | 0.316 | Benign | 0.045 | Benign | 4.16 | Benign | 0.01 | Affected | 0.0450 | 0.6009 | -2 | -3 | 8.0 | -0.94 | ||||||||||||||||||||||||||||||
| c.107A>C | H36P 2D  AIThe SynGAP1 missense variant H36P is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -2.285 | Likely Benign | 0.066 | Likely Benign | Likely Benign | 0.171 | Likely Benign | -1.23 | Neutral | 0.182 | Benign | 0.046 | Benign | 4.16 | Benign | 0.00 | Affected | 0.2097 | 0.4723 | 0 | -2 | 1.6 | -40.02 | |||||||||||||||||||||||||||||||||||||||
| c.108T>A | H36Q 2D  AIThe SynGAP1 missense variant H36Q is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -3.642 | Likely Benign | 0.135 | Likely Benign | Likely Benign | 0.044 | Likely Benign | -0.16 | Neutral | 0.182 | Benign | 0.046 | Benign | 4.34 | Benign | 0.00 | Affected | 0.2042 | 0.4572 | 3 | 0 | -0.3 | -9.01 | |||||||||||||||||||||||||||||||||||||||
| c.108T>G | H36Q 2D  AIThe SynGAP1 missense variant H36Q is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.433974 | Uncertain | 0.334 | 0.834 | 0.375 | -3.642 | Likely Benign | 0.135 | Likely Benign | Likely Benign | 0.044 | Likely Benign | -0.16 | Neutral | 0.182 | Benign | 0.046 | Benign | 4.34 | Benign | 0.00 | Affected | 0.2042 | 0.4572 | 3 | 0 | -0.3 | -9.01 | |||||||||||||||||||||||||||||||||||||||
| c.1289T>G | M430R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M430R is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are PROVEAN, polyPhen‑2 HumDiv, and ESM1b. The remaining tools—FoldX, Foldetta, premPS, and AlphaMissense‑Default—return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta as uncertain. Overall, the majority of consensus tools lean toward a benign classification, while a subset of high‑accuracy methods suggest pathogenicity, leaving the variant’s impact ambiguous. Based on the aggregate predictions, the variant is most likely benign, and this assessment does not contradict the absence of a ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.071867 | Structured | 0.385298 | Uncertain | 0.952 | 0.306 | 0.000 | -10.636 | Likely Pathogenic | 0.558 | Ambiguous | Likely Benign | 0.98 | Ambiguous | 0.1 | 0.47 | Likely Benign | 0.73 | Ambiguous | 0.83 | Ambiguous | 0.153 | Likely Benign | -2.87 | Deleterious | 0.620 | Possibly Damaging | 0.046 | Benign | 3.48 | Benign | 0.38 | Tolerated | 0.1775 | 0.1652 | 0 | -1 | -6.4 | 24.99 | ||||||||||||||||||||||||||||||
| c.1750A>C | I584L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I584L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are ESM1b and FATHMM, while premPS is uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive due to a 2‑vs‑2 split. Overall, the preponderance of evidence points to a benign impact. This conclusion does not contradict ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.059222 | Structured | 0.046673 | Uncertain | 0.846 | 0.244 | 0.000 | -8.266 | Likely Pathogenic | 0.285 | Likely Benign | Likely Benign | -0.18 | Likely Benign | 0.1 | -0.30 | Likely Benign | -0.24 | Likely Benign | 0.84 | Ambiguous | 0.420 | Likely Benign | -1.74 | Neutral | 0.008 | Benign | 0.046 | Benign | -1.23 | Pathogenic | 0.18 | Tolerated | 0.0927 | 0.2817 | 2 | 2 | -0.7 | 0.00 | ||||||||||||||||||||||||||||||
| c.2077C>T | H693Y 2D  AIThe SynGAP1 H693Y missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include FoldX, premPS, polyPhen‑2 HumVar, and FATHMM. Those that predict a pathogenic effect are REVEL, PROVEAN, polyPhen‑2 HumDiv, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Uncertain predictions come from Rosetta, Foldetta, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta as uncertain. Overall, the majority of evidence points to a pathogenic impact. This conclusion is consistent with the lack of ClinVar annotation; there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.073402 | Structured | 0.323991 | Uncertain | 0.964 | 0.260 | 0.000 | -7.963 | In-Between | 0.984 | Likely Pathogenic | Likely Pathogenic | -0.16 | Likely Benign | 1.7 | -1.41 | Ambiguous | -0.79 | Ambiguous | 0.10 | Likely Benign | 0.513 | Likely Pathogenic | -5.98 | Deleterious | 0.553 | Possibly Damaging | 0.046 | Benign | 3.13 | Benign | 0.02 | Affected | 0.0819 | 0.3419 | 0 | 2 | 1.9 | 26.03 | ||||||||||||||||||||||||||||||
| c.2353C>T | R785C 2D  AIThe SynGAP1 R785C missense variant is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6‑33442905‑C‑T). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are PROVEAN, SIFT, FATHMM, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—indicates a likely pathogenic outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence points toward a pathogenic impact, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | SH3-binding motif | 0.859585 | Disordered | 0.681730 | Binding | 0.325 | 0.896 | 0.625 | Uncertain | 1 | 6-33442905-C-T | 29 | 1.80e-5 | -5.887 | Likely Benign | 0.662 | Likely Pathogenic | Likely Benign | 0.126 | Likely Benign | -5.06 | Deleterious | 0.144 | Benign | 0.046 | Benign | 2.22 | Pathogenic | 0.00 | Affected | 3.64 | 6 | 0.3775 | 0.3530 | -4 | -3 | 7.0 | -53.05 | |||||||||||||||||||||||||||||||
| c.2530C>A | L844M 2D  AIThe SynGAP1 missense variant L844M is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Based on the collective predictions, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.611301 | Binding | 0.304 | 0.835 | 0.375 | 1.261 | Likely Benign | 0.213 | Likely Benign | Likely Benign | 0.027 | Likely Benign | 0.42 | Neutral | 0.052 | Benign | 0.046 | Benign | 2.72 | Benign | 0.38 | Tolerated | 0.0872 | 0.4122 | 4 | 2 | -1.9 | 18.03 | |||||||||||||||||||||||||||||||||||||||
| c.98A>G | Q33R 2D  AIThe SynGAP1 missense variant Q33R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The high‑accuracy consensus, SGM‑Consensus, classifies the variant as Likely Benign, and AlphaMissense‑Optimized also reports a benign prediction. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect. The predictions do not contradict ClinVar status, as ClinVar contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.436712 | Uncertain | 0.342 | 0.860 | 0.375 | 0.561 | Likely Benign | 0.171 | Likely Benign | Likely Benign | 0.035 | Likely Benign | -0.82 | Neutral | 0.084 | Benign | 0.046 | Benign | 4.20 | Benign | 0.00 | Affected | 0.1936 | 0.2773 | 1 | 1 | -1.0 | 28.06 | |||||||||||||||||||||||||||||||||||||||
| c.192A>G | I64M 2D  AIThe SynGAP1 missense variant I64M is listed in gnomAD (ID 6‑33425800‑A‑G) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic outcome are polyPhen‑2 HumDiv and SIFT, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.422041 | Structured | 0.475481 | Uncertain | 0.478 | 0.747 | 0.125 | 6-33425800-A-G | 2 | 1.24e-6 | -4.327 | Likely Benign | 0.523 | Ambiguous | Likely Benign | 0.047 | Likely Benign | -0.05 | Neutral | 0.637 | Possibly Damaging | 0.047 | Benign | 4.04 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0568 | 0.2310 | 1 | 2 | -2.6 | 18.03 | ||||||||||||||||||||||||||||||||||
| c.20C>A | S7Y 2D  AIThe SynGAP1 missense variant S7Y is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact. The predictions do not contradict any ClinVar annotation, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.590140 | Disordered | 0.548467 | Binding | 0.386 | 0.922 | 0.750 | -5.240 | Likely Benign | 0.230 | Likely Benign | Likely Benign | 0.193 | Likely Benign | -0.36 | Neutral | 0.561 | Possibly Damaging | 0.047 | Benign | 4.06 | Benign | 0.00 | Affected | 0.0708 | 0.5053 | -3 | -2 | -0.5 | 76.10 | |||||||||||||||||||||||||||||||||||||||
| c.2440G>A | A814T 2D  AIThe SynGAP1 missense variant A814T is not reported in ClinVar and is absent from gnomAD. Consensus from multiple in‑silico predictors indicates a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome, while Foldetta results are unavailable. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.411940 | Structured | 0.814830 | Binding | 0.368 | 0.902 | 0.250 | -3.600 | Likely Benign | 0.227 | Likely Benign | Likely Benign | 0.062 | Likely Benign | -1.24 | Neutral | 0.103 | Benign | 0.047 | Benign | 2.64 | Benign | 0.09 | Tolerated | 0.1400 | 0.6655 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||||||||||
| c.2674T>C | S892P 2D  AIThe SynGAP1 missense variant S892P is reported in gnomAD (ID 6‑33443226‑T‑C) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only SIFT predicts it to be pathogenic. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the consensus of the majority of prediction tools and the high‑accuracy methods points to a benign impact. This conclusion is not contradicted by ClinVar, which contains no classification for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.754692 | Disordered | 0.473390 | Uncertain | 0.319 | 0.926 | 0.875 | 6-33443226-T-C | 1 | 6.20e-7 | -3.247 | Likely Benign | 0.222 | Likely Benign | Likely Benign | 0.041 | Likely Benign | -0.87 | Neutral | 0.057 | Benign | 0.047 | Benign | 2.60 | Benign | 0.01 | Affected | 4.32 | 4 | 0.2230 | 0.5251 | -1 | 1 | -0.8 | 10.04 | ||||||||||||||||||||||||||||||||||
| c.2834A>C | H945P 2D  AIThe SynGAP1 missense variant H945P is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the preponderance of benign predictions and the lack of pathogenic evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.982235 | Disordered | 0.849210 | Binding | 0.386 | 0.923 | 0.750 | -1.962 | Likely Benign | 0.055 | Likely Benign | Likely Benign | 0.420 | Likely Benign | -0.34 | Neutral | 0.012 | Benign | 0.047 | Benign | 5.04 | Benign | 0.05 | Affected | 0.2730 | 0.3907 | 0 | -2 | 1.6 | -40.02 | |||||||||||||||||||||||||||||||||||||||
| c.3422C>A | P1141Q 2D  AIThe SynGAP1 missense variant P1141Q is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.930790 | Disordered | 0.716087 | Binding | 0.364 | 0.852 | 1.000 | -4.331 | Likely Benign | 0.274 | Likely Benign | Likely Benign | 0.136 | Likely Benign | -2.72 | Deleterious | 0.074 | Benign | 0.047 | Benign | 0.97 | Pathogenic | 0.00 | Affected | 0.1449 | 0.5186 | 0 | -1 | -1.9 | 31.01 | ||||||||||||||||||||||||||||||||||||||||
| c.3587A>G | K1196R 2D  AIThe SynGAP1 missense variant K1196R is not reported in ClinVar and is absent from gnomAD, indicating no known population frequency data. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a neutral effect, and the SGM‑Consensus score is “Likely Benign.” No tool in the dataset predicts pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates a likely benign effect. Foldetta results are unavailable, so they do not influence the assessment. Overall, the variant is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.599170 | Disordered | 0.435699 | Uncertain | 0.851 | 0.595 | 0.250 | -2.077 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.164 | Likely Benign | -0.78 | Neutral | 0.031 | Benign | 0.047 | Benign | 5.39 | Benign | 0.53 | Tolerated | 0.3970 | 0.0721 | 3 | 2 | -0.6 | 28.01 | ||||||||||||||||||||||||||||||||||||||
| c.358G>C | G120R 2D  AIThe SynGAP1 missense variant G120R is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign. Only AlphaMissense‑Default predicts a pathogenic outcome. When predictions are grouped, the benign consensus includes eight tools, whereas the pathogenic consensus contains a single tool. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—also yields a likely benign classification. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that G120R is most likely benign, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.659993 | Binding | 0.359 | 0.887 | 0.750 | -4.406 | Likely Benign | 0.745 | Likely Pathogenic | Likely Benign | 0.022 | Likely Benign | -0.33 | Neutral | 0.089 | Benign | 0.047 | Benign | 4.26 | Benign | 0.14 | Tolerated | 0.1050 | 0.4345 | -3 | -2 | -4.1 | 99.14 | |||||||||||||||||||||||||||||||||||||||
| c.3610T>G | S1204A 2D  AIThe SynGAP1 missense variant S1204A is not reported in ClinVar and is absent from gnomAD, indicating no known population frequency data. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a neutral effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.595080 | Disordered | 0.541098 | Binding | 0.887 | 0.587 | 0.375 | -5.134 | Likely Benign | 0.144 | Likely Benign | Likely Benign | 0.213 | Likely Benign | -0.53 | Neutral | 0.061 | Benign | 0.047 | Benign | 5.45 | Benign | 1.00 | Tolerated | 0.4943 | 0.3167 | 1 | 1 | 2.6 | -16.00 | ||||||||||||||||||||||||||||||||||||||
| c.401G>C | S134T 2D  AIThe SynGAP1 missense variant S134T is listed in gnomAD (ID 6‑33432698‑G‑C) but has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also favors benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact; this conclusion is not contradicted by ClinVar, which contains no classification for S134T. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.447574 | Structured | 0.695837 | Binding | 0.333 | 0.898 | 0.250 | 6-33432698-G-C | 1 | 6.32e-7 | -5.187 | Likely Benign | 0.563 | Ambiguous | Likely Benign | 0.069 | Likely Benign | -1.76 | Neutral | 0.034 | Benign | 0.047 | Benign | 3.85 | Benign | 0.00 | Affected | 3.61 | 5 | 0.1083 | 0.5326 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||||||
| c.416G>A | S139N 2D  AIThe SynGAP1 missense variant S139N is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33432713‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as likely benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.553315 | Disordered | 0.600637 | Binding | 0.353 | 0.900 | 0.250 | Uncertain | 1 | 6-33432713-G-A | 3 | 2.22e-6 | -4.584 | Likely Benign | 0.688 | Likely Pathogenic | Likely Benign | 0.109 | Likely Benign | -0.75 | Neutral | 0.149 | Benign | 0.047 | Benign | 4.14 | Benign | 0.24 | Tolerated | 3.61 | 5 | 0.1526 | 0.3614 | 1 | 1 | -2.7 | 27.03 | ||||||||||||||||||||||||||||||||
| c.50C>A | S17Y 2D  AIThe SynGAP1 missense variant S17Y is listed in gnomAD (ID 6‑33420314‑C‑A) but has no ClinVar entry. Functional prediction tools cluster into two groups: benign predictions include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Pathogenic predictions come from polyPhen‑2 HumDiv and SIFT, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments reinforce the benign trend: AlphaMissense‑Optimized scores the variant as benign, and the SGM‑Consensus (derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also favors benign. No Foldetta stability data are available, so it does not influence the conclusion. Overall, the majority of evidence indicates that S17Y is most likely benign, and this assessment does not contradict any ClinVar status, as none is reported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.483068 | Structured | 0.452228 | Uncertain | 0.341 | 0.910 | 0.375 | 6-33420314-C-A | -4.492 | Likely Benign | 0.551 | Ambiguous | Likely Benign | 0.050 | Likely Benign | -1.06 | Neutral | 0.742 | Possibly Damaging | 0.047 | Benign | 3.99 | Benign | 0.00 | Affected | 4.32 | 1 | 0.0818 | 0.6366 | -2 | -3 | -0.5 | 76.10 | ||||||||||||||||||||||||||||||||||||
| c.1526C>T | A509V 2D  3DClick to see structure in 3D Viewer AISynGAP1 A509V is not reported in ClinVar and is absent from gnomAD. High‑accuracy predictors give mixed results: AlphaMissense‑Optimized classifies the variant as benign, while the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—labels it pathogenic; Foldetta, which integrates FoldX‑MD and Rosetta outputs, is inconclusive because FoldX is uncertain and Rosetta is benign. Among the remaining tools, benign predictions come from REVEL, Rosetta, premPS, polyPhen2_HumDiv, polyPhen2_HumVar, and SIFT, whereas pathogenic predictions come from SGM‑Consensus, PROVEAN, ESM1b, and FATHMM. AlphaMissense‑Default and FoldX remain uncertain. Overall, the majority of evidence points toward a benign effect, and this assessment does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.025762 | Structured | 0.250110 | Uncertain | 0.923 | 0.256 | 0.000 | -11.987 | Likely Pathogenic | 0.382 | Ambiguous | Likely Benign | 0.52 | Ambiguous | 0.5 | 0.35 | Likely Benign | 0.44 | Likely Benign | -0.25 | Likely Benign | 0.474 | Likely Benign | -3.11 | Deleterious | 0.064 | Benign | 0.048 | Benign | -1.15 | Pathogenic | 0.18 | Tolerated | 0.1333 | 0.5706 | 0 | 0 | 2.4 | 28.05 | |||||||||||||||||||||||||||||
| c.2200C>T | P734S 2D  AIThe SynGAP1 missense variant P734S is listed in ClinVar with an uncertain significance (ClinVar ID 2283225.0) and is present in the gnomAD database (gnomAD ID 6‑33441665‑C‑T). Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign effects. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this benign assessment: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability predictor combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign classification, which is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.879233 | Disordered | 0.411273 | Uncertain | 0.368 | 0.721 | 0.875 | Uncertain | 2 | 6-33441665-C-T | 2 | 1.24e-6 | -4.291 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.030 | Likely Benign | -2.44 | Neutral | 0.344 | Benign | 0.048 | Benign | 2.77 | Benign | 0.11 | Tolerated | 3.64 | 6 | 0.3775 | 0.3650 | 1 | -1 | 0.8 | -10.04 | 10.1016/j.ajhg.2020.11.011 | |||||||||||||||||||||||||||||||
| c.2624C>G | A875G 2D  AIThe SynGAP1 missense variant A875G is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign classification. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of the majority of tools, including the high‑accuracy methods, points to a benign impact, and this assessment does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.545602 | Disordered | 0.632173 | Binding | 0.273 | 0.872 | 0.250 | -2.904 | Likely Benign | 0.111 | Likely Benign | Likely Benign | 0.078 | Likely Benign | -1.22 | Neutral | 0.022 | Benign | 0.048 | Benign | 2.68 | Benign | 0.04 | Affected | 0.2308 | 0.5046 | 1 | 0 | -2.2 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3115A>C | I1039L 2D  AIThe SynGAP1 missense variant I1039L is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” verdict. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, is not available for this variant. Overall, the consensus of all available predictions strongly supports a benign classification, with no contradiction to ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.969315 | Disordered | 0.979204 | Binding | 0.292 | 0.806 | 0.625 | -2.035 | Likely Benign | 0.269 | Likely Benign | Likely Benign | 0.083 | Likely Benign | -0.14 | Neutral | 0.264 | Benign | 0.048 | Benign | 2.82 | Benign | 0.98 | Tolerated | 0.1057 | 0.4605 | 2 | 2 | -0.7 | 0.00 | |||||||||||||||||||||||||||||||||||||||
| c.3115A>G | I1039V 2D  AIThe SynGAP1 missense variant I1039V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Based on the collective predictions, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.969315 | Disordered | 0.979204 | Binding | 0.292 | 0.806 | 0.625 | -2.455 | Likely Benign | 0.164 | Likely Benign | Likely Benign | 0.060 | Likely Benign | 0.21 | Neutral | 0.264 | Benign | 0.048 | Benign | 2.76 | Benign | 0.41 | Tolerated | 0.1405 | 0.4112 | 4 | 3 | -0.3 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.314C>T | S105L 2D  AIThe SynGAP1 missense variant S105L is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33432179‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. High‑accuracy methods both support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote) is benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.788093 | Disordered | 0.669201 | Binding | 0.364 | 0.870 | 0.625 | Uncertain | 2 | 6-33432179-C-T | 4 | 2.48e-6 | -3.710 | Likely Benign | 0.233 | Likely Benign | Likely Benign | 0.095 | Likely Benign | -1.52 | Neutral | 0.828 | Possibly Damaging | 0.048 | Benign | 4.06 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1161 | 0.5023 | -3 | -2 | 4.6 | 26.08 | ||||||||||||||||||||||||||||||||
| c.3209G>A | R1070K 2D  AIThe SynGAP1 missense variant R1070K is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. All evaluated in‑silico predictors agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” High‑accuracy tools reinforce this view: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta results are unavailable. Based on the unanimous benign predictions, the variant is most likely benign, and this assessment does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.930790 | Disordered | 0.982693 | Binding | 0.297 | 0.906 | 0.875 | Conflicting | 2 | -5.093 | Likely Benign | 0.326 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -1.42 | Neutral | 0.049 | Benign | 0.048 | Benign | 3.86 | Benign | 0.09 | Tolerated | 3.77 | 5 | 0.4997 | 0.4867 | 3 | 2 | 0.6 | -28.01 | |||||||||||||||||||||||||||||||||||
| c.50C>G | S17C 2D  AIThe SynGAP1 missense variant S17C is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only two tools (polyPhen‑2 HumDiv and SIFT) predict pathogenicity, while the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.483068 | Structured | 0.452228 | Uncertain | 0.341 | 0.910 | 0.375 | -4.364 | Likely Benign | 0.279 | Likely Benign | Likely Benign | 0.044 | Likely Benign | 0.17 | Neutral | 0.486 | Possibly Damaging | 0.048 | Benign | 3.99 | Benign | 0.00 | Affected | 0.1226 | 0.6721 | 0 | -1 | 3.3 | 16.06 | |||||||||||||||||||||||||||||||||||||||
| c.563G>A | S188N 2D  AIThe SynGAP1 missense variant S188N is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments are inconclusive: the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a 2‑vs‑2 split, and Foldetta results are unavailable. Overall, the majority of available predictions (five benign vs three pathogenic) suggest a benign impact. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.490133 | Structured | 0.428502 | Uncertain | 0.298 | 0.603 | 0.500 | -10.307 | Likely Pathogenic | 0.952 | Likely Pathogenic | Ambiguous | 0.095 | Likely Benign | -2.34 | Neutral | 0.259 | Benign | 0.048 | Benign | 3.92 | Benign | 0.01 | Affected | 0.1420 | 0.5658 | 1 | 1 | -2.7 | 27.03 | ||||||||||||||||||||||||||||||||||||||||
| c.637A>G | I213V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 I213V missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). No tool predicts a pathogenic outcome. Predictions that are inconclusive are FoldX, Rosetta, Foldetta, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, while Foldetta’s stability analysis is uncertain. Overall, the evidence strongly favors a benign classification, and this is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.158265 | Structured | 0.372201 | Uncertain | 0.850 | 0.295 | 0.125 | -6.133 | Likely Benign | 0.364 | Ambiguous | Likely Benign | 0.67 | Ambiguous | 0.2 | 0.58 | Ambiguous | 0.63 | Ambiguous | 0.47 | Likely Benign | 0.413 | Likely Benign | -0.69 | Neutral | 0.128 | Benign | 0.048 | Benign | 5.82 | Benign | 0.10 | Tolerated | 0.1008 | 0.2959 | 4 | 3 | -0.3 | -14.03 | |||||||||||||||||||||||||||||
| c.677C>T | S226L 2D  AIThe SynGAP1 missense variant S226L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include FoldX, Rosetta, Foldetta, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are REVEL, PROVEAN, and SIFT. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign; the SGM‑Consensus (majority vote) also indicates benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, predicts benign. No prediction or folding result is missing or inconclusive. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.129801 | Structured | 0.334959 | Uncertain | 0.800 | 0.324 | 0.250 | -6.098 | Likely Benign | 0.239 | Likely Benign | Likely Benign | -0.12 | Likely Benign | 0.5 | -0.14 | Likely Benign | -0.13 | Likely Benign | 0.36 | Likely Benign | 0.578 | Likely Pathogenic | -3.87 | Deleterious | 0.437 | Benign | 0.048 | Benign | 5.73 | Benign | 0.03 | Affected | 0.1525 | 0.5907 | -3 | -2 | 4.6 | 26.08 | |||||||||||||||||||||||||||||
| c.709G>T | A237S 2D  AIThe SynGAP1 missense variant A237S is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, AlphaMissense‑Optimized, and FATHMM. No tool predicts a pathogenic outcome; the only inconclusive results come from premPS and ESM1b, which are treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.200174 | Structured | 0.334699 | Uncertain | 0.719 | 0.352 | 0.000 | -7.696 | In-Between | 0.112 | Likely Benign | Likely Benign | 0.48 | Likely Benign | 0.6 | 0.34 | Likely Benign | 0.41 | Likely Benign | 0.61 | Ambiguous | 0.421 | Likely Benign | -1.41 | Neutral | 0.259 | Benign | 0.048 | Benign | 5.82 | Benign | 0.31 | Tolerated | 0.2209 | 0.4357 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.1120T>A | S374T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S374T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on benign impact include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is benign, and Foldetta, which integrates FoldX‑MD and Rosetta outputs, is uncertain but does not indicate destabilization. Overall, the evidence strongly favors a benign effect for S374T, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.642678 | Disordered | 0.428948 | Uncertain | 0.333 | 0.812 | 0.625 | -5.415 | Likely Benign | 0.088 | Likely Benign | Likely Benign | 0.39 | Likely Benign | 0.1 | 0.80 | Ambiguous | 0.60 | Ambiguous | -0.02 | Likely Benign | 0.176 | Likely Benign | -0.47 | Neutral | 0.118 | Benign | 0.049 | Benign | 5.32 | Benign | 0.12 | Tolerated | 0.2212 | 0.6466 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.1199T>C | V400A 2D  3DClick to see structure in 3D Viewer AISynGAP1 V400A is not reported in ClinVar and is absent from gnomAD. Consensus from standard predictors shows a split: benign calls from REVEL, polyPhen‑2 (HumDiv and HumVar) and FATHMM, while pathogenic calls come from FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT and AlphaMissense‑Default. Two high‑accuracy tools give a clear signal: AlphaMissense‑Optimized is uncertain, but the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is pathogenic, and Foldetta also predicts pathogenic. With most evidence pointing to deleterious effects, the variant is most likely pathogenic, and this assessment does not conflict with the absence of a ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.398279 | Structured | 0.415488 | Uncertain | 0.951 | 0.451 | 0.000 | -7.564 | In-Between | 0.871 | Likely Pathogenic | Ambiguous | 3.14 | Destabilizing | 0.1 | 3.12 | Destabilizing | 3.13 | Destabilizing | 2.29 | Destabilizing | 0.479 | Likely Benign | -3.12 | Deleterious | 0.435 | Benign | 0.049 | Benign | 5.32 | Benign | 0.01 | Affected | 0.3291 | 0.2767 | 0 | 0 | -2.4 | -28.05 | ||||||||||||||||||||||||||||||
| c.2374G>C | E792Q 2D  AIThe SynGAP1 missense variant E792Q is not reported in ClinVar and has no entry in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts it to be pathogenic, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” verdict, reflecting the majority of benign calls. High‑accuracy assessments further support this view: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote) is benign; Foldetta results are unavailable. Overall, the preponderance of evidence indicates that E792Q is most likely benign, and this conclusion does not contradict the absence of a ClinVar entry. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.974374 | Disordered | 0.452261 | Uncertain | 0.352 | 0.896 | 0.875 | -3.782 | Likely Benign | 0.411 | Ambiguous | Likely Benign | 0.052 | Likely Benign | -1.60 | Neutral | 0.077 | Benign | 0.049 | Benign | 3.89 | Benign | 0.02 | Affected | 0.1469 | 0.7401 | 2 | 2 | 0.0 | -0.98 | ||||||||||||||||||||||||||||||||||||||
| c.2894A>G | H965R 2D  AIThe SynGAP1 missense variant H965R is catalogued in gnomAD (ID 6‑33443446‑A‑G) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all classify the change as benign or likely benign. No tool predicts pathogenicity; ESM1b is uncertain but does not contradict the benign consensus. High‑accuracy assessments confirm this view: AlphaMissense‑Optimized reports a benign effect, and the SGM‑Consensus likewise indicates a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant, so it does not influence the assessment. Overall, the available predictions strongly suggest that H965R is most likely benign, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.988505 | Disordered | 0.978700 | Binding | 0.342 | 0.882 | 0.750 | 6-33443446-A-G | 1 | 6.20e-7 | -7.056 | In-Between | 0.156 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -0.88 | Neutral | 0.065 | Benign | 0.049 | Benign | 4.08 | Benign | 0.38 | Tolerated | 3.77 | 5 | 0.2394 | 0.3610 | 0 | 2 | -1.3 | 19.05 | ||||||||||||||||||||||||||||||||||
| c.3127A>G | R1043G 2D  AIThe SynGAP1 missense variant R1043G is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none exists). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.978672 | Disordered | 0.954069 | Binding | 0.299 | 0.853 | 0.625 | -2.168 | Likely Benign | 0.201 | Likely Benign | Likely Benign | 0.383 | Likely Benign | -3.17 | Deleterious | 0.130 | Benign | 0.049 | Benign | 5.94 | Benign | 0.00 | Affected | 0.3179 | 0.3697 | -3 | -2 | 4.1 | -99.14 | |||||||||||||||||||||||||||||||||||||||
| c.3139T>A | S1047T 2D  AIThe SynGAP1 missense variant S1047T is not reported in ClinVar and is absent from gnomAD, indicating no documented allele frequency data. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign status. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is that the variant is most likely benign, and this conclusion is consistent with the lack of ClinVar evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.963420 | Disordered | 0.933764 | Binding | 0.409 | 0.909 | 0.750 | -4.222 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.029 | Likely Benign | 0.23 | Neutral | 0.069 | Benign | 0.049 | Benign | 2.63 | Benign | 0.63 | Tolerated | 0.1796 | 0.6265 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.32G>T | G11V 2D  AIThe SynGAP1 missense variant G11V is reported in gnomAD (variant ID 6‑33420296‑G‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. In contrast, polyPhen‑2 HumDiv and SIFT predict a pathogenic impact. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also likely benign. No Foldetta stability prediction is available, so it does not influence the assessment. Overall, the preponderance of evidence from both general and high‑accuracy tools indicates that G11V is most likely benign, and this conclusion is not contradicted by any ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.444081 | Structured | 0.501027 | Binding | 0.348 | 0.915 | 0.375 | 6-33420296-G-T | -4.079 | Likely Benign | 0.160 | Likely Benign | Likely Benign | 0.146 | Likely Benign | -0.38 | Neutral | 0.668 | Possibly Damaging | 0.049 | Benign | 3.93 | Benign | 0.00 | Affected | 4.32 | 1 | 0.1292 | 0.4522 | -3 | -1 | 4.6 | 42.08 | ||||||||||||||||||||||||||||||||||||
| c.521T>C | M174T 2D  AIThe SynGAP1 missense variant M174T is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect comprise PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Pathogenic. High‑accuracy assessments further support a deleterious impact: AlphaMissense‑Optimized predicts pathogenicity, and the SGM‑Consensus (majority vote) also indicates Likely Pathogenic. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence points to a pathogenic effect, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.661982 | Disordered | 0.485854 | Uncertain | 0.373 | 0.620 | 0.375 | -9.174 | Likely Pathogenic | 0.985 | Likely Pathogenic | Likely Pathogenic | 0.286 | Likely Benign | -3.12 | Deleterious | 0.244 | Benign | 0.049 | Benign | 4.08 | Benign | 0.01 | Affected | 0.1949 | 0.2334 | -1 | -1 | -2.6 | -30.09 | |||||||||||||||||||||||||||||||||||||||
| c.673T>A | S225T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S225T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, PROVEAN, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is also benign; Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is inconclusive. No pathogenic predictions are present. Therefore, based on the available computational evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.150080 | Structured | 0.344191 | Uncertain | 0.817 | 0.319 | 0.250 | -6.351 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.64 | Ambiguous | 0.4 | 0.59 | Ambiguous | 0.62 | Ambiguous | 0.03 | Likely Benign | 0.244 | Likely Benign | -1.98 | Neutral | 0.118 | Benign | 0.049 | Benign | 5.73 | Benign | 0.55 | Tolerated | 0.1419 | 0.7395 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.71T>G | V24G 2D  AIThe SynGAP1 missense variant V24G is not reported in ClinVar (ClinVar status: not present) and is absent from gnomAD (gnomAD ID: none). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is benign, and Foldetta results are unavailable. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.541878 | Disordered | 0.438970 | Uncertain | 0.382 | 0.890 | 0.500 | -2.673 | Likely Benign | 0.256 | Likely Benign | Likely Benign | 0.178 | Likely Benign | -1.67 | Neutral | 0.026 | Benign | 0.049 | Benign | 3.77 | Benign | 0.00 | Affected | 0.2081 | 0.3105 | -1 | -3 | -4.6 | -42.08 | ||||||||||||||||||||||||||||||||||||||||
| c.142T>A | F48I 2D  AIThe SynGAP1 missense variant F48I is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), and FATHMM, while SIFT and AlphaMissense‑Default predict a pathogenic impact. Two tools—ESM1b and AlphaMissense‑Optimized—return uncertain results. High‑accuracy assessments further clarify the variant’s likely benign nature: the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, favors benign; AlphaMissense‑Optimized remains uncertain, and Foldetta (which would evaluate protein‑folding stability) is unavailable. Overall, the balance of evidence points to a benign classification, and this conclusion does not contradict any ClinVar annotation, as none exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.298791 | Structured | 0.440452 | Uncertain | 0.558 | 0.707 | 0.125 | -7.994 | In-Between | 0.808 | Likely Pathogenic | Ambiguous | 0.108 | Likely Benign | -1.67 | Neutral | 0.092 | Benign | 0.050 | Benign | 3.99 | Benign | 0.00 | Affected | 0.2561 | 0.2301 | 1 | 0 | 1.7 | -34.02 | ||||||||||||||||||||||||||||||||||||||||
| c.152T>C | I51T 2D  AIThe SynGAP1 missense variant I51T is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include SGM‑Consensus, REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default, while AlphaMissense‑Optimized is uncertain. High‑accuracy assessments show that the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is Likely Benign, AlphaMissense‑Optimized remains Uncertain, and no Foldetta stability result is available. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.291804 | Structured | 0.454181 | Uncertain | 0.606 | 0.710 | 0.000 | -5.861 | Likely Benign | 0.881 | Likely Pathogenic | Ambiguous | 0.135 | Likely Benign | -1.07 | Neutral | 0.084 | Benign | 0.050 | Benign | 4.16 | Benign | 0.00 | Affected | 0.1180 | 0.1265 | 0 | -1 | -5.2 | -12.05 | |||||||||||||||||||||||||||||||||||||||
| c.1769G>C | S590T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S590T is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and ESM1b. Two tools (premPS and AlphaMissense‑Default) return uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) as benign. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.022667 | Structured | 0.088943 | Uncertain | 0.918 | 0.199 | 0.000 | -12.472 | Likely Pathogenic | 0.458 | Ambiguous | Likely Benign | 0.23 | Likely Benign | 0.1 | 0.42 | Likely Benign | 0.33 | Likely Benign | 0.64 | Ambiguous | 0.459 | Likely Benign | -2.89 | Deleterious | 0.183 | Benign | 0.050 | Benign | 3.15 | Benign | 0.08 | Tolerated | 0.1420 | 0.5723 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||
| c.2101C>A | P701T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant P701T is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the substitution as benign: REVEL, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, PROVEAN, AlphaMissense‑Default, and AlphaMissense‑Optimized all indicate a tolerated change. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign; the SGM Consensus, derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports benign. Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is inconclusive, and FoldX alone is uncertain, so these results are treated as unavailable. Overall, the evidence strongly supports a benign effect for P701T, and this assessment does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.074921 | Structured | 0.404318 | Uncertain | 0.918 | 0.345 | 0.000 | -6.920 | Likely Benign | 0.272 | Likely Benign | Likely Benign | 1.61 | Ambiguous | 0.0 | 0.11 | Likely Benign | 0.86 | Ambiguous | -0.18 | Likely Benign | 0.144 | Likely Benign | -0.43 | Neutral | 0.084 | Benign | 0.050 | Benign | 3.44 | Benign | 0.70 | Tolerated | 0.1398 | 0.4160 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||
| c.316A>G | R106G 2D  AIThe SynGAP1 missense variant R106G is listed in gnomAD (ID 6‑33432181‑A‑G) but has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM. Those that predict a pathogenic effect are SIFT and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, SGM‑Consensus as likely benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. There is no ClinVar classification to contradict this assessment. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.663409 | Binding | 0.345 | 0.862 | 0.875 | 6-33432181-A-G | 1 | 6.20e-7 | -2.617 | Likely Benign | 0.835 | Likely Pathogenic | Ambiguous | 0.161 | Likely Benign | -2.21 | Neutral | 0.421 | Benign | 0.050 | Benign | 3.65 | Benign | 0.00 | Affected | 4.05 | 3 | 0.3680 | 0.3980 | -2 | -3 | 4.1 | -99.14 | ||||||||||||||||||||||||||||||||||
| c.319A>G | R107G 2D  AIThe SynGAP1 missense variant R107G is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM, while those that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments are inconclusive: AlphaMissense‑Optimized is uncertain, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is a tie and thus unavailable, and Foldetta stability analysis is missing. Overall, the majority of tools (five benign vs. three pathogenic) suggest a benign impact, but the lack of consensus from the most accurate predictors means the variant’s effect remains uncertain. This assessment does not contradict any ClinVar status, as none is available. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.666105 | Disordered | 0.663448 | Binding | 0.331 | 0.863 | 0.875 | -3.486 | Likely Benign | 0.948 | Likely Pathogenic | Ambiguous | 0.180 | Likely Benign | -3.15 | Deleterious | 0.421 | Benign | 0.050 | Benign | 2.98 | Benign | 0.00 | Affected | 0.3280 | 0.4000 | -3 | -2 | 4.1 | -99.14 | ||||||||||||||||||||||||||||||||||||||||
| c.320G>C | R107T 2D  AIThe SynGAP1 missense variant R107T is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized classifying the variant as pathogenic, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive (2 pathogenic vs. 2 benign) and Foldetta results are unavailable. Overall, the majority of predictions (five benign vs. four pathogenic) lean toward a benign interpretation. Thus, the variant is most likely benign based on current computational evidence, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.666105 | Disordered | 0.663448 | Binding | 0.331 | 0.863 | 0.875 | -2.902 | Likely Benign | 0.992 | Likely Pathogenic | Likely Pathogenic | 0.191 | Likely Benign | -2.63 | Deleterious | 0.421 | Benign | 0.050 | Benign | 2.98 | Benign | 0.00 | Affected | 0.1549 | 0.4761 | -1 | -1 | 3.8 | -55.08 | ||||||||||||||||||||||||||||||||||||||||
| c.326G>C | S109T 2D  AIThe SynGAP1 missense variant S109T is catalogued in gnomAD (6‑33432191‑G‑C) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all report benign or likely benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments confirm the benign consensus: AlphaMissense‑Optimized is benign, and the SGM‑Consensus, derived from the majority of the high‑accuracy predictors, is benign. Foldetta, a protein‑folding stability method that integrates FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that S109T is most likely benign, and this assessment does not contradict any ClinVar status (none). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.622677 | Disordered | 0.669335 | Binding | 0.328 | 0.864 | 0.750 | 6-33432191-G-C | 2 | 1.24e-6 | -4.065 | Likely Benign | 0.449 | Ambiguous | Likely Benign | 0.090 | Likely Benign | -1.19 | Neutral | 0.231 | Benign | 0.050 | Benign | 3.59 | Benign | 0.00 | Affected | 3.61 | 5 | 0.1519 | 0.5212 | 1 | 1 | 0.1 | 14.03 | ||||||||||||||||||||||||||||||||||
| c.331C>A | P111T 2D  AIThe SynGAP1 missense variant P111T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is also benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.650020 | Binding | 0.438 | 0.858 | 0.750 | -2.800 | Likely Benign | 0.305 | Likely Benign | Likely Benign | 0.061 | Likely Benign | -1.44 | Neutral | 0.421 | Benign | 0.050 | Benign | 4.11 | Benign | 0.02 | Affected | 0.1650 | 0.5402 | 0 | -1 | 0.9 | 3.99 | |||||||||||||||||||||||||||||||||||||||
| c.362C>A | A121D 2D  AIThe SynGAP1 missense variant A121D is not reported in ClinVar and has no entry in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates Likely Benign. No Foldetta stability prediction is available for this variant. Overall, the majority of computational evidence points to a benign impact, and this is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.779859 | Disordered | 0.661304 | Binding | 0.362 | 0.888 | 0.750 | -3.626 | Likely Benign | 0.595 | Likely Pathogenic | Likely Benign | 0.096 | Likely Benign | -0.89 | Neutral | 0.244 | Benign | 0.050 | Benign | 4.06 | Benign | 0.03 | Affected | 0.1876 | 0.1945 | 0 | -2 | -5.3 | 44.01 | |||||||||||||||||||||||||||||||||||||||
| c.362C>T | A121V 2D  AIThe SynGAP1 missense variant A121V is reported in gnomAD (ID 6‑33432227‑C‑T) but has no ClinVar entry. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign, while only SIFT predicts it as pathogenic. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Overall, the preponderance of evidence supports a benign classification for A121V, and this conclusion is not contradicted by any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.779859 | Disordered | 0.661304 | Binding | 0.362 | 0.888 | 0.750 | 6-33432227-C-T | 3 | 1.86e-6 | -3.818 | Likely Benign | 0.086 | Likely Benign | Likely Benign | 0.046 | Likely Benign | -1.44 | Neutral | 0.213 | Benign | 0.050 | Benign | 4.04 | Benign | 0.02 | Affected | 3.61 | 5 | 0.1450 | 0.6898 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||||
| c.676T>G | S226A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S226A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Functional prediction tools uniformly indicate a benign effect: REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all classify the variant as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus predicts likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also predicts benign. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status (none available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.129801 | Structured | 0.334959 | Uncertain | 0.800 | 0.324 | 0.250 | -4.665 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.06 | Likely Benign | 0.0 | 0.25 | Likely Benign | 0.16 | Likely Benign | -0.02 | Likely Benign | 0.339 | Likely Benign | -1.64 | Neutral | 0.123 | Benign | 0.050 | Benign | 5.76 | Benign | 0.06 | Tolerated | 0.4812 | 0.5599 | 1 | 1 | 2.6 | -16.00 | |||||||||||||||||||||||||||||
| c.94A>T | T32S 2D  AIThe SynGAP1 missense variant T32S is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.437154 | Uncertain | 0.349 | 0.879 | 0.375 | -2.618 | Likely Benign | 0.072 | Likely Benign | Likely Benign | 0.062 | Likely Benign | -0.39 | Neutral | 0.171 | Benign | 0.050 | Benign | 4.18 | Benign | 0.00 | Affected | 0.3603 | 0.5174 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.95C>G | T32S 2D  AIThe SynGAP1 missense variant T32S is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the consensus of the majority of tools and the high‑accuracy predictions point to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.437154 | Uncertain | 0.349 | 0.879 | 0.375 | -2.618 | Likely Benign | 0.072 | Likely Benign | Likely Benign | 0.024 | Likely Benign | -0.39 | Neutral | 0.171 | Benign | 0.050 | Benign | 4.18 | Benign | 0.00 | Affected | 0.3603 | 0.5174 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2015C>A | T672K 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant T672K is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are SGM‑Consensus, PROVEAN, polyPhen‑2 HumDiv, ESM1b, and AlphaMissense‑Default. Uncertain predictions come from Foldetta, premPS, and Rosetta. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta as inconclusive. Overall, the majority of tools lean toward a benign interpretation, but the high‑accuracy consensus is split, leaving the variant’s impact uncertain. Thus, the variant is most likely benign based on the bulk of predictions, and this does not contradict its ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.116183 | Structured | 0.102069 | Uncertain | 0.586 | 0.362 | 0.000 | Uncertain | 1 | -12.192 | Likely Pathogenic | 0.698 | Likely Pathogenic | Likely Benign | 0.20 | Likely Benign | 0.5 | 1.21 | Ambiguous | 0.71 | Ambiguous | 0.72 | Ambiguous | 0.065 | Likely Benign | -4.31 | Deleterious | 0.745 | Possibly Damaging | 0.051 | Benign | 3.40 | Benign | 0.07 | Tolerated | 3.40 | 25 | 0.1152 | 0.3250 | 0 | -1 | -3.2 | 27.07 | 195.1 | 7.0 | 0.4 | 0.7 | 0.4 | 0.1 | X | X | Potentially Pathogenic | The hydroxyl group of Thr672, located in an entangled α-α loop connecting the two α-helices (res. Ser641-Glu666 and res. Leu685-Val699), is involved in a highly coordinated hydrogen-bonding network between residues from two α-helices (res. Ser641-Glu666 and res. Arg563-Glu578) and from the α-α loop itself, such as Lys566, Glu666, and Asn669. In the variant simulations, Lys672 can only form a hydrogen bond with the amino group of the Lys566 side chain via its backbone carbonyl group. Consequently, it cannot maintain the Lys566-Glu666 salt bridge through hydrogen bonding. However, the amino group of Lys periodically forms a salt bridge with the carboxylate group of Glu666, which prevents a drastic disruption of the hydrogen-bond network that keeps the loop close to the helices. | |||||||||||||||
| c.2116G>C | E706Q 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 E706Q missense variant has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are AlphaMissense‑Default and ESM1b. Rosetta and Foldetta provide uncertain results and are treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 pathogenic vs. 2 benign). Foldetta also yields an uncertain outcome. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict ClinVar status, as no ClinVar classification exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.200174 | Structured | 0.377033 | Uncertain | 0.929 | 0.363 | 0.000 | -8.171 | Likely Pathogenic | 0.588 | Likely Pathogenic | Likely Benign | 0.44 | Likely Benign | 0.0 | 0.61 | Ambiguous | 0.53 | Ambiguous | -0.14 | Likely Benign | 0.059 | Likely Benign | -1.00 | Neutral | 0.433 | Benign | 0.051 | Benign | 4.13 | Benign | 0.31 | Tolerated | 0.0941 | 0.3831 | 2 | 2 | 0.0 | -0.98 | ||||||||||||||||||||||||||||||
| c.1931A>T | D644V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 D644V missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, Rosetta, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive because it yields a 2‑to‑2 split. Overall, the majority of evidence points to a benign impact. Thus, the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.066181 | Structured | 0.248888 | Uncertain | 0.883 | 0.320 | 0.000 | -6.230 | Likely Benign | 0.636 | Likely Pathogenic | Likely Benign | 0.50 | Ambiguous | 0.0 | -0.35 | Likely Benign | 0.08 | Likely Benign | -0.03 | Likely Benign | 0.347 | Likely Benign | -4.96 | Deleterious | 0.198 | Benign | 0.052 | Benign | 3.49 | Benign | 0.11 | Tolerated | 0.0951 | 0.6267 | -2 | -3 | 7.7 | -15.96 | ||||||||||||||||||||||||||||||
| c.1999A>G | I667V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 I667V missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Only ESM1b predicts a pathogenic outcome; all other tools (FoldX, Rosetta, premPS, AlphaMissense‑Default, Foldetta) are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized classifying the variant as benign, while the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign majority vote. Foldetta’s stability prediction is uncertain. Overall, the preponderance of evidence points to a benign effect. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.142424 | Structured | 0.083597 | Uncertain | 0.927 | 0.379 | 0.000 | -8.482 | Likely Pathogenic | 0.406 | Ambiguous | Likely Benign | 1.32 | Ambiguous | 0.0 | 1.06 | Ambiguous | 1.19 | Ambiguous | 0.85 | Ambiguous | 0.234 | Likely Benign | -0.86 | Neutral | 0.341 | Benign | 0.052 | Benign | 3.35 | Benign | 0.15 | Tolerated | 0.1178 | 0.3059 | 4 | 3 | -0.3 | -14.03 | ||||||||||||||||||||||||||||||
| c.3194C>G | P1065R 2D  AIThe SynGAP1 missense variant P1065R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the preponderance of evidence from multiple independent predictors indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.979741 | Disordered | 0.959518 | Binding | 0.424 | 0.917 | 0.875 | -3.237 | Likely Benign | 0.228 | Likely Benign | Likely Benign | 0.043 | Likely Benign | -2.46 | Neutral | 0.102 | Benign | 0.052 | Benign | 2.00 | Pathogenic | 0.00 | Affected | 0.1439 | 0.4369 | 0 | -2 | -2.9 | 59.07 | |||||||||||||||||||||||||||||||||||||||
| c.3288G>C | E1096D 2D  AIThe SynGAP1 missense variant E1096D is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the collective evidence strongly supports a benign interpretation, and this conclusion does not contradict the ClinVar status, which currently contains no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.926919 | Disordered | 0.976475 | Binding | 0.308 | 0.858 | 1.000 | -3.818 | Likely Benign | 0.192 | Likely Benign | Likely Benign | 0.116 | Likely Benign | -0.40 | Neutral | 0.115 | Benign | 0.052 | Benign | 2.71 | Benign | 0.38 | Tolerated | 0.2034 | 0.4994 | 3 | 2 | 0.0 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3288G>T | E1096D 2D  AIThe SynGAP1 missense variant E1096D is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the collective evidence strongly supports a benign classification, and this conclusion is not contradicted by any ClinVar status, as no ClinVar entry exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.926919 | Disordered | 0.976475 | Binding | 0.308 | 0.858 | 1.000 | -3.818 | Likely Benign | 0.192 | Likely Benign | Likely Benign | 0.116 | Likely Benign | -0.40 | Neutral | 0.115 | Benign | 0.052 | Benign | 2.71 | Benign | 0.38 | Tolerated | 0.2034 | 0.4994 | 3 | 2 | 0.0 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.440A>G | Q147R 2D  AIThe SynGAP1 missense variant Q147R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect are SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments are inconclusive: the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a 2‑to‑2 split, and Foldetta results are unavailable. Overall, more tools predict a benign outcome (five vs. three pathogenic predictions, with one uncertain). Therefore, the variant is most likely benign based on current computational evidence, and this assessment does not contradict any ClinVar status because no ClinVar claim exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.541878 | Disordered | 0.503877 | Binding | 0.349 | 0.840 | 0.625 | -12.301 | Likely Pathogenic | 0.952 | Likely Pathogenic | Ambiguous | 0.166 | Likely Benign | -2.22 | Neutral | 0.079 | Benign | 0.052 | Benign | 3.91 | Benign | 0.02 | Affected | 0.1760 | 0.1042 | 1 | 1 | -1.0 | 28.06 | ||||||||||||||||||||||||||||||||||||||||
| c.505G>A | D169N 2D  AIThe SynGAP1 missense variant D169N is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools show a split: six methods (REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Optimized) predict a benign effect, while three (SIFT, ESM1b, AlphaMissense‑Default) predict pathogenicity. High‑accuracy assessments are mixed: AlphaMissense‑Optimized indicates benign, whereas the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—yields a tie and is therefore inconclusive. No Foldetta stability data are available. Overall, the balance of evidence leans toward a benign impact, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.418646 | Structured | 0.497160 | Uncertain | 0.420 | 0.675 | 0.125 | Uncertain | 1 | -10.713 | Likely Pathogenic | 0.761 | Likely Pathogenic | Likely Benign | 0.110 | Likely Benign | -2.04 | Neutral | 0.079 | Benign | 0.052 | Benign | 4.07 | Benign | 0.01 | Affected | 3.74 | 4 | 0.1430 | 0.7391 | 2 | 1 | 0.0 | -0.98 | ||||||||||||||||||||||||||||||||||||
| c.1061C>A | A354D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A354D is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, PROVEAN, polyPhen‑2 (HumDiv and HumVar), and SIFT. Those that predict a pathogenic effect are FATHMM and AlphaMissense‑Default. Four tools (Foldetta, premPS, ESM1b, and Rosetta) return uncertain results and are treated as unavailable evidence. High‑accuracy assessments show AlphaMissense‑Optimized predicts a benign outcome, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) favors a pathogenic interpretation. Foldetta’s stability prediction is uncertain. Overall, the majority of consensus tools lean toward a benign effect, and there is no ClinVar annotation to contradict this assessment. Thus, the variant is most likely benign based on the available predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.203355 | Structured | 0.381329 | Uncertain | 0.882 | 0.335 | 0.125 | -7.914 | In-Between | 0.725 | Likely Pathogenic | Likely Benign | 0.22 | Likely Benign | 0.1 | 1.10 | Ambiguous | 0.66 | Ambiguous | 0.57 | Ambiguous | 0.087 | Likely Benign | -1.49 | Neutral | 0.255 | Benign | 0.053 | Benign | 1.75 | Pathogenic | 0.07 | Tolerated | 0.1759 | 0.1345 | 0 | -2 | -5.3 | 44.01 | ||||||||||||||||||||||||||||||
| c.1660G>C | V554L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V554L has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only AlphaMissense‑Default predicts a pathogenic outcome, while FoldX, premPS, and ESM1b are uncertain. High‑accuracy assessments reinforce the benign interpretation: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign consensus; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also predicts benign. No prediction or stability result is missing or inconclusive. Overall, the variant is most likely benign based on the available computational evidence, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.020522 | Structured | 0.007349 | Uncertain | 0.955 | 0.226 | 0.000 | -7.884 | In-Between | 0.645 | Likely Pathogenic | Likely Benign | -1.14 | Ambiguous | 0.0 | 0.29 | Likely Benign | -0.43 | Likely Benign | 0.56 | Ambiguous | 0.162 | Likely Benign | -1.54 | Neutral | 0.204 | Benign | 0.053 | Benign | 3.49 | Benign | 0.08 | Tolerated | 0.0991 | 0.2998 | 2 | 1 | -0.4 | 14.03 | ||||||||||||||||||||||||||||||
| c.1660G>T | V554L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V554L has no ClinVar entry and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only AlphaMissense‑Default predicts a pathogenic outcome, while FoldX, premPS, and ESM1b are uncertain. High‑accuracy assessments reinforce the benign interpretation: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign consensus; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also predicts benign. No prediction or stability result is missing or inconclusive. Overall, the variant is most likely benign based on the available computational evidence, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.020522 | Structured | 0.007349 | Uncertain | 0.955 | 0.226 | 0.000 | -7.884 | In-Between | 0.645 | Likely Pathogenic | Likely Benign | -1.14 | Ambiguous | 0.0 | 0.29 | Likely Benign | -0.43 | Likely Benign | 0.56 | Ambiguous | 0.162 | Likely Benign | -1.54 | Neutral | 0.204 | Benign | 0.053 | Benign | 3.49 | Benign | 0.08 | Tolerated | 0.0991 | 0.2998 | 2 | 1 | -0.4 | 14.03 | ||||||||||||||||||||||||||||||
| c.2033G>C | S678T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S678T is not reported in ClinVar and is absent from gnomAD. All available in‑silico predictors classify the change as benign: REVEL, FoldX, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). No tool predicts pathogenicity. High‑accuracy assessments likewise support a benign effect: AlphaMissense‑Optimized is benign, the SGM Consensus is “Likely Benign,” and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign stability. Based on the unanimous computational evidence, the variant is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.301917 | Structured | 0.123585 | Uncertain | 0.660 | 0.321 | 0.000 | -5.389 | Likely Benign | 0.083 | Likely Benign | Likely Benign | 0.32 | Likely Benign | 0.1 | 0.41 | Likely Benign | 0.37 | Likely Benign | 0.14 | Likely Benign | 0.053 | Likely Benign | -1.82 | Neutral | 0.255 | Benign | 0.053 | Benign | 3.41 | Benign | 0.11 | Tolerated | 0.1494 | 0.6351 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||
| c.2185A>T | N729Y 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N729Y is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Foldetta, premPS, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only polyPhen‑2 HumDiv predicts a pathogenic outcome, while Rosetta remains uncertain. The high‑accuracy consensus methods give a consistent benign signal: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. Taken together, the overwhelming majority of evidence points to a benign effect. Thus, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.750527 | Disordered | 0.426547 | Uncertain | 0.651 | 0.583 | 0.625 | -2.284 | Likely Benign | 0.216 | Likely Benign | Likely Benign | 0.00 | Likely Benign | 0.1 | 0.82 | Ambiguous | 0.41 | Likely Benign | 0.08 | Likely Benign | 0.060 | Likely Benign | -2.35 | Neutral | 0.575 | Possibly Damaging | 0.053 | Benign | 3.27 | Benign | 0.14 | Tolerated | 0.0570 | 0.4073 | -2 | -2 | 2.2 | 49.07 | ||||||||||||||||||||||||||||||
| c.2264T>C | M755T 2D  AIThe SynGAP1 missense variant M755T is not reported in ClinVar and is absent from gnomAD. Consensus from multiple in silico predictors indicates a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM all classify the change as benign, and AlphaMissense‑Optimized also predicts benign. No tool predicts pathogenicity; AlphaMissense‑Default is uncertain. The SGM‑Consensus, which aggregates majority votes from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as likely benign. High‑accuracy assessments further support this: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta data are not available. Overall, the computational evidence overwhelmingly suggests the variant is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.490133 | Structured | 0.783855 | Binding | 0.336 | 0.873 | 0.375 | -4.116 | Likely Benign | 0.358 | Ambiguous | Likely Benign | 0.040 | Likely Benign | -1.23 | Neutral | 0.159 | Benign | 0.053 | Benign | 2.65 | Benign | 0.19 | Tolerated | 0.1944 | 0.1767 | -1 | -1 | -2.6 | -30.09 | |||||||||||||||||||||||||||||||||||||||
| c.2507G>T | S836I 2D  AIThe SynGAP1 missense variant S836I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Consensus from standard in silico predictors shows five tools (REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, AlphaMissense‑Optimized) predict a benign effect, while two tools (PROVEAN, SIFT) predict pathogenicity. Two additional predictors (ESM1b, AlphaMissense‑Default) return uncertain results. High‑accuracy assessment further indicates a benign prediction from AlphaMissense‑Optimized; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive (one benign, one pathogenic, two uncertain). Foldetta stability analysis is unavailable. Overall, the balance of evidence favors a benign classification, and this conclusion does not conflict with the absence of a ClinVar annotation. Therefore, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.525368 | Disordered | 0.634582 | Binding | 0.269 | 0.859 | 0.250 | -7.751 | In-Between | 0.517 | Ambiguous | Likely Benign | 0.149 | Likely Benign | -3.33 | Deleterious | 0.057 | Benign | 0.053 | Benign | 2.51 | Benign | 0.03 | Affected | 0.0756 | 0.5023 | -1 | -2 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||||||||||||
| c.253A>C | T85P 2D  AIThe SynGAP1 missense variant T85P is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, and FATHMM. Tools that agree on a pathogenic effect include polyPhen‑2 HumDiv, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments are mixed: AlphaMissense‑Optimized predicts pathogenic, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields no clear majority and is therefore unavailable; Foldetta results are not provided. Overall, the majority of available predictions (five pathogenic vs. four benign) indicate a pathogenic effect. There is no ClinVar entry to contradict this assessment, so the variant is most likely pathogenic based on current computational evidence. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.680603 | Disordered | 0.542004 | Binding | 0.288 | 0.888 | 0.500 | -8.406 | Likely Pathogenic | 0.978 | Likely Pathogenic | Likely Pathogenic | 0.125 | Likely Benign | -2.28 | Neutral | 0.813 | Possibly Damaging | 0.053 | Benign | 3.80 | Benign | 0.00 | Affected | 0.1499 | 0.4485 | 0 | -1 | -0.9 | -3.99 | ||||||||||||||||||||||||||||||||||||||||
| c.1106C>A | T369K 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T369K is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are FATHMM and AlphaMissense‑Default. Foldetta and Rosetta give uncertain results, which are treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (two benign vs two pathogenic). Foldetta’s stability prediction is uncertain. Overall, the majority of evidence (nine benign vs two pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.468512 | Structured | 0.437011 | Uncertain | 0.417 | 0.707 | 0.500 | -5.884 | Likely Benign | 0.616 | Likely Pathogenic | Likely Benign | -0.10 | Likely Benign | 0.1 | 1.13 | Ambiguous | 0.52 | Ambiguous | 0.27 | Likely Benign | 0.102 | Likely Benign | -1.87 | Neutral | 0.118 | Benign | 0.054 | Benign | 1.84 | Pathogenic | 0.34 | Tolerated | 0.1387 | 0.3855 | 0 | -1 | -3.2 | 27.07 | ||||||||||||||||||||||||||||||
| c.1196C>A | A399D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A399D is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Those that agree on a pathogenic effect are REVEL, FoldX, SIFT, ESM1b, and AlphaMissense‑Default. The remaining tools—Rosetta, Foldetta, premPS, and AlphaMissense‑Optimized—return uncertain or inconclusive results. High‑accuracy methods give no definitive verdict: AlphaMissense‑Optimized is uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is a tie and thus unavailable, and Foldetta is uncertain. Overall, the majority of available predictions (five pathogenic vs. four benign) lean toward pathogenicity. Therefore, the variant is most likely pathogenic, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.394753 | Structured | 0.407674 | Uncertain | 0.939 | 0.490 | 0.125 | -10.441 | Likely Pathogenic | 0.943 | Likely Pathogenic | Ambiguous | 2.45 | Destabilizing | 0.2 | 1.17 | Ambiguous | 1.81 | Ambiguous | 0.91 | Ambiguous | 0.525 | Likely Pathogenic | -1.84 | Neutral | 0.421 | Benign | 0.054 | Benign | 5.38 | Benign | 0.05 | Affected | 0.1533 | 0.1760 | 0 | -2 | -5.3 | 44.01 | ||||||||||||||||||||||||||||||
| c.1585A>T | I529F 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I529F is not reported in ClinVar and is present in gnomAD (variant ID 6‑33438828‑A‑T). Consensus from most in‑silico predictors classifies the change as benign: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic effect. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, labels the variant as Likely Benign. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign; the SGM Consensus is Likely Benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also reports a benign effect. Overall, the preponderance of evidence indicates that I529F is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.318242 | Structured | 0.019545 | Uncertain | 0.901 | 0.403 | 0.000 | 6-33438828-A-T | 4 | 2.48e-6 | -5.669 | Likely Benign | 0.235 | Likely Benign | Likely Benign | -0.21 | Likely Benign | 0.0 | 0.06 | Likely Benign | -0.08 | Likely Benign | 0.06 | Likely Benign | 0.320 | Likely Benign | -0.97 | Neutral | 0.266 | Benign | 0.054 | Benign | -1.30 | Pathogenic | 0.23 | Tolerated | 3.37 | 35 | 0.0522 | 0.2713 | 0 | 1 | -1.7 | 34.02 | ||||||||||||||||||||||||
| c.240A>C | K80N 2D  AIThe SynGAP1 K80N missense variant has no ClinVar record and is not reported in gnomAD. Functional prediction tools that agree on benign impact include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, and FATHMM, while those that predict pathogenicity are polyPhen‑2 HumDiv, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized predicting pathogenicity, whereas the SGM‑Consensus (majority vote) predicts benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Based on the overall distribution of predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.477530 | Uncertain | 0.331 | 0.873 | 0.500 | -5.072 | Likely Benign | 0.958 | Likely Pathogenic | Likely Pathogenic | 0.078 | Likely Benign | -1.07 | Neutral | 0.588 | Possibly Damaging | 0.054 | Benign | 3.89 | Benign | 0.00 | Affected | 0.3749 | 0.1237 | 1 | 0 | 0.4 | -14.07 | |||||||||||||||||||||||||||||||||||||||
| c.240A>T | K80N 2D  AIThe SynGAP1 K80N missense variant has no ClinVar record and is not listed in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, and FATHMM. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus (majority vote) remains benign; Foldetta results are unavailable. Based on the overall distribution of predictions, the variant is most likely benign, and this assessment does not contradict any ClinVar status (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.637480 | Disordered | 0.477530 | Uncertain | 0.331 | 0.873 | 0.500 | -5.072 | Likely Benign | 0.958 | Likely Pathogenic | Likely Pathogenic | 0.078 | Likely Benign | -1.07 | Neutral | 0.588 | Possibly Damaging | 0.054 | Benign | 3.89 | Benign | 0.00 | Affected | 0.3749 | 0.1237 | 1 | 0 | 0.4 | -14.07 | |||||||||||||||||||||||||||||||||||||||
| c.2612A>T | H871L 2D  AIThe SynGAP1 missense variant H871L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification—there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.480142 | Structured | 0.679301 | Binding | 0.279 | 0.858 | 0.250 | -4.562 | Likely Benign | 0.149 | Likely Benign | Likely Benign | 0.167 | Likely Benign | -2.16 | Neutral | 0.069 | Benign | 0.054 | Benign | 2.65 | Benign | 0.14 | Tolerated | 0.0953 | 0.4714 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.3178G>A | G1060S 2D  AIThe SynGAP1 missense variant G1060S is listed in ClinVar with an uncertain significance (ClinVar ID 1512003.0) and is present in gnomAD (variant ID 6‑33443730‑G‑A). All evaluated in‑silico predictors classify the change as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this benign view: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence overwhelmingly supports a benign effect, which is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.979242 | Disordered | 0.913048 | Binding | 0.407 | 0.928 | 0.875 | Uncertain | 1 | 6-33443730-G-A | -4.759 | Likely Benign | 0.082 | Likely Benign | Likely Benign | 0.376 | Likely Benign | -0.08 | Neutral | 0.271 | Benign | 0.054 | Benign | 2.69 | Benign | 0.49 | Tolerated | 4.32 | 2 | 0.2468 | 0.5311 | 1 | 0 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||||
| c.3199C>T | P1067S 2D  AIThe SynGAP1 missense variant P1067S is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta results are not available, so they do not influence the assessment. Based on the collective predictions, the variant is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.966441 | Disordered | 0.975099 | Binding | 0.459 | 0.907 | 0.875 | -4.673 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.079 | Likely Benign | -1.43 | Neutral | 0.271 | Benign | 0.054 | Benign | 2.91 | Benign | 0.48 | Tolerated | 0.3086 | 0.5753 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||||||||||||
| c.332C>A | P111Q 2D  AIThe SynGAP1 missense variant P111Q is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign status. High‑accuracy assessments further support this view: AlphaMissense‑Optimized indicates benign, SGM‑Consensus confirms likely benign, and Foldetta data are unavailable. Taken together, the preponderance of evidence points to a benign effect for P111Q, and this assessment does not conflict with the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.707965 | Disordered | 0.650020 | Binding | 0.438 | 0.858 | 0.750 | -4.726 | Likely Benign | 0.543 | Ambiguous | Likely Benign | 0.079 | Likely Benign | -2.10 | Neutral | 0.421 | Benign | 0.054 | Benign | 4.06 | Benign | 0.00 | Affected | 0.1593 | 0.4232 | 0 | -1 | -1.9 | 31.01 | |||||||||||||||||||||||||||||||||||||||
| c.335G>C | G112A 2D  AIThe SynGAP1 missense variant G112A is listed in ClinVar with an uncertain significance (ClinVar ID 1425533.0) and is present in gnomAD (6‑33432200‑G‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is also benign. Foldetta results are unavailable. Overall, the majority of computational evidence indicates that the variant is most likely benign, which is consistent with its ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.728858 | Disordered | 0.640153 | Binding | 0.332 | 0.867 | 0.750 | Uncertain | 1 | 6-33432200-G-C | 15 | 9.30e-6 | -2.456 | Likely Benign | 0.119 | Likely Benign | Likely Benign | 0.114 | Likely Benign | -2.34 | Neutral | 0.231 | Benign | 0.054 | Benign | 4.07 | Benign | 0.00 | Affected | 3.61 | 5 | 0.3817 | 0.4429 | 1 | 0 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||||
| c.527G>C | S176T 2D  AIThe SynGAP1 missense variant S176T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts Benign, and the SGM‑Consensus also indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of available predictions indicates that the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which is currently unreported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.562014 | Disordered | 0.466016 | Uncertain | 0.380 | 0.597 | 0.375 | -3.682 | Likely Benign | 0.351 | Ambiguous | Likely Benign | 0.045 | Likely Benign | -0.73 | Neutral | 0.421 | Benign | 0.054 | Benign | 4.09 | Benign | 0.19 | Tolerated | 0.1103 | 0.4979 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.56C>A | A19D 2D  AIThe SynGAP1 missense variant A19D is listed in gnomAD (ID 6‑33420320‑C‑A) but has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic outcome are polyPhen‑2 HumDiv, SIFT, and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar status (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.529623 | Disordered | 0.443393 | Uncertain | 0.378 | 0.906 | 0.500 | 6-33420320-C-A | 2 | 1.30e-6 | -3.746 | Likely Benign | 0.573 | Likely Pathogenic | Likely Benign | 0.055 | Likely Benign | -0.08 | Neutral | 0.588 | Possibly Damaging | 0.054 | Benign | 4.07 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2508 | 0.2734 | -2 | 0 | -5.3 | 44.01 | ||||||||||||||||||||||||||||||||||
| c.865A>G | M289V 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant M289V is reported in ClinVar as Benign (ClinVar ID 2122760.0) and is not present in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus all predict benign, while only FATHMM predicts pathogenic. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also indicates benign. No prediction or stability result is inconclusive. Overall, the computational evidence strongly supports a benign classification, consistent with the ClinVar status and showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.127496 | Structured | 0.403499 | Uncertain | 0.886 | 0.276 | 0.000 | Benign | 1 | -4.239 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 1.09 | Ambiguous | 0.1 | -0.27 | Likely Benign | 0.41 | Likely Benign | 0.24 | Likely Benign | 0.150 | Likely Benign | -0.36 | Neutral | 0.136 | Benign | 0.054 | Benign | 1.80 | Pathogenic | 1.00 | Tolerated | 3.38 | 23 | 0.2280 | 0.2808 | 2 | 1 | 2.3 | -32.06 | 204.2 | 51.0 | 0.0 | 0.0 | 0.2 | 0.0 | X | Potentially Benign | The hydrophobic residue Met289, located in a β hairpin linking two anti-parallel β sheet strands (res. Met289-Arg299, res. Arg272-Leu286), is swapped for another hydrophobic residue, valine. In the variant simulations, the branched hydrocarbon side chain of Val289 packs against the phenol group of the Tyr291 side chain but is unable to form methionine-aromatic interactions. β hairpins are potential nucleation sites during the initial stages of protein folding, so even minor changes in them could be significant. However, based on the simulations, the residue swap does not cause adverse effects on the structure. | ||||||||||||||||
| c.1936C>A | L646M 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L646M is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome. Predictions that are inconclusive are FoldX, Rosetta, and Foldetta. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as likely benign, while Foldetta’s stability analysis remains uncertain. Overall, the evidence overwhelmingly supports a benign classification for this variant, and this conclusion does not contradict the ClinVar status, which currently has no entry for the variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.048328 | Structured | 0.303751 | Uncertain | 0.941 | 0.344 | 0.000 | -1.911 | Likely Benign | 0.152 | Likely Benign | Likely Benign | 0.70 | Ambiguous | 0.1 | 1.66 | Ambiguous | 1.18 | Ambiguous | -1.10 | Stabilizing | 0.106 | Likely Benign | 1.86 | Neutral | 0.211 | Benign | 0.055 | Benign | 3.57 | Benign | 1.00 | Tolerated | 0.2041 | 0.3427 | 4 | 2 | -1.9 | 18.03 | |||||||||||||||||||||||||||||
| c.3266G>C | G1089A 2D  AIThe SynGAP1 missense variant G1089A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this is consistent with the lack of ClinVar annotation—there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.891961 | Disordered | 0.976771 | Binding | 0.366 | 0.890 | 1.000 | -2.864 | Likely Benign | 0.224 | Likely Benign | Likely Benign | 0.142 | Likely Benign | -1.73 | Neutral | 0.186 | Benign | 0.055 | Benign | 2.45 | Pathogenic | 0.02 | Affected | 0.3475 | 0.5331 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3271C>A | L1091I 2D  AIThe SynGAP1 missense variant L1091I is reported in gnomAD (ID 6‑33443823‑C‑A) but has no ClinVar entry. Across the available in‑silico predictors, every tool classified the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all returned benign scores. No tool predicted pathogenicity, so the pathogenic‑prediction group is empty. High‑accuracy assessments reinforce this benign view: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available for this variant. Overall, the computational evidence strongly supports a benign effect, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.924947 | Disordered | 0.984454 | Binding | 0.376 | 0.889 | 1.000 | 6-33443823-C-A | -4.304 | Likely Benign | 0.273 | Likely Benign | Likely Benign | 0.057 | Likely Benign | -0.66 | Neutral | 0.186 | Benign | 0.055 | Benign | 2.55 | Benign | 0.13 | Tolerated | 3.77 | 5 | 0.1073 | 0.3911 | 2 | 2 | 0.7 | 0.00 | ||||||||||||||||||||||||||||||||||||
| c.332C>T | P111L 2D  AIThe SynGAP1 missense variant P111L is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN and SIFT, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also yields a benign prediction (2 benign vs. 1 pathogenic votes). Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of available predictions indicates that P111L is most likely benign, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.707965 | Disordered | 0.650020 | Binding | 0.438 | 0.858 | 0.750 | -4.430 | Likely Benign | 0.486 | Ambiguous | Likely Benign | 0.089 | Likely Benign | -2.81 | Deleterious | 0.421 | Benign | 0.055 | Benign | 4.06 | Benign | 0.00 | Affected | 0.2355 | 0.7085 | -3 | -3 | 5.4 | 16.04 | ||||||||||||||||||||||||||||||||||||||||
| c.3449C>T | A1150V 2D  AIThe SynGAP1 missense variant A1150V is listed in ClinVar (ID 589625.0) with an “Uncertain” status and is present in gnomAD (variant ID 6‑33444484‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are SIFT and FATHMM. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is also benign. Foldetta, a protein‑folding stability method, did not provide a result for this variant. Overall, the majority of computational evidence indicates that A1150V is most likely benign, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.795712 | Binding | 0.371 | 0.831 | 0.625 | Uncertain | 1 | 6-33444484-C-T | 3 | 1.86e-6 | -3.648 | Likely Benign | 0.192 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -2.22 | Neutral | 0.114 | Benign | 0.055 | Benign | 2.32 | Pathogenic | 0.04 | Affected | 3.77 | 5 | 0.1242 | 0.6335 | 0 | 0 | 2.4 | 28.05 | ||||||||||||||||||||||||||||||||
| c.356A>G | E119G 2D  AIThe SynGAP1 missense variant E119G is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as likely benign; the Foldetta protein‑folding stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar status because no ClinVar claim exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.690604 | Disordered | 0.661946 | Binding | 0.346 | 0.881 | 0.750 | -4.349 | Likely Benign | 0.584 | Likely Pathogenic | Likely Benign | 0.143 | Likely Benign | -2.40 | Neutral | 0.421 | Benign | 0.055 | Benign | 3.82 | Benign | 0.01 | Affected | 0.3246 | 0.6405 | 0 | -2 | 3.1 | -72.06 | |||||||||||||||||||||||||||||||||||||||
| c.2047A>C | I683L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I683L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and ESM1b. Remaining methods (FoldX, Rosetta, Foldetta, premPS, AlphaMissense‑Default) give uncertain or inconclusive results and are treated as unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields a benign prediction, while Foldetta remains uncertain. Overall, the balance of evidence favors a benign impact for I683L, and this conclusion does not contradict any ClinVar annotation (none is available). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.200174 | Structured | 0.143268 | Uncertain | 0.848 | 0.314 | 0.000 | -9.988 | Likely Pathogenic | 0.488 | Ambiguous | Likely Benign | 0.63 | Ambiguous | 0.1 | 0.76 | Ambiguous | 0.70 | Ambiguous | 0.69 | Ambiguous | 0.286 | Likely Benign | -2.00 | Neutral | 0.011 | Benign | 0.056 | Benign | 3.43 | Benign | 0.04 | Affected | 0.0957 | 0.3185 | 2 | 2 | -0.7 | 0.00 | ||||||||||||||||||||||||||||||
| c.3705G>A | M1235I 2D  AIThe SynGAP1 missense variant M1235I is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that classify the variant as benign include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only SIFT predicts a pathogenic effect. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as “Likely Benign”; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.690604 | Disordered | 0.577958 | Binding | 0.872 | 0.532 | 0.125 | Uncertain | 1 | -4.312 | Likely Benign | 0.310 | Likely Benign | Likely Benign | 0.027 | Likely Benign | -1.44 | Neutral | 0.139 | Benign | 0.056 | Benign | 2.69 | Benign | 0.04 | Affected | 3.77 | 5 | 0.1224 | 0.2691 | 1 | 2 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||||||
| c.3705G>C | M1235I 2D  AIThe SynGAP1 missense variant M1235I is reported in gnomAD (6-33446697‑G‑C) and has no ClinVar entry. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion is not contradicted by any ClinVar status (none). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.690604 | Disordered | 0.577958 | Binding | 0.872 | 0.532 | 0.125 | 6-33446697-G-C | 1 | 6.20e-7 | -4.312 | Likely Benign | 0.310 | Likely Benign | Likely Benign | 0.027 | Likely Benign | -1.44 | Neutral | 0.139 | Benign | 0.056 | Benign | 2.69 | Benign | 0.04 | Affected | 3.77 | 5 | 0.1224 | 0.2691 | 1 | 2 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||
| c.3705G>T | M1235I 2D  AIThe SynGAP1 missense variant M1235I is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign; Foldetta results are not available. Overall, the preponderance of evidence supports a benign classification for M1235I, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.690604 | Disordered | 0.577958 | Binding | 0.872 | 0.532 | 0.125 | -4.312 | Likely Benign | 0.310 | Likely Benign | Likely Benign | 0.027 | Likely Benign | -1.44 | Neutral | 0.139 | Benign | 0.056 | Benign | 2.69 | Benign | 0.04 | Affected | 3.77 | 5 | 0.1224 | 0.2691 | 1 | 2 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||||||||
| c.500A>C | D167A 2D  AIThe SynGAP1 D167A missense variant has no ClinVar entry and is not reported in gnomAD. Functional prediction tools split evenly: benign calls come from REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM, while pathogenic calls come from PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. High‑accuracy assessment shows AlphaMissense‑Optimized as uncertain, whereas the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—labels the variant as Likely Pathogenic. Foldetta, a protein‑folding stability predictor, has no available result for this residue. Taken together, the preponderance of evidence, especially the SGM Consensus, points to a pathogenic effect. This conclusion is consistent with the absence of a ClinVar classification, so there is no contradiction with existing database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.429200 | Structured | 0.502306 | Binding | 0.377 | 0.667 | 0.375 | -13.473 | Likely Pathogenic | 0.921 | Likely Pathogenic | Ambiguous | 0.291 | Likely Benign | -3.61 | Deleterious | 0.141 | Benign | 0.056 | Benign | 3.97 | Benign | 0.00 | Affected | 0.3429 | 0.6549 | 0 | -2 | 5.3 | -44.01 | |||||||||||||||||||||||||||||||||||||||
| c.1055C>A | T352N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T352N is listed in ClinVar as Benign (ClinVar ID 590151.0) and is present in the gnomAD database (gnomAD ID 6‑33437960‑C‑A). Across the broad panel of in‑silico predictors, 13 tools (REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus) uniformly report a benign effect, whereas only FATHMM predicts pathogenicity. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is also benign. No predictions or stability analyses are missing or inconclusive. Overall, the computational evidence strongly supports a benign classification, consistent with the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.137348 | Structured | 0.367886 | Uncertain | 0.926 | 0.329 | 0.000 | Likely Benign | 1 | 6-33437960-C-A | 2 | 1.24e-6 | -4.817 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.20 | Likely Benign | 0.0 | -0.04 | Likely Benign | 0.08 | Likely Benign | 0.45 | Likely Benign | 0.027 | Likely Benign | -0.92 | Neutral | 0.255 | Benign | 0.057 | Benign | 1.75 | Pathogenic | 0.19 | Tolerated | 3.37 | 25 | 0.1311 | 0.4358 | 0 | 0 | -2.8 | 13.00 | 208.4 | -14.5 | -0.2 | 0.1 | -0.1 | 0.0 | X | Potentially Benign | Thr352 is located in a short α helical section within a loop connecting two β strands (res. Gly341-Pro349, res. Thr359-Pro364) originating from two different anti-parallel β sheets of the C2 domain. In the WT simulations, the side chain hydroxyl and backbone amide groups of Thr354 form hydrogen bonds with the backbone carbonyl group of Pro349 at the end of the preceding β strand. This arrangement likely stabilizes the α helical section and aids in folding, keeping the short secondary structure element intact in the variant simulations. However, the carboxamide group of the Asn352 side chain does not form hydrogen bonds with the backbone carbonyl group of Pro349. Instead, it packs against the cyclic ring and forms hydrogen bonds with the phenol group of the Tyr363 side chain in the other β strand. | |||||||||||||
| c.1100T>G | L367R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 L367R missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and ESM1b. Those that predict a pathogenic effect are SIFT, FATHMM, and Rosetta. Tools with uncertain or inconclusive results are FoldX, premPS, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as pathogenic. Overall, the majority of evidence points to a benign impact, with a minority of pathogenic predictions. The variant’s status is not contradicted by ClinVar, as it is not yet classified there. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | 0.370445 | Structured | 0.441805 | Uncertain | 0.790 | 0.657 | 0.250 | -6.527 | Likely Benign | 0.515 | Ambiguous | Likely Benign | 0.68 | Ambiguous | 0.4 | 6.63 | Destabilizing | 3.66 | Destabilizing | 0.70 | Ambiguous | 0.196 | Likely Benign | -0.39 | Neutral | 0.146 | Benign | 0.057 | Benign | 1.66 | Pathogenic | 0.02 | Affected | 0.1608 | 0.1173 | -3 | -2 | -8.3 | 43.03 | ||||||||||||||||||||||||||||||
| c.1985A>T | Q662L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant Q662L is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, SIFT, AlphaMissense‑Default, AlphaMissense‑Optimized, polyPhen2_HumVar, and FATHMM. Tools that predict a pathogenic effect are PROVEAN, polyPhen2_HumDiv, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign and Foldetta as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs 2 pathogenic). Overall, the majority of evidence supports a benign impact. This conclusion is consistent with the lack of ClinVar annotation and gnomAD presence, so there is no contradiction with existing database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.046336 | Structured | 0.103446 | Uncertain | 0.932 | 0.323 | 0.000 | -10.291 | Likely Pathogenic | 0.300 | Likely Benign | Likely Benign | 0.04 | Likely Benign | 0.0 | 0.12 | Likely Benign | 0.08 | Likely Benign | 0.17 | Likely Benign | 0.186 | Likely Benign | -4.09 | Deleterious | 0.699 | Possibly Damaging | 0.057 | Benign | 3.42 | Benign | 0.10 | Tolerated | 0.1070 | 0.5255 | -2 | -2 | 7.3 | -14.97 | ||||||||||||||||||||||||||||||
| c.2032A>C | S678R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S678R is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumVar, and FATHMM. Those that predict a pathogenic effect are polyPhen‑2 HumDiv, SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 pathogenic vs. 2 benign), and Foldetta predicts a benign effect. Overall, the majority of tools (8 benign vs. 4 pathogenic) support a benign classification. Thus, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.301917 | Structured | 0.123585 | Uncertain | 0.660 | 0.321 | 0.000 | -9.708 | Likely Pathogenic | 0.878 | Likely Pathogenic | Ambiguous | -0.37 | Likely Benign | 0.2 | 0.48 | Likely Benign | 0.06 | Likely Benign | 0.32 | Likely Benign | 0.106 | Likely Benign | -2.07 | Neutral | 0.454 | Possibly Damaging | 0.057 | Benign | 3.44 | Benign | 0.02 | Affected | 0.0950 | 0.3519 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||
| c.2033G>T | S678I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S678I is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, polyPhen‑2 (HumDiv and HumVar), FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑vs‑2 split. Overall, the majority of evidence (10 benign vs. 3 pathogenic) supports a benign classification. This consensus does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.301917 | Structured | 0.123585 | Uncertain | 0.660 | 0.321 | 0.000 | -9.735 | Likely Pathogenic | 0.253 | Likely Benign | Likely Benign | 0.36 | Likely Benign | 0.3 | -0.01 | Likely Benign | 0.18 | Likely Benign | -0.11 | Likely Benign | 0.119 | Likely Benign | -3.99 | Deleterious | 0.294 | Benign | 0.057 | Benign | 3.45 | Benign | 0.01 | Affected | 0.0941 | 0.5954 | -1 | -2 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||
| c.2034C>A | S678R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S678R is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SIFT, polyPhen‑2 HumDiv, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is classified as uncertain. High‑accuracy assessments show AlphaMissense‑Optimized uncertain, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 pathogenic vs. 2 benign), and Foldetta predicts a benign effect. Overall, the majority of evidence (8 benign vs. 4 pathogenic) supports a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.301917 | Structured | 0.123585 | Uncertain | 0.660 | 0.321 | 0.000 | -9.708 | Likely Pathogenic | 0.878 | Likely Pathogenic | Ambiguous | -0.37 | Likely Benign | 0.2 | 0.48 | Likely Benign | 0.06 | Likely Benign | 0.32 | Likely Benign | 0.158 | Likely Benign | -2.07 | Neutral | 0.454 | Possibly Damaging | 0.057 | Benign | 3.44 | Benign | 0.02 | Affected | 0.0950 | 0.3519 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||
| c.2034C>G | S678R 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S678R is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumVar, and FATHMM. Those that predict a pathogenic effect are polyPhen‑2 HumDiv, SIFT, ESM1b, and AlphaMissense‑Default. AlphaMissense‑Optimized is classified as uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, and Foldetta predicts a benign stability change. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict the ClinVar status, which currently has no classification for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.301917 | Structured | 0.123585 | Uncertain | 0.660 | 0.321 | 0.000 | -9.708 | Likely Pathogenic | 0.878 | Likely Pathogenic | Ambiguous | -0.37 | Likely Benign | 0.2 | 0.48 | Likely Benign | 0.06 | Likely Benign | 0.32 | Likely Benign | 0.157 | Likely Benign | -2.07 | Neutral | 0.454 | Possibly Damaging | 0.057 | Benign | 3.44 | Benign | 0.02 | Affected | 0.0950 | 0.3519 | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||
| c.3194C>A | P1065Q 2D  AIThe SynGAP1 missense variant P1065Q is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and FATHMM. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. This conclusion is consistent with the lack of ClinVar annotation, so there is no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.979741 | Disordered | 0.959518 | Binding | 0.424 | 0.917 | 0.875 | -3.928 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.026 | Likely Benign | -2.44 | Neutral | 0.102 | Benign | 0.057 | Benign | 2.00 | Pathogenic | 0.00 | Affected | 0.1487 | 0.5478 | 0 | -1 | -1.9 | 31.01 | |||||||||||||||||||||||||||||||||||||||
| c.3944G>C | W1315S 2D  AIThe SynGAP1 missense variant W1315S is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.973142 | Binding | 0.403 | 0.889 | 0.750 | 0.896 | Likely Benign | 0.279 | Likely Benign | Likely Benign | 0.118 | Likely Benign | -0.79 | Neutral | 0.115 | Benign | 0.057 | Benign | 4.26 | Benign | 0.13 | Tolerated | 0.3974 | 0.1211 | -2 | -3 | 0.1 | -99.14 | |||||||||||||||||||||||||||||||||||||||
| c.3944G>T | W1315L 2D  AIThe SynGAP1 missense variant W1315L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.885302 | Disordered | 0.973142 | Binding | 0.403 | 0.889 | 0.750 | 0.369 | Likely Benign | 0.304 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -0.65 | Neutral | 0.115 | Benign | 0.057 | Benign | 4.29 | Benign | 0.16 | Tolerated | 0.2256 | 0.3338 | -2 | -2 | 4.7 | -73.05 | |||||||||||||||||||||||||||||||||||||||
| c.4021G>C | A1341P 2D  AIThe SynGAP1 missense variant A1341P is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the preponderance of evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.823549 | Disordered | 0.980111 | Binding | 0.383 | 0.696 | 1.000 | -3.168 | Likely Benign | 0.112 | Likely Benign | Likely Benign | 0.117 | Likely Benign | -1.59 | Neutral | 0.131 | Benign | 0.057 | Benign | 4.02 | Benign | 0.02 | Affected | 0.2154 | 0.5703 | 1 | -1 | -3.4 | 26.04 | |||||||||||||||||||||||||||||||||||||||
| c.1525G>A | A509T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A509T is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are ESM1b and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive (2 benign vs 2 pathogenic). Foldetta, which integrates FoldX‑MD and Rosetta outputs, is uncertain and therefore unavailable for interpretation. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the lack of ClinVar annotation. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.025762 | Structured | 0.250110 | Uncertain | 0.923 | 0.256 | 0.000 | -8.961 | Likely Pathogenic | 0.171 | Likely Benign | Likely Benign | 0.95 | Ambiguous | 0.4 | 0.61 | Ambiguous | 0.78 | Ambiguous | 0.04 | Likely Benign | 0.360 | Likely Benign | -2.15 | Neutral | 0.031 | Benign | 0.058 | Benign | -1.25 | Pathogenic | 0.32 | Tolerated | 0.1344 | 0.5593 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||
| c.223G>C | E75Q 2D  AIThe SynGAP1 missense variant E75Q is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as Likely Benign, and AlphaMissense‑Optimized also predicts Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this assessment does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.595080 | Disordered | 0.443881 | Uncertain | 0.303 | 0.822 | 0.500 | -3.772 | Likely Benign | 0.194 | Likely Benign | Likely Benign | 0.110 | Likely Benign | -0.76 | Neutral | 0.731 | Possibly Damaging | 0.058 | Benign | 4.04 | Benign | 0.00 | Affected | 0.1461 | 0.6634 | 2 | 2 | 0.0 | -0.98 | |||||||||||||||||||||||||||||||||||||||
| c.2605C>A | L869M 2D  AIThe SynGAP1 missense variant L869M is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions is benign, and this conclusion does not contradict any ClinVar annotation (none present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.501700 | Disordered | 0.688653 | Binding | 0.272 | 0.839 | 0.250 | -4.294 | Likely Benign | 0.119 | Likely Benign | Likely Benign | 0.093 | Likely Benign | -0.09 | Neutral | 0.149 | Benign | 0.058 | Benign | 2.61 | Benign | 0.11 | Tolerated | 0.0710 | 0.3795 | 4 | 2 | -1.9 | 18.03 | |||||||||||||||||||||||||||||||||||||||
| c.266C>T | P89L 2D  AISynGAP1 missense variant P89L is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools show a split: benign calls come from REVEL, polyPhen‑2 HumVar, ESM1b, and FATHMM, while pathogenic calls are made by PROVEAN, polyPhen‑2 HumDiv, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments indicate that AlphaMissense‑Optimized predicts pathogenicity, whereas the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive due to a 2‑to‑2 split, and Foldetta results are unavailable. Overall, the majority of tools favor a pathogenic effect, but the evidence is not unanimous. Therefore, the variant is most likely pathogenic according to the current predictions, and this assessment does not contradict its ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.703578 | Disordered | 0.545797 | Binding | 0.316 | 0.865 | 0.500 | Uncertain | 2 | -6.775 | Likely Benign | 0.982 | Likely Pathogenic | Likely Pathogenic | 0.119 | Likely Benign | -3.29 | Deleterious | 0.889 | Possibly Damaging | 0.058 | Benign | 3.73 | Benign | 0.00 | Affected | 4.32 | 1 | 0.2399 | 0.5638 | -3 | -3 | 5.4 | 16.04 | ||||||||||||||||||||||||||||||||||||
| c.2869C>A | H957N 2D  AIThe SynGAP1 missense variant H957N is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. Only FATHMM predicts a pathogenic outcome. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign status. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign and the SGM‑Consensus is likely benign; Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.985964 | Disordered | 0.968874 | Binding | 0.362 | 0.915 | 0.750 | -6.804 | Likely Benign | 0.090 | Likely Benign | Likely Benign | 0.053 | Likely Benign | -0.45 | Neutral | 0.144 | Benign | 0.058 | Benign | 2.45 | Pathogenic | 0.50 | Tolerated | 0.2343 | 0.3788 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.2869C>G | H957D 2D  AIThe SynGAP1 missense variant H957D is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are ESM1b and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive (2 benign vs 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence (7 benign vs 2 pathogenic) supports a benign classification. This prediction is consistent with the lack of ClinVar annotation and gnomAD presence, indicating no contradiction with existing clinical data. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.985964 | Disordered | 0.968874 | Binding | 0.362 | 0.915 | 0.750 | -8.777 | Likely Pathogenic | 0.237 | Likely Benign | Likely Benign | 0.149 | Likely Benign | -0.77 | Neutral | 0.144 | Benign | 0.058 | Benign | 2.45 | Pathogenic | 0.44 | Tolerated | 0.2646 | 0.3066 | 1 | -1 | -0.3 | -22.05 | ||||||||||||||||||||||||||||||||||||||||
| c.2875C>A | H959N 2D  AIThe SynGAP1 missense variant H959N is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while only ESM1b predicts it as pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” consensus. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) also indicates benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence from multiple independent predictors and high‑accuracy tools indicates that H959N is most likely benign, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.985964 | Disordered | 0.980566 | Binding | 0.333 | 0.905 | 0.750 | -8.811 | Likely Pathogenic | 0.104 | Likely Benign | Likely Benign | 0.115 | Likely Benign | -0.10 | Neutral | 0.144 | Benign | 0.058 | Benign | 4.15 | Benign | 0.21 | Tolerated | 0.2298 | 0.3838 | 2 | 1 | -0.3 | -23.04 | |||||||||||||||||||||||||||||||||||||||
| c.2875C>G | H959D 2D  AIThe SynGAP1 missense variant H959D is listed in gnomAD (ID 6‑33443427‑C‑G) but has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, resolves to “Likely Benign” (three benign votes versus one pathogenic). High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar status (none reported). Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.985964 | Disordered | 0.980566 | Binding | 0.333 | 0.905 | 0.750 | 6-33443427-C-G | 1 | 6.20e-7 | -12.060 | Likely Pathogenic | 0.235 | Likely Benign | Likely Benign | 0.176 | Likely Benign | -0.73 | Neutral | 0.144 | Benign | 0.058 | Benign | 4.14 | Benign | 0.29 | Tolerated | 3.77 | 5 | 0.2586 | 0.3066 | -1 | 1 | -0.3 | -22.05 | ||||||||||||||||||||||||||||||||||
| c.2909A>G | E970G 2D  AIThe SynGAP1 missense variant E970G is listed in ClinVar (ID 2013677.0) as Benign and is not reported in gnomAD. All available in‑silico predictors classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts a benign effect, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant. Overall, the computational evidence overwhelmingly supports a benign classification, which is consistent with the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.951925 | Disordered | 0.953422 | Binding | 0.342 | 0.902 | 0.750 | Benign | 1 | -0.167 | Likely Benign | 0.139 | Likely Benign | Likely Benign | 0.139 | Likely Benign | -0.93 | Neutral | 0.144 | Benign | 0.058 | Benign | 4.09 | Benign | 0.10 | Tolerated | 4.32 | 2 | 0.2701 | 0.5833 | 0 | -2 | 3.1 | -72.06 | |||||||||||||||||||||||||||||||||||
| c.3140C>T | S1047L 2D  AIThe SynGAP1 missense variant S1047L is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443692‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only SIFT predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized scores benign, and the SGM‑Consensus (derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates benign. Foldetta stability analysis is not available for this variant. Overall, the preponderance of computational evidence points to a benign effect, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.963420 | Disordered | 0.933764 | Binding | 0.409 | 0.909 | 0.750 | Uncertain | 1 | 6-33443692-C-T | 1 | 6.20e-7 | -4.062 | Likely Benign | 0.132 | Likely Benign | Likely Benign | 0.032 | Likely Benign | -0.63 | Neutral | 0.144 | Benign | 0.058 | Benign | 2.60 | Benign | 0.02 | Affected | 3.77 | 5 | 0.1575 | 0.5555 | -2 | -3 | 4.6 | 26.08 | ||||||||||||||||||||||||||||||||
| c.3188G>A | G1063D 2D  AIThe SynGAP1 missense variant G1063D is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign classification. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign. Foldetta results are unavailable, so they do not influence the assessment. Overall, the computational evidence overwhelmingly supports a benign impact for G1063D, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.975134 | Disordered | 0.945784 | Binding | 0.394 | 0.913 | 0.875 | -6.950 | Likely Benign | 0.367 | Ambiguous | Likely Benign | 0.057 | Likely Benign | -0.30 | Neutral | 0.411 | Benign | 0.058 | Benign | 4.24 | Benign | 0.08 | Tolerated | 0.1878 | 0.3026 | 1 | -1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||||||||||||
| c.3244C>A | Q1082K 2D  AIThe SynGAP1 missense variant Q1082K is not reported in ClinVar and is absent from gnomAD. Consensus predictions from multiple in silico tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM all score benign, and AlphaMissense‑Optimized also predicts benign. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. No tools predict pathogenicity, and AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, SGM‑Consensus is Likely Benign, while Foldetta results are unavailable. Consequently, the variant is most likely benign, and this assessment does not contradict any ClinVar annotation, as none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.819762 | Disordered | 0.979325 | Binding | 0.347 | 0.896 | 0.875 | -4.488 | Likely Benign | 0.460 | Ambiguous | Likely Benign | 0.087 | Likely Benign | -1.13 | Neutral | 0.224 | Benign | 0.058 | Benign | 4.19 | Benign | 0.12 | Tolerated | 0.2008 | 0.4972 | 1 | 1 | -0.4 | 0.04 | |||||||||||||||||||||||||||||||||||||||
| c.3245A>G | Q1082R 2D  AIThe SynGAP1 missense variant Q1082R is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Benign. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of available predictions indicates that the variant is most likely benign, and this conclusion is not contradicted by any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.819762 | Disordered | 0.979325 | Binding | 0.347 | 0.896 | 0.875 | -3.584 | Likely Benign | 0.418 | Ambiguous | Likely Benign | 0.076 | Likely Benign | -0.96 | Neutral | 0.224 | Benign | 0.058 | Benign | 4.14 | Benign | 0.10 | Tolerated | 0.1590 | 0.3014 | 1 | 1 | -1.0 | 28.06 | |||||||||||||||||||||||||||||||||||||||
| c.3245A>T | Q1082L 2D  AIThe SynGAP1 missense variant Q1082L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are unavailable. Overall, the variant is most likely benign, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.819762 | Disordered | 0.979325 | Binding | 0.347 | 0.896 | 0.875 | -3.284 | Likely Benign | 0.171 | Likely Benign | Likely Benign | 0.097 | Likely Benign | -1.30 | Neutral | 0.224 | Benign | 0.058 | Benign | 4.12 | Benign | 1.00 | Tolerated | 0.0979 | 0.6824 | -2 | -2 | 7.3 | -14.97 | |||||||||||||||||||||||||||||||||||||||
| c.3341G>A | S1114N 2D  AIThe SynGAP1 missense variant S1114N is not reported in ClinVar and is absent from gnomAD, indicating no documented allele frequency data. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool predicts pathogenicity. The high‑accuracy consensus, SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN), also reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.908098 | Disordered | 0.895196 | Binding | 0.295 | 0.908 | 0.875 | -6.089 | Likely Benign | 0.106 | Likely Benign | Likely Benign | 0.047 | Likely Benign | -0.51 | Neutral | 0.071 | Benign | 0.058 | Benign | 2.71 | Benign | 0.06 | Tolerated | 0.1416 | 0.4610 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||||||||||||||
| c.517C>G | L173V 2D  AIThe SynGAP1 missense variant L173V is not reported in ClinVar (ClinVar ID: None) and is absent from gnomAD (gnomAD ID: None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized. Only ESM1b predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized is benign, and the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a benign majority vote. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.534167 | Disordered | 0.491566 | Uncertain | 0.390 | 0.631 | 0.375 | -8.320 | Likely Pathogenic | 0.495 | Ambiguous | Likely Benign | 0.100 | Likely Benign | -0.95 | Neutral | 0.131 | Benign | 0.058 | Benign | 4.05 | Benign | 0.22 | Tolerated | 0.1343 | 0.3217 | 2 | 1 | 0.4 | -14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.529T>A | F177I 2D  AIThe SynGAP1 missense variant F177I is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Tools that predict a pathogenic effect are ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 vs 2), and Foldetta results are unavailable. Overall, the majority of predictions (six benign vs. three pathogenic) suggest a benign impact, though the presence of pathogenic signals from high‑accuracy tools introduces uncertainty. The variant is most likely benign based on the current evidence, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.505461 | Disordered | 0.461817 | Uncertain | 0.357 | 0.598 | 0.500 | -10.208 | Likely Pathogenic | 0.980 | Likely Pathogenic | Likely Pathogenic | 0.148 | Likely Benign | -0.60 | Neutral | 0.131 | Benign | 0.058 | Benign | 4.18 | Benign | 0.11 | Tolerated | 0.2282 | 0.3299 | 1 | 0 | 1.7 | -34.02 | ||||||||||||||||||||||||||||||||||||||||
| c.548A>T | H183L 2D  AIThe SynGAP1 missense variant H183L is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. In contrast, tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The high‑accuracy consensus, SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a “Likely Pathogenic” verdict (3 pathogenic vs. 1 benign votes). AlphaMissense‑Optimized alone also predicts pathogenic. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a pathogenic impact, and this assessment does not contradict any ClinVar status because no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.476583 | Structured | 0.432952 | Uncertain | 0.421 | 0.622 | 0.500 | -12.898 | Likely Pathogenic | 0.968 | Likely Pathogenic | Likely Pathogenic | 0.325 | Likely Benign | -8.12 | Deleterious | 0.421 | Benign | 0.058 | Benign | 3.79 | Benign | 0.01 | Affected | 0.0911 | 0.5304 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.571A>G | S191G 2D  AIThe SynGAP1 missense variant S191G is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while ESM1b and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also leans toward benign (two benign versus two uncertain). Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence indicates that S191G is most likely benign, and this conclusion does not contradict the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.429200 | Structured | 0.428475 | Uncertain | 0.322 | 0.615 | 0.125 | -7.161 | In-Between | 0.506 | Ambiguous | Likely Benign | 0.173 | Likely Benign | -2.43 | Neutral | 0.131 | Benign | 0.058 | Benign | 3.77 | Benign | 0.03 | Affected | 0.2893 | 0.5335 | 1 | 0 | 0.4 | -30.03 | ||||||||||||||||||||||||||||||||||||||||
| c.661G>A | E221K 2D  AIThe SynGAP1 E221K missense variant is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include Rosetta, premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Those that predict a pathogenic effect are REVEL, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus. FoldX and Foldetta give uncertain results. High‑accuracy methods show AlphaMissense‑Optimized as pathogenic; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also predicts pathogenic (3 pathogenic vs 1 benign). Foldetta remains uncertain. Overall, the majority of reliable predictors indicate a pathogenic impact. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.127496 | Structured | 0.413334 | Uncertain | 0.891 | 0.283 | 0.000 | -12.331 | Likely Pathogenic | 0.997 | Likely Pathogenic | Likely Pathogenic | -1.01 | Ambiguous | 0.4 | -0.18 | Likely Benign | -0.60 | Ambiguous | 0.19 | Likely Benign | 0.815 | Likely Pathogenic | -2.92 | Deleterious | 0.131 | Benign | 0.058 | Benign | 5.92 | Benign | 0.02 | Affected | 0.2491 | 0.8002 | 0 | 1 | -0.4 | -0.94 | |||||||||||||||||||||||||||||
| c.667A>G | T223A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T223A is listed in ClinVar with an “Uncertain” status and is present in the gnomAD database (ID 6‑33435518‑A‑G). Functional prediction tools that agree on a benign effect include FoldX, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are REVEL and PROVEAN. Predictions that are inconclusive are Rosetta, Foldetta, premPS, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also resolves to benign; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, remains uncertain. Overall, the majority of evidence points to a benign impact, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.070400 | Structured | 0.382605 | Uncertain | 0.867 | 0.316 | 0.125 | Uncertain | 1 | 6-33435518-A-G | 3 | 1.86e-6 | -7.076 | In-Between | 0.316 | Likely Benign | Likely Benign | 0.30 | Likely Benign | 0.1 | 0.77 | Ambiguous | 0.54 | Ambiguous | 0.74 | Ambiguous | 0.574 | Likely Pathogenic | -3.36 | Deleterious | 0.231 | Benign | 0.058 | Benign | 5.74 | Benign | 0.09 | Tolerated | 3.41 | 13 | 0.2982 | 0.3031 | 1 | 0 | 2.5 | -30.03 | 186.4 | 44.0 | 0.0 | 0.0 | 0.0 | 0.0 | X | X | Uncertain | The introduced residue Ala223 is located on the outer surface of an anti-parallel β sheet strand (res. Cys219-Thr224). Unlike the hydroxyl group of the Thr223 side chain in the WT protein, the methyl side chain of Ala223 cannot form hydrogen bonds with nearby residues Thr228 and Lys207. Without these hydrogen-bonding interactions at the β sheet surface, the secondary structure element becomes unstable and partially unfolds in the variant simulations. However, since the model ends abruptly at the N-terminus, no definite conclusions can be drawn from the simulations. | |||||||||||||
| c.667A>T | T223S 2D  3DClick to see structure in 3D Viewer AISynGAP1 T223S is listed in ClinVar as a variant of uncertain significance and is present in the gnomAD database (ID 6‑33435518‑A‑T). Functional prediction tools that reach consensus classify the variant as benign: FoldX, Foldetta, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, FATHMM, and AlphaMissense‑Optimized. Tools that predict pathogenicity include REVEL, PROVEAN, and SIFT. Predictions that are inconclusive or uncertain are Rosetta, premPS, AlphaMissense‑Default, and ESM1b. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, Foldetta is benign, while the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive due to a 1‑to‑1 split between benign and pathogenic calls. Overall, the majority of evidence points to a benign effect, which does not contradict the ClinVar designation of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | 0.070400 | Structured | 0.382605 | Uncertain | 0.867 | 0.316 | 0.125 | Conflicting | 2 | 6-33435518-A-T | 3 | 1.86e-6 | -7.714 | In-Between | 0.410 | Ambiguous | Likely Benign | 0.26 | Likely Benign | 0.1 | 0.50 | Ambiguous | 0.38 | Likely Benign | 0.62 | Ambiguous | 0.535 | Likely Pathogenic | -2.86 | Deleterious | 0.421 | Benign | 0.058 | Benign | 5.80 | Benign | 0.02 | Affected | 3.41 | 13 | 0.2388 | 0.2972 | 1 | 1 | -0.1 | -14.03 | 200.7 | 17.3 | -0.2 | 0.2 | 0.0 | 0.0 | X | Uncertain | The introduced residue Ser223 is located on the outer surface of an anti-parallel β sheet strand (res. Cys219-Thr224). Its hydroxyl group forms hydrogen bonds with nearby residues Thr228 and Lys207 in the variant simulations, similar to the hydroxyl group of Thr223 in the WT simulations. These hydrogen-bonding interactions at the β sheet surface contribute to the stability of the secondary structure element and may prevent it from unfolding. However, since the model ends abruptly at the N-terminus, no definite conclusions can be drawn from the simulations. | ||||||||||||||
| c.908G>C | G303A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant G303A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from FoldX, Rosetta, and Foldetta, which are treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also predicts benign, while Foldetta’s stability analysis is uncertain. Overall, the variant is most likely benign based on the consensus of predictive tools, and this assessment does not contradict the ClinVar status, which currently has no entry for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.450668 | Structured | 0.271087 | Uncertain | 0.630 | 0.254 | 0.250 | -1.965 | Likely Benign | 0.105 | Likely Benign | Likely Benign | 1.66 | Ambiguous | 0.2 | -0.51 | Ambiguous | 0.58 | Ambiguous | 0.10 | Likely Benign | 0.034 | Likely Benign | -0.90 | Neutral | 0.112 | Benign | 0.058 | Benign | 4.07 | Benign | 0.35 | Tolerated | 0.3724 | 0.5325 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||
| c.1258T>A | F420I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F420I is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a deleterious effect: benign calls come from REVEL, polyPhen‑2 HumVar, and FATHMM, while pathogenic calls are made by FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized predicts pathogenicity, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is labeled Likely Pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) also predicts pathogenicity. No prediction or folding stability result is missing or inconclusive. Based on the overwhelming agreement among both general and high‑accuracy tools, the variant is most likely pathogenic, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | 0.104810 | Structured | 0.384475 | Uncertain | 0.974 | 0.255 | 0.000 | -12.567 | Likely Pathogenic | 0.995 | Likely Pathogenic | Likely Pathogenic | 2.88 | Destabilizing | 0.0 | 3.64 | Destabilizing | 3.26 | Destabilizing | 1.25 | Destabilizing | 0.330 | Likely Benign | -5.46 | Deleterious | 0.575 | Possibly Damaging | 0.059 | Benign | 3.22 | Benign | 0.02 | Affected | 0.1864 | 0.2113 | 1 | 0 | 1.7 | -34.02 | |||||||||||||||||||||||||||||
| c.3440C>T | T1147I 2D  AIThe SynGAP1 missense variant T1147I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status because no ClinVar claim exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.831250 | Disordered | 0.746520 | Binding | 0.345 | 0.839 | 0.875 | -3.552 | Likely Benign | 0.151 | Likely Benign | Likely Benign | 0.423 | Likely Benign | -2.03 | Neutral | 0.259 | Benign | 0.059 | Benign | 5.42 | Benign | 0.01 | Affected | 0.1029 | 0.4968 | 0 | -1 | 5.2 | 12.05 | |||||||||||||||||||||||||||||||||||||||
| c.3902C>G | P1301R 2D  AIThe SynGAP1 missense variant P1301R is listed in ClinVar with an uncertain significance (ClinVar ID 2092739.0) and is present in gnomAD (ID 6‑33451776‑C‑G). Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign effects. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are unavailable. In summary, all available predictions agree on a benign impact, and this consensus does not contradict the ClinVar uncertain status. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.728858 | Disordered | 0.885064 | Binding | 0.447 | 0.841 | 0.875 | Uncertain | 1 | 6-33451776-C-G | 15 | 9.30e-6 | -4.753 | Likely Benign | 0.162 | Likely Benign | Likely Benign | 0.076 | Likely Benign | -1.13 | Neutral | 0.077 | Benign | 0.059 | Benign | 2.81 | Benign | 0.10 | Tolerated | 3.77 | 5 | 0.1352 | 0.1929 | 0 | -2 | -2.9 | 59.07 | ||||||||||||||||||||||||||||||||
| c.212A>T | D71V 2D  AIThe SynGAP1 D71V missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that indicate a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are SIFT and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, resolves to a likely benign classification (3 benign vs. 1 pathogenic votes). High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this assessment does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.575842 | Disordered | 0.456046 | Uncertain | 0.350 | 0.799 | 0.375 | -3.549 | Likely Benign | 0.780 | Likely Pathogenic | Likely Benign | 0.183 | Likely Benign | -2.28 | Neutral | 0.334 | Benign | 0.060 | Benign | 4.01 | Benign | 0.00 | Affected | 0.0771 | 0.6197 | -2 | -3 | 7.7 | -15.96 | |||||||||||||||||||||||||||||||||||||||
| c.898T>G | S300A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S300A is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. Rosetta and Foldetta give uncertain results and are therefore not considered evidence for either side. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is also benign (3 benign vs. 1 pathogenic). Foldetta remains uncertain. Overall, the preponderance of evidence indicates that the variant is most likely benign, and this conclusion does not contradict the absence of a ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.356642 | Structured | 0.256848 | Uncertain | 0.742 | 0.280 | 0.375 | -5.420 | Likely Benign | 0.060 | Likely Benign | Likely Benign | 0.08 | Likely Benign | 0.3 | 1.07 | Ambiguous | 0.58 | Ambiguous | 0.08 | Likely Benign | 0.031 | Likely Benign | -1.18 | Neutral | 0.139 | Benign | 0.060 | Benign | 1.56 | Pathogenic | 0.20 | Tolerated | 0.5135 | 0.4949 | Weaken | 1 | 1 | 2.6 | -16.00 | ||||||||||||||||||||||||||||
| c.991T>G | S331A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S331A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only FATHMM predicts pathogenic. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a benign majority vote (3 benign vs. 1 pathogenic). High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote) is benign; and Foldetta, combining FoldX‑MD and Rosetta outputs, is benign. No prediction or folding‑stability result is missing or inconclusive. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | 0.433034 | Structured | 0.346458 | Uncertain | 0.658 | 0.475 | 0.250 | -2.199 | Likely Benign | 0.101 | Likely Benign | Likely Benign | 0.22 | Likely Benign | 0.1 | -0.08 | Likely Benign | 0.07 | Likely Benign | -0.15 | Likely Benign | 0.071 | Likely Benign | -0.44 | Neutral | 0.139 | Benign | 0.060 | Benign | 1.89 | Pathogenic | 0.67 | Tolerated | 0.5314 | 0.3008 | Weaken | 1 | 1 | 2.6 | -16.00 | ||||||||||||||||||||||||||||
| c.2698A>G | T900A 2D  AIThe SynGAP1 missense variant T900A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta results are unavailable. Overall, the consensus of all available predictions points to a benign impact, and this conclusion is consistent with the lack of a ClinVar classification, so there is no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.675549 | Disordered | 0.465347 | Uncertain | 0.289 | 0.924 | 0.375 | -2.289 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.028 | Likely Benign | -0.49 | Neutral | 0.059 | Benign | 0.061 | Benign | 2.73 | Benign | 0.40 | Tolerated | 0.4018 | 0.4059 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||||||||||||||
| c.2824C>G | P942A 2D  AIThe SynGAP1 missense variant P942A is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.919029 | Disordered | 0.878102 | Binding | 0.365 | 0.915 | 0.625 | -4.404 | Likely Benign | 0.047 | Likely Benign | Likely Benign | 0.023 | Likely Benign | -0.90 | Neutral | 0.059 | Benign | 0.061 | Benign | 2.52 | Benign | 0.00 | Affected | 0.3362 | 0.4621 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||||||
| c.2849G>C | G950A 2D  AIThe SynGAP1 missense variant G950A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. Only FATHMM predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a “Likely Benign” verdict, reflecting the majority of benign calls. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (majority vote) is benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of available predictions indicates that the variant is most likely benign, and this assessment does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.987317 | Disordered | 0.888649 | Binding | 0.368 | 0.923 | 0.750 | -6.557 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.339 | Likely Benign | -0.13 | Neutral | 0.059 | Benign | 0.061 | Benign | 2.28 | Pathogenic | 0.08 | Tolerated | 0.3343 | 0.4957 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2857C>G | P953A 2D  AIThe SynGAP1 missense variant P953A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools that assess functional impact all converge on a benign outcome: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. No tool in the dataset indicates pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available, so they do not alter the overall assessment. Consequently, the variant is most likely benign based on the collective predictions, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.983019 | Disordered | 0.920633 | Binding | 0.403 | 0.926 | 0.750 | -4.879 | Likely Benign | 0.059 | Likely Benign | Likely Benign | 0.082 | Likely Benign | -0.98 | Neutral | 0.124 | Benign | 0.061 | Benign | 2.81 | Benign | 0.41 | Tolerated | 0.3476 | 0.5203 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||||||
| c.2923A>T | T975S 2D  AIThe SynGAP1 missense variant T975S is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity. High‑accuracy assessments corroborate this view: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, is not available for this variant. Overall, the predictions strongly suggest that T975S is most likely benign, and this conclusion is consistent with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.871313 | Disordered | 0.969331 | Binding | 0.332 | 0.890 | 0.625 | -2.743 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.127 | Likely Benign | -0.57 | Neutral | 0.059 | Benign | 0.061 | Benign | 4.16 | Benign | 0.20 | Tolerated | 0.3228 | 0.4366 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.2924C>G | T975S 2D  AIThe SynGAP1 missense variant T975S is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized scores the variant as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the available predictions strongly suggest that T975S is most likely benign, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.871313 | Disordered | 0.969331 | Binding | 0.332 | 0.890 | 0.625 | Uncertain | 1 | -2.743 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.109 | Likely Benign | -0.57 | Neutral | 0.059 | Benign | 0.061 | Benign | 4.16 | Benign | 0.20 | Tolerated | 0.3228 | 0.4366 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||||||||||
| c.3077A>C | D1026A 2D  AIThe SynGAP1 D1026A variant is not reported in ClinVar (ClinVar status: not listed) and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, and FATHMM, while those that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments are inconclusive: AlphaMissense‑Optimized is uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is a tie, and Foldetta results are not available. Overall, the majority of standard tools favor a benign interpretation, and no ClinVar entry contradicts this assessment. Thus, the variant is most likely benign based on current predictions, with no conflict with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.894241 | Disordered | 0.993931 | Binding | 0.324 | 0.739 | 0.500 | -4.211 | Likely Benign | 0.849 | Likely Pathogenic | Ambiguous | 0.070 | Likely Benign | -2.69 | Deleterious | 0.112 | Benign | 0.061 | Benign | 2.53 | Benign | 0.02 | Affected | 0.3392 | 0.5279 | 0 | -2 | 5.3 | -44.01 | ||||||||||||||||||||||||||||||||||||||||
| c.3100C>G | P1034A 2D  AIThe SynGAP1 missense variant P1034A is listed in ClinVar as Benign (ClinVar ID 1901716.0) and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only FATHMM predicts a pathogenic outcome, representing the sole discordant signal. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the consensus of the majority of tools, including the high‑accuracy methods, indicates a benign impact. This prediction aligns with the ClinVar status, with no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.926919 | Disordered | 0.991713 | Binding | 0.343 | 0.752 | 0.625 | Benign | 1 | -4.174 | Likely Benign | 0.178 | Likely Benign | Likely Benign | 0.060 | Likely Benign | -2.44 | Neutral | 0.059 | Benign | 0.061 | Benign | 2.47 | Pathogenic | 0.06 | Tolerated | 3.77 | 5 | 0.3028 | 0.5622 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||
| c.3130C>G | P1044A 2D  AIThe SynGAP1 missense variant P1044A is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the evidence strongly supports a benign classification, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.979741 | Disordered | 0.952126 | Binding | 0.331 | 0.855 | 0.750 | -3.957 | Likely Benign | 0.059 | Likely Benign | Likely Benign | 0.359 | Likely Benign | -1.07 | Neutral | 0.059 | Benign | 0.061 | Benign | 5.50 | Benign | 0.18 | Tolerated | 0.3194 | 0.5850 | 1 | -1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||||||
| c.3377G>C | G1126A 2D  AIThe SynGAP1 missense variant G1126A is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.894241 | Disordered | 0.837209 | Binding | 0.345 | 0.918 | 0.875 | -6.402 | Likely Benign | 0.080 | Likely Benign | Likely Benign | 0.228 | Likely Benign | -0.63 | Neutral | 0.124 | Benign | 0.061 | Benign | 4.83 | Benign | 0.56 | Tolerated | 0.3505 | 0.5333 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.3757G>A | A1253T 2D  AISynGAP1 missense variant A1253T is predicted to be benign by all evaluated in‑silico tools. The consensus group of predictors—REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—uniformly classify the change as benign, with no tool indicating pathogenicity. High‑accuracy methods corroborate this view: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus (a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also labels the variant as likely benign. Foldetta stability analysis is unavailable. ClinVar contains no entry for this variant, and it is not present in gnomAD. Overall, the predictive evidence strongly supports a benign classification, consistent with the lack of a ClinVar pathogenic report. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.661982 | Disordered | 0.391377 | Uncertain | 0.881 | 0.550 | 0.750 | -4.116 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.046 | Likely Benign | -0.08 | Neutral | 0.042 | Benign | 0.061 | Benign | 2.79 | Benign | 0.41 | Tolerated | 0.1016 | 0.5339 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||||||||||
| c.1895A>C | N632T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N632T is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include Foldetta, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are REVEL, PROVEAN, and FATHMM. The remaining tools—FoldX, Rosetta, and AlphaMissense‑Default—return uncertain results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic. Overall, the majority of predictions lean toward a benign impact, and this does not contradict any ClinVar annotation (none is available). Thus, based on the current computational evidence, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.042364 | Structured | 0.041437 | Uncertain | 0.938 | 0.254 | 0.000 | -6.797 | Likely Benign | 0.371 | Ambiguous | Likely Benign | 1.14 | Ambiguous | 0.2 | -0.58 | Ambiguous | 0.28 | Likely Benign | 0.39 | Likely Benign | 0.541 | Likely Pathogenic | -4.48 | Deleterious | 0.214 | Benign | 0.062 | Benign | -1.49 | Pathogenic | 0.13 | Tolerated | 0.1295 | 0.6809 | 0 | 0 | 2.8 | -13.00 | ||||||||||||||||||||||||||||||
| c.2252C>T | P751L 2D  AIThe SynGAP1 missense variant P751L is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present among the evaluated tools. High‑accuracy assessments confirm this trend: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.618285 | Disordered | 0.667683 | Binding | 0.386 | 0.866 | 0.625 | -3.558 | Likely Benign | 0.143 | Likely Benign | Likely Benign | 0.207 | Likely Benign | -1.62 | Neutral | 0.316 | Benign | 0.062 | Benign | 2.94 | Benign | 0.24 | Tolerated | 0.2318 | 0.6451 | -3 | -3 | 5.4 | 16.04 | |||||||||||||||||||||||||||||||||||||||
| c.2822C>G | P941R 2D  AIThe SynGAP1 missense variant P941R is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the only pathogenic prediction comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates a benign outcome. Foldetta results are not available, so they do not influence the assessment. Overall, the majority of evidence points to a benign impact for P941R, and this conclusion is consistent with the lack of ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.874069 | Disordered | 0.900790 | Binding | 0.403 | 0.906 | 0.625 | -5.463 | Likely Benign | 0.181 | Likely Benign | Likely Benign | 0.056 | Likely Benign | -0.34 | Neutral | 0.144 | Benign | 0.062 | Benign | 2.75 | Benign | 0.01 | Affected | 0.1487 | 0.3775 | 0 | -2 | -2.9 | 59.07 | |||||||||||||||||||||||||||||||||||||||
| c.2827G>C | G943R 2D  AIThe SynGAP1 missense variant G943R is reported in gnomAD (variant ID 6-33443379‑G‑C) but has no ClinVar entry. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. No tool predicts pathogenicity; only ESM1b is uncertain. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports the variant as likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of available predictions strongly supports a benign classification, and this is consistent with the absence of a ClinVar pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.973328 | Disordered | 0.860437 | Binding | 0.372 | 0.910 | 0.750 | 6-33443379-G-C | 1 | 6.20e-7 | -7.159 | In-Between | 0.335 | Likely Benign | Likely Benign | 0.308 | Likely Benign | 1.00 | Neutral | 0.411 | Benign | 0.062 | Benign | 2.86 | Benign | 0.94 | Tolerated | 4.32 | 4 | 0.0976 | 0.4933 | -2 | -3 | -4.1 | 99.14 | ||||||||||||||||||||||||||||||||||
| c.2828G>T | G943V 2D  AIThe SynGAP1 missense variant G943V is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. The only tool with an uncertain outcome is ESM1b, which does not alter the overall consensus. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign classification. High‑accuracy predictors corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates likely benign; Foldetta results are not available. Taken together, the evidence overwhelmingly supports a benign interpretation, and there is no conflict with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.973328 | Disordered | 0.860437 | Binding | 0.372 | 0.910 | 0.750 | -7.658 | In-Between | 0.092 | Likely Benign | Likely Benign | 0.265 | Likely Benign | -0.43 | Neutral | 0.224 | Benign | 0.062 | Benign | 2.85 | Benign | 0.16 | Tolerated | 0.1399 | 0.3507 | -1 | -3 | 4.6 | 42.08 | |||||||||||||||||||||||||||||||||||||||
| c.286G>C | G96R 2D  AIThe SynGAP1 missense variant G96R is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and SIFT. AlphaMissense‑Default is uncertain, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates a likely benign outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact for G96R, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.657645 | Disordered | 0.599491 | Binding | 0.335 | 0.871 | 0.625 | -3.872 | Likely Benign | 0.349 | Ambiguous | Likely Benign | 0.059 | Likely Benign | -0.91 | Neutral | 0.687 | Possibly Damaging | 0.062 | Benign | 4.19 | Benign | 0.00 | Affected | 0.1300 | 0.5031 | -3 | -2 | -4.1 | 99.14 | |||||||||||||||||||||||||||||||||||||||
| c.2897A>T | H966L 2D  AIThe SynGAP1 missense variant H966L is not reported in ClinVar and is absent from gnomAD. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. No pathogenic predictions are present among the evaluated algorithms. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) reports likely benign. Foldetta results are not available for this variant. Based on the collective evidence, the variant is most likely benign, and this conclusion is consistent with the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.976962 | Disordered | 0.974672 | Binding | 0.378 | 0.879 | 0.750 | -6.119 | Likely Benign | 0.095 | Likely Benign | Likely Benign | 0.183 | Likely Benign | -1.74 | Neutral | 0.174 | Benign | 0.062 | Benign | 4.05 | Benign | 0.35 | Tolerated | 0.1514 | 0.5316 | -2 | -3 | 7.0 | -23.98 | |||||||||||||||||||||||||||||||||||||||
| c.3106C>G | Q1036E 2D  AIThe SynGAP1 missense variant Q1036E is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. Only SIFT predicts a pathogenic outcome. When predictions are grouped by consensus, the benign group contains nine tools, while the pathogenic group contains one (SIFT). High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign.” Foldetta results are unavailable, so they do not influence the assessment. Overall, the evidence strongly suggests the variant is most likely benign, and this conclusion is not contradicted by any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.948786 | Disordered | 0.987955 | Binding | 0.275 | 0.765 | 0.625 | -3.797 | Likely Benign | 0.313 | Likely Benign | Likely Benign | 0.056 | Likely Benign | -1.22 | Neutral | 0.264 | Benign | 0.062 | Benign | 2.59 | Benign | 0.03 | Affected | 0.1629 | 0.3484 | 2 | 2 | 0.0 | 0.98 | |||||||||||||||||||||||||||||||||||||||
| c.3444G>A | M1148I 2D  AIThe SynGAP1 missense variant M1148I is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact. There is no ClinVar annotation to contradict this conclusion, so the variant is most likely benign based on current predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.774279 | Binding | 0.343 | 0.835 | 0.875 | -3.017 | Likely Benign | 0.348 | Ambiguous | Likely Benign | 0.021 | Likely Benign | -1.55 | Neutral | 0.144 | Benign | 0.062 | Benign | 2.57 | Benign | 0.00 | Affected | 0.1453 | 0.3041 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3444G>C | M1148I 2D  AIThe SynGAP1 missense variant M1148I has no ClinVar entry and is not reported in gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and AlphaMissense‑Optimized all indicate a benign or likely benign outcome. Only SIFT predicts a pathogenic effect, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign, the SGM‑Consensus also reports likely benign, and Foldetta data are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar classification exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.774279 | Binding | 0.343 | 0.835 | 0.875 | -3.017 | Likely Benign | 0.348 | Ambiguous | Likely Benign | 0.021 | Likely Benign | -1.55 | Neutral | 0.144 | Benign | 0.062 | Benign | 2.57 | Benign | 0.00 | Affected | 0.1453 | 0.3041 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.3444G>T | M1148I 2D  AIThe SynGAP1 missense variant M1148I has no ClinVar entry and is not reported in gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and AlphaMissense‑Optimized all indicate a benign or likely benign outcome. Only SIFT predicts a pathogenic effect, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign, the SGM‑Consensus also reports likely benign, and Foldetta data are unavailable. Based on the preponderance of evidence, the variant is most likely benign, and this conclusion does not contradict any ClinVar status because no ClinVar classification exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.762850 | Disordered | 0.774279 | Binding | 0.343 | 0.835 | 0.875 | -3.017 | Likely Benign | 0.348 | Ambiguous | Likely Benign | 0.021 | Likely Benign | -1.55 | Neutral | 0.144 | Benign | 0.062 | Benign | 2.57 | Benign | 0.00 | Affected | 0.1453 | 0.3041 | 2 | 1 | 2.6 | -18.03 | |||||||||||||||||||||||||||||||||||||||
| c.445A>G | K149E 2D  AIThe SynGAP1 missense variant K149E is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Tools that predict a pathogenic effect are SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. The SGM Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields an equal split (2 vs 2) and is therefore considered unavailable. AlphaMissense‑Optimized, a high‑accuracy predictor, classifies the variant as pathogenic, while Foldetta (combining FoldX‑MD and Rosetta) has no available result for this variant. Overall, the majority of standard tools (five benign vs. four pathogenic) lean toward a benign interpretation, and this is consistent with the lack of ClinVar evidence. Thus, the variant is most likely benign, with no contradiction to ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.562014 | Disordered | 0.501681 | Binding | 0.302 | 0.839 | 0.625 | -12.148 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 0.231 | Likely Benign | -2.17 | Neutral | 0.141 | Benign | 0.062 | Benign | 3.59 | Benign | 0.00 | Affected | 0.4636 | 0.1022 | 0 | 1 | 0.4 | 0.94 | ||||||||||||||||||||||||||||||||||||||||
| c.501C>A | D167E 2D  AIThe SynGAP1 D167E missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while ESM1b and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive because it receives two benign votes and two uncertain votes, yielding no clear majority. Foldetta, which would provide a protein‑folding stability estimate, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.429200 | Structured | 0.502306 | Binding | 0.377 | 0.667 | 0.375 | -7.636 | In-Between | 0.516 | Ambiguous | Likely Benign | 0.067 | Likely Benign | -1.52 | Neutral | 0.063 | Benign | 0.062 | Benign | 4.07 | Benign | 0.00 | Affected | 0.1392 | 0.7144 | 3 | 2 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.501C>G | D167E 2D  AIThe SynGAP1 D167E missense variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome, while ESM1b and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive because it receives two benign votes and two uncertain votes, yielding no clear majority. Foldetta, which would provide a protein‑folding stability estimate, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none exists). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.429200 | Structured | 0.502306 | Binding | 0.377 | 0.667 | 0.375 | -7.636 | In-Between | 0.516 | Ambiguous | Likely Benign | 0.067 | Likely Benign | -1.52 | Neutral | 0.063 | Benign | 0.062 | Benign | 4.07 | Benign | 0.00 | Affected | 0.1392 | 0.7144 | 3 | 2 | 0.0 | 14.03 | ||||||||||||||||||||||||||||||||||||||||
| c.502C>T | H168Y 2D  AIThe SynGAP1 missense variant H168Y is listed in ClinVar (ID 956914.0) as benign and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic effect are SIFT and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion is consistent with the ClinVar benign classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.433034 | Structured | 0.502450 | Binding | 0.402 | 0.678 | 0.125 | Benign | 1 | -8.914 | Likely Pathogenic | 0.264 | Likely Benign | Likely Benign | 0.065 | Likely Benign | -1.53 | Neutral | 0.192 | Benign | 0.062 | Benign | 4.18 | Benign | 0.01 | Affected | 4.32 | 3 | 0.0695 | 0.4657 | 0 | 2 | 1.9 | 26.03 | |||||||||||||||||||||||||||||||||||
| c.595A>G | N199D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N199D is not reported in ClinVar and is absent from gnomAD. Functional prediction tools that reach a consensus classify the variant as benign: REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. The only inconclusive results come from ESM1b and Rosetta, which are treated as unavailable. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, also predicts a benign outcome. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict any ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | 0.390993 | Structured | 0.431347 | Uncertain | 0.571 | 0.473 | 0.125 | -7.874 | In-Between | 0.338 | Likely Benign | Likely Benign | -0.14 | Likely Benign | 0.1 | -0.67 | Ambiguous | -0.41 | Likely Benign | 0.13 | Likely Benign | 0.028 | Likely Benign | -1.41 | Neutral | 0.276 | Benign | 0.062 | Benign | 4.22 | Benign | 0.35 | Tolerated | 0.1516 | 0.3514 | 2 | 1 | 0.0 | 0.98 | |||||||||||||||||||||||||||||
| c.1587C>G | I529M 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I529M is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, FoldX, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome; Rosetta is uncertain and therefore treated as unavailable. High‑accuracy assessments—AlphaMissense‑Optimized, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and Foldetta (combining FoldX‑MD and Rosetta outputs)—all uniformly indicate a benign effect. Consequently, the variant is most likely benign based on the collective predictions, and this assessment does not contradict any ClinVar status (none reported). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | 0.318242 | Structured | 0.019545 | Uncertain | 0.901 | 0.403 | 0.000 | 1.489 | Likely Benign | 0.063 | Likely Benign | Likely Benign | 0.10 | Likely Benign | 0.1 | 0.68 | Ambiguous | 0.39 | Likely Benign | -0.17 | Likely Benign | 0.215 | Likely Benign | 0.71 | Neutral | 0.035 | Benign | 0.063 | Benign | -1.27 | Pathogenic | 0.15 | Tolerated | 0.0673 | 0.2876 | 2 | 1 | -2.6 | 18.03 | |||||||||||||||||||||||||||||
| c.2027G>T | S676I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S676I is not reported in ClinVar and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are polyPhen‑2 HumDiv, SIFT, and ESM1b. FoldX, Rosetta, Foldetta, and AlphaMissense‑Default are inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields a benign prediction, while Foldetta remains uncertain. Overall, the balance of evidence (five benign versus three pathogenic predictions) indicates that the variant is most likely benign, and this conclusion does not contradict any ClinVar annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.209395 | Structured | 0.113632 | Uncertain | 0.551 | 0.338 | 0.125 | -10.165 | Likely Pathogenic | 0.383 | Ambiguous | Likely Benign | 1.25 | Ambiguous | 0.3 | 1.12 | Ambiguous | 1.19 | Ambiguous | 0.14 | Likely Benign | 0.170 | Likely Benign | -2.46 | Neutral | 0.801 | Possibly Damaging | 0.063 | Benign | 3.38 | Benign | 0.02 | Affected | 0.0879 | 0.5720 | -1 | -2 | 5.3 | 26.08 | ||||||||||||||||||||||||||||||
| c.2320G>A | A774T 2D  AIThe SynGAP1 missense variant A774T is not reported in ClinVar and is absent from gnomAD. In silico prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a likely benign outcome. No pathogenic predictions are present. High‑accuracy assessments further support this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign; Foldetta results are unavailable. Consequently, the variant is most likely benign based on current predictions, and this assessment does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.450668 | Structured | 0.905168 | Binding | 0.336 | 0.897 | 0.250 | -3.238 | Likely Benign | 0.093 | Likely Benign | Likely Benign | 0.062 | Likely Benign | -0.46 | Neutral | 0.037 | Benign | 0.063 | Benign | 4.20 | Benign | 0.39 | Tolerated | 0.1436 | 0.7578 | 1 | 0 | -2.5 | 30.03 | |||||||||||||||||||||||||||||||||||||||
| c.2386C>G | P796A 2D  AIThe SynGAP1 missense variant P796A is not reported in ClinVar (ClinVar ID: None) but is present in gnomAD (gnomAD ID: 6‑33442938‑C‑G). All evaluated in silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized return benign or benign‑like scores. No tool predicts pathogenicity. Grouping by agreement, the benign‑predicting tools comprise the entire set, while no pathogenic predictions are available. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method, did not provide a result for this variant. Overall, the computational evidence strongly suggests that P796A is most likely benign, and this conclusion does not contradict the absence of a ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | 0.936162 | Disordered | 0.426363 | Uncertain | 0.427 | 0.900 | 0.875 | 6-33442938-C-G | -6.077 | Likely Benign | 0.050 | Likely Benign | Likely Benign | 0.023 | Likely Benign | -0.31 | Neutral | 0.174 | Benign | 0.063 | Benign | 4.28 | Benign | 0.08 | Tolerated | 4.32 | 2 | 0.3598 | 0.3776 | -1 | 1 | 3.4 | -26.04 | |||||||||||||||||||||||||||||||||||
| c.3914C>T | T1305I 2D  AIThe SynGAP1 missense variant T1305I is not reported in ClinVar and is absent from gnomAD. Prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the change as benign, while the single pathogenic call comes from SIFT. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.791621 | Disordered | 0.894658 | Binding | 0.390 | 0.873 | 0.875 | -4.202 | Likely Benign | 0.221 | Likely Benign | Likely Benign | 0.154 | Likely Benign | -1.35 | Neutral | 0.027 | Benign | 0.063 | Benign | 2.77 | Benign | 0.02 | Affected | 0.0923 | 0.6149 | 0 | -1 | 5.2 | 12.05 | |||||||||||||||||||||||||||||||||||||||
| c.3948T>A | N1316K 2D  AIThe SynGAP1 missense variant N1316K is not reported in ClinVar (ClinVar status: none) but is present in gnomAD (ID 6‑33451822‑T‑A). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign vs 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict any ClinVar annotation (none). Thus, the variant is most likely benign based on current predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.899122 | Disordered | 0.971970 | Binding | 0.380 | 0.885 | 0.750 | 6-33451822-T-A | -3.815 | Likely Benign | 0.699 | Likely Pathogenic | Likely Benign | 0.044 | Likely Benign | -3.27 | Deleterious | 0.414 | Benign | 0.063 | Benign | 4.00 | Benign | 0.00 | Affected | 3.77 | 5 | 0.2065 | 0.5500 | 0 | 1 | -0.4 | 14.07 | |||||||||||||||||||||||||||||||||||||
| c.3948T>G | N1316K 2D  AIThe SynGAP1 missense variant N1316K is not reported in ClinVar (ClinVar status: none) but is present in gnomAD (ID 6‑33451822‑T‑G). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive (2 benign + 2 pathogenic), and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar status, which is currently unreported. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.899122 | Disordered | 0.971970 | Binding | 0.380 | 0.885 | 0.750 | 6-33451822-T-G | -3.815 | Likely Benign | 0.699 | Likely Pathogenic | Likely Benign | 0.044 | Likely Benign | -3.27 | Deleterious | 0.414 | Benign | 0.063 | Benign | 4.00 | Benign | 0.00 | Affected | 3.77 | 5 | 0.2065 | 0.5500 | 0 | 1 | -0.4 | 14.07 | |||||||||||||||||||||||||||||||||||||
| c.4001A>C | N1334T 2D  AIThe SynGAP1 missense variant N1334T is not reported in ClinVar (ClinVar ID = None) and is absent from gnomAD (gnomAD ID = None). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PROVEAN, SIFT, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, while the SGM Consensus (a majority vote among AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is inconclusive (two pathogenic vs. two benign votes). Foldetta, which evaluates protein‑folding stability, has no available result for this variant. Overall, the majority of evidence points to a benign impact, and there is no ClinVar entry to contradict this assessment. Thus, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | 0.915074 | Disordered | 0.960403 | Binding | 0.406 | 0.734 | 0.875 | -4.249 | Likely Benign | 0.576 | Likely Pathogenic | Likely Benign | 0.103 | Likely Benign | -3.45 | Deleterious | 0.047 | Benign | 0.063 | Benign | 3.59 | Benign | 0.00 | Affected | 0.1600 | 0.5519 | 0 | 0 | 2.8 | -13.00 | ||||||||||||||||||||||||||||||||||||||||
| c.431C>A | T144K 2D  AIThe SynGAP1 T144K variant is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD. Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Those that predict a pathogenic effect comprise PROVEAN, SIFT, ESM1b, and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as Likely Pathogenic. High‑accuracy assessments further support a deleterious outcome: AlphaMissense‑Optimized predicts Pathogenic, and the SGM‑Consensus also indicates Likely Pathogenic. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of computational evidence points to a pathogenic impact, and this assessment does not contradict any ClinVar status because none is assigned. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.666105 | Disordered | 0.524000 | Binding | 0.335 | 0.838 | 0.625 | -15.436 | Likely Pathogenic | 0.956 | Likely Pathogenic | Likely Pathogenic | 0.164 | Likely Benign | -2.83 | Deleterious | 0.180 | Benign | 0.063 | Benign | 3.76 | Benign | 0.00 | Affected | 0.1462 | 0.3179 | 0 | -1 | -3.2 | 27.07 | |||||||||||||||||||||||||||||||||||||||
| c.437C>T | S146L 2D  AIThe SynGAP1 missense variant S146L is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. In contrast, tools that predict a pathogenic effect are PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) which classifies the variant as Likely Pathogenic. High‑accuracy assessments show AlphaMissense‑Optimized as Pathogenic and the SGM‑Consensus as Likely Pathogenic; Foldetta results are not available. Overall, the majority of predictions indicate a pathogenic impact, and this is consistent with the lack of ClinVar annotation—there is no contradiction with ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | 0.541878 | Disordered | 0.508612 | Binding | 0.349 | 0.837 | 0.625 | -15.179 | Likely Pathogenic | 0.976 | Likely Pathogenic | Likely Pathogenic | 0.130 | Likely Benign | -3.77 | Deleterious | 0.084 | Benign | 0.063 | Benign | 3.63 | Benign | 0.00 | Affected | 0.0856 | 0.4975 | -3 | -2 | 4.6 | 26.08 | |||||||||||||||||||||||||||||||||||||||
| c.1929G>C | E643D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant E643D is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, AlphaMissense‑Default, AlphaMissense‑Optimized, FATHMM, and polyPhen2_HumVar. Tools that predict a pathogenic effect are premPS, PROVEAN, polyPhen2_HumDiv, SIFT, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑vs‑2 split. Overall, the majority of predictions (8 benign vs. 5 pathogenic) support a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.033407 | Structured | 0.215915 | Uncertain | 0.871 | 0.315 | 0.000 | -8.083 | Likely Pathogenic | 0.223 | Likely Benign | Likely Benign | 0.46 | Likely Benign | 0.2 | -0.34 | Likely Benign | 0.06 | Likely Benign | 1.09 | Destabilizing | 0.292 | Likely Benign | -2.96 | Deleterious | 0.694 | Possibly Damaging | 0.064 | Benign | 2.98 | Benign | 0.01 | Affected | 0.2040 | 0.4276 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||
| c.1929G>T | E643D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant E643D is not reported in ClinVar (ClinVar ID None) and is absent from gnomAD (gnomAD ID None). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, AlphaMissense‑Default, AlphaMissense‑Optimized, FATHMM, and polyPhen2_HumVar. Tools that predict a pathogenic effect are premPS, PROVEAN, polyPhen2_HumDiv, SIFT, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑vs‑2 split. Overall, the majority of predictions (8 benign vs. 5 pathogenic) support a benign classification. This conclusion does not contradict ClinVar status, as no ClinVar assertion exists for this variant. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | 0.033407 | Structured | 0.215915 | Uncertain | 0.871 | 0.315 | 0.000 | -8.083 | Likely Pathogenic | 0.223 | Likely Benign | Likely Benign | 0.46 | Likely Benign | 0.2 | -0.34 | Likely Benign | 0.06 | Likely Benign | 1.09 | Destabilizing | 0.292 | Likely Benign | -2.96 | Deleterious | 0.694 | Possibly Damaging | 0.064 | Benign | 2.98 | Benign | 0.01 | Affected | 0.2040 | 0.4276 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||||||
| c.3029T>A | F1010Y 2D  AIThe SynGAP1 missense variant F1010Y is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict any ClinVar annotation (none is present). Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.741537 | Disordered | 0.912572 | Binding | 0.286 | 0.881 | 0.625 | -3.297 | Likely Benign | 0.138 | Likely Benign | Likely Benign | 0.071 | Likely Benign | -0.96 | Neutral | 0.031 | Benign | 0.064 | Benign | 2.64 | Benign | 0.05 | Affected | 0.1651 | 0.2514 | 7 | 3 | -4.1 | 16.00 | |||||||||||||||||||||||||||||||||||||||
| c.3740G>A | R1247K 2D  AIThe SynGAP1 missense variant R1247K is reported in gnomAD (6-33446732‑G‑A) and has no ClinVar entry. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are SIFT and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact. There is no ClinVar classification to contradict this assessment. Thus, based on current predictions, the variant is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | 0.736850 | Disordered | 0.374141 | Uncertain | 0.875 | 0.557 | 0.625 | 6-33446732-G-A | 4 | 2.48e-6 | -4.713 | Likely Benign | 0.179 | Likely Benign | Likely Benign | 0.082 | Likely Benign | -2.39 | Neutral | 0.122 | Benign | 0.064 | Benign | 1.80 | Pathogenic | 0.00 | Affected | 3.77 | 5 | 0.3428 | 0.2175 | 2 | 3 | 0.6 | -28.01 | |||||||||||||||||||||||||||||||||
| c.3871C>G | L1291V 2D  AIThe SynGAP1 missense variant L1291V is not reported in ClinVar and is absent from gnomAD, indicating no documented clinical or population evidence. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict a benign effect. No tool in the dataset indicates pathogenicity, so the pathogenic prediction group is empty. High‑accuracy assessments corroborate this benign consensus: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification. Foldetta results are not available, so they do not influence the assessment. Overall, the variant is most likely benign, and this prediction aligns with the absence of any ClinVar pathogenic annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.827927 | Disordered | 0.863458 | Binding | 0.579 | 0.797 | 0.625 | -4.583 | Likely Benign | 0.103 | Likely Benign | Likely Benign | 0.028 | Likely Benign | 0.28 | Neutral | 0.149 | Benign | 0.064 | Benign | 2.61 | Benign | 0.07 | Tolerated | 0.1435 | 0.2946 | 2 | 1 | 0.4 | -14.03 | |||||||||||||||||||||||||||||||||||||||
| c.494G>C | S165T 2D  AIThe SynGAP1 missense variant S165T is not reported in ClinVar (no ClinVar ID) and is absent from gnomAD (no gnomAD ID). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only SIFT predicts a pathogenic outcome. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect. The variant is most likely benign, and this conclusion does not contradict any ClinVar status, as no ClinVar entry exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | 0.429200 | Structured | 0.509123 | Binding | 0.324 | 0.644 | 0.250 | -6.952 | Likely Benign | 0.205 | Likely Benign | Likely Benign | 0.076 | Likely Benign | -0.94 | Neutral | 0.155 | Benign | 0.064 | Benign | 4.02 | Benign | 0.00 | Affected | 0.1754 | 0.5779 | 1 | 1 | 0.1 | 14.03 | |||||||||||||||||||||||||||||||||||||||
| c.630C>A | H210Q 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant H210Q is not reported in ClinVar and is absent from gnomAD. Functional prediction tools show mixed results: benign calls come from REVEL, FoldX, polyPhen‑2 (HumDiv and HumVar) and FATHMM, whereas pathogenic calls are made by premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default and AlphaMissense‑Optimized. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM and PROVEAN, favors a pathogenic outcome (3/4). High‑accuracy assessments further support pathogenicity: AlphaMissense‑Optimized predicts pathogenic, and the SGM‑Consensus (majority vote) also predicts pathogenic. Foldetta, a protein‑folding stability predictor that integrates FoldX‑MD and Rosetta, is inconclusive and therefore not used as evidence. Overall, the majority of reliable predictors indicate a pathogenic effect for H210Q, and this conclusion does not conflict with any ClinVar annotation because none exists. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | PH | 0.144935 | Structured | 0.390904 | Uncertain | 0.872 | 0.298 | 0.125 | -12.639 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 0.26 | Likely Benign | 0.3 | 1.96 | Ambiguous | 1.11 | Ambiguous | 1.20 | Destabilizing | 0.258 | Likely Benign | -6.84 | Deleterious | 0.141 | Benign | 0.064 | Benign | 3.10 | Benign | 0.00 | Affected | 0.1016 | 0.2852 | 3 | 0 | -0.3 | -9.01 | |||||||||||||||||||||||||||||
Found 8751 rows. Show 800 rows per page. Page 2/11 « Previous | Next »