

Table of SynGAP1 Isoform α2 (UniProt Q96PV0-1) Missense Variants.
| c.dna | Variant | SGM Consensus | Domain | ClinVar | gnomAD | ESM1b | AlphaMissense | REVEL | FoldX | Rosetta | Foldetta | PremPS | PROVEAN | PolyPhen-2 HumDiv | PolyPhen-2 HumVar | FATHMM | SIFT | PAM | Physical | SASA | Normalized B-factor backbone | Normalized B-factor sidechain | SynGAP Structural Annotation | DOI | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Clinical Status | Review | Subm. | ID | Allele count | Allele freq. | LLR score | Prediction | Pathogenicity | Class | Optimized | Score | Prediction | Average ΔΔG | Prediction | StdDev | ΔΔG | Prediction | ΔΔG | Prediction | ΔΔG | Prediction | Score | Prediction | pph2_prob | Prediction | pph2_prob | Prediction | Nervous System Score | Prediction | Prediction | Status | Conservation | Sequences | PAM250 | PAM120 | Hydropathy Δ | MW Δ | Average | Δ | Δ | StdDev | Δ | StdDev | Secondary | Tertiary bonds | Inside out | GAP-Ras interface | At membrane | No effect | MD Alert | Verdict | Description | |||||
| c.1436G>C | R479P 2D  3DClick to see structure in 3D Viewer AIClinVar lists the SynGAP1 R479P variant as Uncertain, and it is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, SIFT, and FATHMM. Those that predict a pathogenic effect are FoldX, Rosetta, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, AlphaMissense‑Default, and ESM1b. Predictions that are inconclusive are AlphaMissense‑Optimized and premPS. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta (combining FoldX‑MD and Rosetta) as pathogenic. Overall, the majority of tools and the high‑accuracy methods support a pathogenic classification, which is in contrast to the ClinVar designation of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -11.795 | Likely Pathogenic | 0.938 | Likely Pathogenic | Ambiguous | 0.277 | Likely Benign | 2.86 | Destabilizing | 0.2 | 3.88 | Destabilizing | 3.37 | Destabilizing | 0.81 | Ambiguous | -3.52 | Deleterious | 1.000 | Probably Damaging | 1.000 | Probably Damaging | 3.41 | Benign | 0.18 | Tolerated | 0 | -2 | 2.9 | -59.07 | ||||||||||||||||||||||
| c.1594A>C | T532P 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T532P is listed in ClinVar as Benign (ClinVar ID 1598909.0) and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote). Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments—AlphaMissense‑Optimized, the SGM Consensus, and Foldetta (combining FoldX‑MD and Rosetta outputs)—all indicate a benign impact. No prediction or folding‑stability result is missing or inconclusive. Based on the collective evidence, the variant is most likely benign, and this conclusion is consistent with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Benign | 1 | -2.143 | Likely Benign | 0.061 | Likely Benign | Likely Benign | 0.201 | Likely Benign | -0.30 | Likely Benign | 0.2 | 0.06 | Likely Benign | -0.12 | Likely Benign | 0.08 | Likely Benign | -0.90 | Neutral | 0.005 | Benign | 0.008 | Benign | -1.28 | Pathogenic | 0.18 | Tolerated | 3.37 | 35 | 0 | -1 | -0.9 | -3.99 | 174.2 | 35.1 | 0.4 | 0.0 | 0.1 | 0.0 | X | Potentially Benign | Thr532 is located on an α-α loop between the two α-helices (res. Gly502-Tyr518 and Ala533-Val560) facing the membrane. In the WT simulations, the hydroxyl group of Thr532 occasionally forms hydrogen bonds with the backbone atoms of other loop residues without any specific interaction. In the variant simulations, the Pro532 residue swap does not cause structural changes. Although hydrophilic residues seem more favorable in the loop, the pyrrolidine side chain of proline is well suited for unstructured protein regions such as loops. However, due to its location at the SynGAP-membrane interface, the effect of the residue swap cannot be fully addressed using the SynGAP solvent-only simulations. | |||||||||||
| c.2632A>G | T878A 2D  AIThe SynGAP1 missense variant T878A is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. All available in‑silico predictors agree on a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” High‑accuracy tools likewise support a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is benign; Foldetta results are unavailable. Based on the unanimous benign predictions, the variant is most likely benign, and this assessment does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.154 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.088 | Likely Benign | -0.67 | Neutral | 0.003 | Benign | 0.006 | Benign | 2.73 | Benign | 0.18 | Tolerated | 3.77 | 5 | 1 | 0 | 2.5 | -30.03 | ||||||||||||||||||||||||||||||
| c.2711T>C | M904T 2D  AIThe SynGAP1 missense variant M904T is listed in ClinVar (ID 1311496.0) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.721 | Likely Benign | 0.668 | Likely Pathogenic | Likely Benign | 0.042 | Likely Benign | -1.15 | Neutral | 0.277 | Benign | 0.103 | Benign | 2.78 | Benign | 0.18 | Tolerated | 3.77 | 5 | -1 | -1 | -2.6 | -30.09 | ||||||||||||||||||||||||||||||
| c.3907G>A | G1303S 2D  AIThe SynGAP1 missense variant G1303S is listed in ClinVar (ID 1736068.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only polyPhen‑2 HumDiv predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this is consistent with the ClinVar “Uncertain” status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.271 | Likely Benign | 0.125 | Likely Benign | Likely Benign | 0.155 | Likely Benign | -0.19 | Neutral | 0.649 | Possibly Damaging | 0.433 | Benign | 2.84 | Benign | 0.18 | Tolerated | 1 | 0 | -0.4 | 30.03 | ||||||||||||||||||||||||||||||||
| c.3932T>C | L1311P 2D  AIThe SynGAP1 missense variant L1311P is listed in ClinVar (ID 833866.0) as Benign and is present in gnomAD (variant ID 6‑33451806‑T‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only polyPhen‑2 HumDiv predicts a pathogenic outcome. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a Likely Benign classification, and AlphaMissense‑Optimized also reports Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, is not available for this variant. Overall, the majority of computational evidence supports a benign effect, which is consistent with the ClinVar benign annotation and does not contradict the database status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Likely Benign | 1 | 6-33451806-T-C | 1 | 6.21e-7 | -1.831 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.123 | Likely Benign | -0.52 | Neutral | 0.579 | Possibly Damaging | 0.335 | Benign | 2.72 | Benign | 0.18 | Tolerated | 3.77 | 5 | -3 | -3 | -5.4 | -16.04 | |||||||||||||||||||||||||||
| c.1055C>A | T352N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T352N is listed in ClinVar as Benign (ClinVar ID 590151.0) and is present in the gnomAD database (gnomAD ID 6‑33437960‑C‑A). Across the broad panel of in‑silico predictors, 13 tools (REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus) uniformly report a benign effect, whereas only FATHMM predicts pathogenicity. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign; the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, is also benign. No predictions or stability analyses are missing or inconclusive. Overall, the computational evidence strongly supports a benign classification, consistent with the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Likely Benign | 1 | 6-33437960-C-A | 2 | 1.24e-6 | -4.817 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.027 | Likely Benign | 0.20 | Likely Benign | 0.0 | -0.04 | Likely Benign | 0.08 | Likely Benign | 0.45 | Likely Benign | -0.92 | Neutral | 0.255 | Benign | 0.057 | Benign | 1.75 | Pathogenic | 0.19 | Tolerated | 3.37 | 25 | 0 | 0 | -2.8 | 13.00 | 208.4 | -14.5 | -0.2 | 0.1 | -0.1 | 0.0 | X | Potentially Benign | Thr352 is located in a short α helical section within a loop connecting two β strands (res. Gly341-Pro349, res. Thr359-Pro364) originating from two different anti-parallel β sheets of the C2 domain. In the WT simulations, the side chain hydroxyl and backbone amide groups of Thr354 form hydrogen bonds with the backbone carbonyl group of Pro349 at the end of the preceding β strand. This arrangement likely stabilizes the α helical section and aids in folding, keeping the short secondary structure element intact in the variant simulations. However, the carboxamide group of the Asn352 side chain does not form hydrogen bonds with the backbone carbonyl group of Pro349. Instead, it packs against the cyclic ring and forms hydrogen bonds with the phenol group of the Tyr363 side chain in the other β strand. | ||||||||
| c.1835A>C | Q612P 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant Q612P is listed in ClinVar (ID 3660462.0) with an uncertain significance annotation and is not reported in gnomAD. Functional prediction tools cluster into two groups: benign predictions come from FoldX, SIFT, and AlphaMissense‑Optimized; pathogenic predictions arise from REVEL, PolyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, PROVEAN, and the SGM Consensus score (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). The high‑accuracy AlphaMissense‑Optimized predicts benign, whereas the SGM Consensus predicts likely pathogenic; Foldetta, a folding‑stability method combining FoldX‑MD and Rosetta outputs, returns an uncertain result and is therefore not factored into the consensus. Overall, the majority of evidence supports a pathogenic effect, which contrasts with the ClinVar uncertain classification. Thus, based on current predictions, the variant is most likely pathogenic, contradicting the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -9.684 | Likely Pathogenic | 0.673 | Likely Pathogenic | Likely Benign | 0.671 | Likely Pathogenic | -0.19 | Likely Benign | 0.3 | 3.06 | Destabilizing | 1.44 | Ambiguous | 0.56 | Ambiguous | -5.84 | Deleterious | 1.000 | Probably Damaging | 1.000 | Probably Damaging | -1.31 | Pathogenic | 0.19 | Tolerated | 0 | -1 | 1.9 | -31.01 | ||||||||||||||||||||||
| c.3368G>A | G1123D 2D  AISynGAP1 missense variant G1123D is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33443920‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Optimized, and the SGM Consensus (derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic outcome are polyPhen‑2 HumDiv and ESM1b. AlphaMissense‑Default remains uncertain, and Foldetta results are unavailable. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus also benign, while Foldetta provides no data. Overall, the majority of evidence points to a benign effect, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Uncertain | 1 | 6-33443920-G-A | 2 | 1.33e-6 | -10.321 | Likely Pathogenic | 0.405 | Ambiguous | Likely Benign | 0.360 | Likely Benign | -0.78 | Neutral | 0.500 | Possibly Damaging | 0.157 | Benign | 4.34 | Benign | 0.19 | Tolerated | 3.77 | 5 | 1 | -1 | -3.1 | 58.04 | ||||||||||||||||||||||||||||
| c.2458T>A | Y820N 2D  AIThe SynGAP1 Y820N variant is listed in ClinVar with an “Uncertain” significance and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, and FATHMM, whereas polyPhen‑2 (HumDiv and HumVar), ESM1b, and AlphaMissense‑Default all predict a pathogenic outcome. AlphaMissense‑Optimized returns an “Uncertain” result. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive (two benign vs. two pathogenic votes). Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the predictions are evenly split between benign and pathogenic, with no high‑confidence pathogenic or benign signal. Thus, the variant is most likely of uncertain significance, which is consistent with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Uncertain | 1 | -9.032 | Likely Pathogenic | 0.842 | Likely Pathogenic | Ambiguous | 0.143 | Likely Benign | -1.53 | Neutral | 0.999 | Probably Damaging | 0.977 | Probably Damaging | 2.74 | Benign | 0.20 | Tolerated | -2 | -2 | -2.2 | -49.07 | |||||||||||||||||||||||||||||||||
| c.2765G>A | R922Q 2D  AIThe SynGAP1 missense variant R922Q is listed in ClinVar as Benign (ClinVar ID 2917638.0) and is present in gnomAD (ID 6‑33443317‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic effect are PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (derived from the same set of high‑confidence predictors) also as benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, aligning with the ClinVar classification and indicating no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33443317-G-A | 7 | 4.34e-6 | -3.295 | Likely Benign | 0.189 | Likely Benign | Likely Benign | 0.085 | Likely Benign | -0.27 | Neutral | 0.992 | Probably Damaging | 0.736 | Possibly Damaging | 2.57 | Benign | 0.20 | Tolerated | 3.77 | 5 | 1 | 1 | 1.0 | -28.06 | |||||||||||||||||||||||||||
| c.2924C>G | T975S 2D  AIThe SynGAP1 missense variant T975S is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized scores the variant as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the available predictions strongly suggest that T975S is most likely benign, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.743 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.109 | Likely Benign | -0.57 | Neutral | 0.059 | Benign | 0.061 | Benign | 4.16 | Benign | 0.20 | Tolerated | 1 | 1 | -0.1 | -14.03 | ||||||||||||||||||||||||||||||||
| c.1406C>A | A469D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A469D is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Prediction tools that assess the variant’s effect fall into two groups: the single benign prediction from SIFT, and a consensus of pathogenic predictions from the remaining 15 tools (REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus). High‑accuracy methods further support a deleterious impact: AlphaMissense‑Optimized predicts pathogenic, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is pathogenic, and Foldetta (combining FoldX‑MD and Rosetta outputs) also indicates pathogenic. Taken together, the overwhelming majority of evidence points to a pathogenic effect, which is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -14.643 | Likely Pathogenic | 0.999 | Likely Pathogenic | Likely Pathogenic | 0.738 | Likely Pathogenic | 5.09 | Destabilizing | 0.2 | 4.16 | Destabilizing | 4.63 | Destabilizing | 1.68 | Destabilizing | -3.48 | Deleterious | 0.999 | Probably Damaging | 0.996 | Probably Damaging | -1.34 | Pathogenic | 0.21 | Tolerated | 3.37 | 34 | 0 | -2 | -5.3 | 44.01 | 237.0 | -58.2 | -0.2 | 0.1 | 0.8 | 0.1 | X | X | Potentially Pathogenic | The methyl group of Ala469, located in an α helix (res. Ala461–Phe476), interacts with hydrophobic residues (e.g., Trp572, Leu588, Met470) in an inter-helix space formed by two other α helices (res. Glu582–Ser604, res. Arg563–Gly580). In the variant simulations, Asp469 introduces a negatively charged and bulky side chain into the hydrophobic niche. Consequently, the side chain of Asp469 rotates outward, allowing the carboxylate group to form a salt bridge with the guanidinium group of Arg575 on the protein surface. This interaction affects the continuity of the parent α helix (Ala461–Phe476). Due to the importance of hydrophobic packing, the structural effects could be more pronounced during actual protein folding. | ||||||||||
| c.2195G>A | R732K 2D  AIThe SynGAP1 missense variant R732K is listed in ClinVar (ID 537019.0) with an “Uncertain” clinical significance and is present in gnomAD (6‑33441660‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). In contrast, the two polyPhen‑2 implementations (HumDiv and HumVar) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; no Foldetta stability result is available. Overall, the majority of evidence points to a benign impact, and this consensus does not conflict with the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 2 | 6-33441660-G-A | 4 | 2.48e-6 | -5.278 | Likely Benign | 0.240 | Likely Benign | Likely Benign | 0.045 | Likely Benign | -0.82 | Neutral | 0.973 | Probably Damaging | 0.943 | Probably Damaging | 2.69 | Benign | 0.21 | Tolerated | 3.59 | 7 | 3 | 2 | 0.6 | -28.01 | |||||||||||||||||||||||||||
| c.2596G>T | V866L 2D  AIThe SynGAP1 missense variant V866L is listed in ClinVar (ID 469150.0) with an “Uncertain” clinical significance and is present in gnomAD (6‑33443148‑G‑T). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic outcome. High‑accuracy assessments corroborate this benign prediction: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence strongly supports a benign effect, and this conclusion does not contradict the current ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443148-G-T | 1 | 6.20e-7 | -3.352 | Likely Benign | 0.148 | Likely Benign | Likely Benign | 0.046 | Likely Benign | -0.97 | Neutral | 0.217 | Benign | 0.229 | Benign | 2.71 | Benign | 0.21 | Tolerated | 3.82 | 4 | 2 | 1 | -0.4 | 14.03 | |||||||||||||||||||||||||||
| c.2635_2636delinsAA | A879K 2D  AIThe SynGAP1 missense variant A879K is listed in ClinVar (ID 575856.0) as benign and is not reported in gnomAD. Prediction tools that agree on a benign effect include PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this consensus aligns with the ClinVar designation, with no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Likely Benign | 1 | -5.877 | Likely Benign | 0.757 | Likely Pathogenic | Likely Benign | -0.71 | Neutral | 0.969 | Probably Damaging | 0.593 | Possibly Damaging | 2.69 | Benign | 0.21 | Tolerated | 3.77 | 5 | -1 | -1 | -5.7 | 57.10 | ||||||||||||||||||||||||||||||||
| c.2349G>A | M783I 2D  AIThe SynGAP1 missense variant M783I is listed in ClinVar as a benign alteration (ClinVar ID 3618151.0) and is present in the gnomAD database (gnomAD ID 6‑33442901‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also indicates a likely benign effect. The Foldetta protein‑folding stability analysis is not available for this variant. Overall, the computational evidence strongly suggests that the variant is most likely benign, in agreement with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33442901-G-A | 6 | 3.72e-6 | -3.560 | Likely Benign | 0.418 | Ambiguous | Likely Benign | 0.042 | Likely Benign | -0.54 | Neutral | 0.004 | Benign | 0.006 | Benign | 2.87 | Benign | 0.22 | Tolerated | 3.64 | 6 | 1 | 2 | 2.6 | -18.03 | |||||||||||||||||||||||||||
| c.526A>G | S176G 2D  AIThe SynGAP1 missense variant S176G is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33435168‑A‑G). Consensus among most in silico predictors is benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized all report a benign effect. No tool predicts pathogenicity. Two predictors are inconclusive: ESM1b and AlphaMissense‑Default, which are grouped under uncertain. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, while the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) remains uncertain, and Foldetta stability analysis is unavailable. Overall, the computational evidence overwhelmingly favors a benign impact, which does not contradict the ClinVar uncertain classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Uncertain | 1 | 6-33435168-A-G | 1 | 6.20e-7 | -7.541 | In-Between | 0.360 | Ambiguous | Likely Benign | 0.066 | Likely Benign | -1.08 | Neutral | 0.131 | Benign | 0.039 | Benign | 4.08 | Benign | 0.22 | Tolerated | 3.54 | 6 | 0 | 1 | 0.4 | -30.03 | ||||||||||||||||||||||||||||
| c.901G>A | A301T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A301T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33437806‑G‑A). Prediction tools that uniformly indicate a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only the two polyPhen‑2 implementations (HumDiv and HumVar) predict a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, is benign. Overall, the majority of evidence points to a benign effect, and this is not in conflict with the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 5 | 6-33437806-G-A | 2 | 1.24e-6 | -3.448 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.150 | Likely Benign | 0.36 | Likely Benign | 0.2 | -0.33 | Likely Benign | 0.02 | Likely Benign | 0.03 | Likely Benign | -0.25 | Neutral | 0.997 | Probably Damaging | 0.989 | Probably Damaging | 4.15 | Benign | 0.22 | Tolerated | 4.32 | 14 | 1 | 0 | -2.5 | 30.03 | 219.8 | -42.8 | -0.1 | 0.0 | -0.5 | 0.2 | Uncertain | The methyl group of Ala301, located in a β hairpin loop linking two anti-parallel β sheet strands (res. Met289-Pro298, res. Thr305-Asn315), points outward from the β hairpin loop, and its backbone atoms do not participate in the loop formation in the WT simulations. In the variant simulations, the hydroxyl group of the Thr301 side chain also mostly points outward; however, the guanidinium group of Arg299 is moved away from its central hairpin loop position.β hairpins are potential nucleation sites during the initial stages of protein folding, so even minor changes in them could be significant. Due to its location near the membrane surface, the residue swap could also affect the C2 loop dynamics and SynGAP-membrane association. However, this is beyond the scope of the solvent-only simulations to unravel. | |||||||||
| c.3103C>A | P1035T 2D  AIThe SynGAP1 missense variant P1035T is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic outcome are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. AlphaMissense‑Default remains uncertain, and no Foldetta stability assessment is available. High‑accuracy evidence from AlphaMissense‑Optimized and the SGM‑Consensus both support a benign classification, while the absence of a Foldetta result does not alter this view. Overall, the majority of predictions indicate a benign effect, and this is consistent with the ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -4.447 | Likely Benign | 0.426 | Ambiguous | Likely Benign | 0.087 | Likely Benign | -0.96 | Neutral | 0.901 | Possibly Damaging | 0.537 | Possibly Damaging | 2.72 | Benign | 0.23 | Tolerated | 3.77 | 5 | 0 | -1 | 0.9 | 3.99 | ||||||||||||||||||||||||||||||
| c.3344T>C | I1115T 2D  AIThe SynGAP1 missense variant I1115T is listed in ClinVar as a benign alteration (ClinVar ID 130530.0) and is present in the gnomAD database (gnomAD ID 6‑33443896‑T‑C). All evaluated in‑silico predictors classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence strongly supports a benign classification, fully consistent with the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 9 | 6-33443896-T-C | 20536 | 1.36e-2 | -2.670 | Likely Benign | 0.068 | Likely Benign | Likely Benign | 0.100 | Likely Benign | -0.04 | Neutral | 0.000 | Benign | 0.001 | Benign | 2.76 | Benign | 0.23 | Tolerated | 4.32 | 2 | 0 | -1 | -5.2 | -12.05 | |||||||||||||||||||||||||||
| c.3964G>C | A1322P 2D  AIThe SynGAP1 missense variant A1322P is reported in ClinVar (ID 1169945.0) as benign and is present in gnomAD (variant ID 6‑33451838‑G‑C). Across the available in‑silico predictors, all tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign predictions. No tool in the dataset indicates a pathogenic effect. High‑accuracy assessments corroborate this consensus: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” verdict. The Foldetta protein‑folding stability analysis is not available for this variant. Overall, the computational evidence strongly supports a benign classification, which is consistent with the ClinVar status and does not contradict it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33451838-G-C | -1.153 | Likely Benign | 0.063 | Likely Benign | Likely Benign | 0.090 | Likely Benign | 0.03 | Neutral | 0.000 | Benign | 0.000 | Benign | 4.15 | Benign | 0.23 | Tolerated | 3.77 | 5 | 1 | -1 | -3.4 | 26.04 | |||||||||||||||||||||||||||||
| c.1240A>G | M414V 2D  AISynGAP1 M414V is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools show a split: benign calls come from REVEL, SIFT, FATHMM, and AlphaMissense‑Optimized; pathogenic calls come from PROVEAN, polyPhen‑2 (HumDiv and HumVar), and ESM1b; the remaining tools (FoldX, Rosetta, Foldetta, premPS, AlphaMissense‑Default) are inconclusive. The SGM consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, yields a pathogenic majority. High‑accuracy assessments give AlphaMissense‑Optimized benign, SGM consensus pathogenic, and Foldetta uncertain. Because the high‑accuracy predictions are divided and the overall tool set is evenly split, there is no definitive evidence for pathogenicity or benignity. Thus, the variant is most likely inconclusive, and this lack of consensus does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Uncertain | 1 | -8.003 | Likely Pathogenic | 0.541 | Ambiguous | Likely Benign | 0.261 | Likely Benign | 1.81 | Ambiguous | 0.4 | 1.73 | Ambiguous | 1.77 | Ambiguous | 0.95 | Ambiguous | -2.95 | Deleterious | 0.999 | Probably Damaging | 0.987 | Probably Damaging | 3.43 | Benign | 0.24 | Tolerated | 2 | 1 | 2.3 | -32.06 | |||||||||||||||||||||||
| c.1409T>C | M470T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M470T is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include only SIFT, whereas the remaining tools—REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN)—all predict a pathogenic impact. High‑accuracy methods further support this: AlphaMissense‑Optimized is pathogenic, the SGM‑Consensus is “Likely Pathogenic,” and Foldetta (combining FoldX‑MD and Rosetta outputs) is pathogenic. No prediction or folding‑stability result is missing or inconclusive. Based on the overwhelming consensus of pathogenic predictions, the variant is most likely pathogenic, which is consistent with the ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -8.104 | Likely Pathogenic | 0.976 | Likely Pathogenic | Likely Pathogenic | 0.763 | Likely Pathogenic | 3.19 | Destabilizing | 0.1 | 2.68 | Destabilizing | 2.94 | Destabilizing | 1.49 | Destabilizing | -5.30 | Deleterious | 0.996 | Probably Damaging | 0.985 | Probably Damaging | -1.08 | Pathogenic | 0.24 | Tolerated | 3.37 | 34 | -1 | -1 | -2.6 | -30.09 | 213.8 | 46.5 | 0.0 | 0.0 | -0.2 | 0.2 | X | X | Potentially Pathogenic | The thioether group of Met470, located in the middle of an α helix (res. Ala461–Phe476), interacts with hydrophobic residues in the inter-helix space (e.g., Val473, Leu558, Cys576, Trp572) formed by two other α helices (res. Ser604–Arg581, res. Pro562–Arg579). In the WT simulations, the Met470 side chain also packs against the positively charged guanidinium groups of Arg575, Arg429, and Arg579, which form salt bridges with the negatively charged carboxylate groups of the Asp474 and Asp467 side chains at the protein surface. In the variant simulations, the hydroxyl group of the Thr470 side chain forms an H-bond with the backbone carbonyl group of Ser466 in the α helix, potentially lowering its structural integrity. Importantly, the hydroxyl group of Thr470 also forms an H-bond with the guanidinium group of Arg575, which helps it form a more permanent salt bridge with Asp467. | ||||||||||
| c.1610C>T | A537V 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant A537V is listed in ClinVar as Benign (ClinVar ID 766762.0) and is present in gnomAD (ID 6‑33438853‑C‑T). Functional prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign; the SGM Consensus, derived from the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, is benign. FoldX alone is uncertain and therefore not considered evidence. Overall, the consensus of available predictions indicates that the variant is most likely benign, in agreement with its ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Likely Benign | 1 | 6-33438853-C-T | 7 | 4.34e-6 | -6.888 | Likely Benign | 0.120 | Likely Benign | Likely Benign | 0.382 | Likely Benign | 0.54 | Ambiguous | 0.0 | -0.05 | Likely Benign | 0.25 | Likely Benign | 0.41 | Likely Benign | -1.97 | Neutral | 0.977 | Probably Damaging | 0.469 | Possibly Damaging | -1.26 | Pathogenic | 0.24 | Tolerated | 3.37 | 35 | 0 | 0 | 2.4 | 28.05 | 220.3 | -45.1 | 0.0 | 0.0 | -0.7 | 0.1 | X | Potentially Benign | Ala537 is located on the outer surface of an α-helix (res. Ala533-Val560). The methyl group of Ala537 is on the surface and does not form any interactions. In the variant simulations, the iso-propyl side chain of Val537 is also on the surface, similar to Ala537 in the WT, causing no negative structural effects. | ||||||||
| c.2852A>G | H951R 2D  AIThe SynGAP1 missense variant H951R is listed in ClinVar as Pathogenic (ClinVar ID 1003739.0) and is not reported in gnomAD. Functional prediction tools uniformly classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign effect. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign. The Foldetta protein‑folding stability analysis is unavailable for this variant. Based on the collective predictions, the variant is most likely benign, which contradicts its ClinVar pathogenic classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Likely Pathogenic | 1 | -4.964 | Likely Benign | 0.125 | Likely Benign | Likely Benign | 0.185 | Likely Benign | -1.08 | Neutral | 0.048 | Benign | 0.029 | Benign | 5.46 | Benign | 0.24 | Tolerated | 3.77 | 5 | 2 | 0 | -1.3 | 19.05 | ||||||||||||||||||||||||||||||
| c.2888A>G | H963R 2D  AIThe SynGAP1 missense variant H963R is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443440‑A‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Only ESM1b predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized reports benign, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a benign consensus. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant, so its status is unavailable. Overall, the preponderance of evidence indicates the variant is most likely benign, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443440-A-G | 8 | 4.96e-6 | -8.952 | Likely Pathogenic | 0.169 | Likely Benign | Likely Benign | 0.081 | Likely Benign | -1.28 | Neutral | 0.001 | Benign | 0.003 | Benign | 4.15 | Benign | 0.24 | Tolerated | 3.77 | 5 | 2 | 0 | -1.3 | 19.05 | |||||||||||||||||||||||||||
| c.3607C>G | H1203D 2D  AIThe SynGAP1 missense variant H1203D is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only polyPhen‑2 HumDiv predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as “Likely Benign”; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict the ClinVar designation, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Uncertain | 1 | -6.729 | Likely Benign | 0.525 | Ambiguous | Likely Benign | 0.403 | Likely Benign | -1.89 | Neutral | 0.473 | Possibly Damaging | 0.265 | Benign | 5.51 | Benign | 0.24 | Tolerated | 3.77 | 5 | 1 | -1 | -0.3 | -22.05 | |||||||||||||||||||||||||||||
| c.416G>A | S139N 2D  AIThe SynGAP1 missense variant S139N is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33432713‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as likely benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33432713-G-A | 3 | 2.22e-6 | -4.584 | Likely Benign | 0.688 | Likely Pathogenic | Likely Benign | 0.109 | Likely Benign | -0.75 | Neutral | 0.149 | Benign | 0.047 | Benign | 4.14 | Benign | 0.24 | Tolerated | 3.61 | 5 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||
| c.1604G>C | S535T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S535T is catalogued in ClinVar as benign (ClinVar ID 537005.0) and is observed in gnomAD (variant ID 6‑33438847‑G‑C). In silico prediction tools largely agree on a benign effect: REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. Only FATHMM predicts a pathogenic outcome. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as likely benign. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign; the SGM Consensus is likely benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also reports a benign effect. Overall, the consensus of predictive tools and high‑accuracy methods indicates that the variant is most likely benign, consistent with its ClinVar classification and presence in gnomAD. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Benign | 1 | 6-33438847-G-C | 14 | 8.67e-6 | -3.886 | Likely Benign | 0.069 | Likely Benign | Likely Benign | 0.177 | Likely Benign | 0.45 | Likely Benign | 0.1 | -0.27 | Likely Benign | 0.09 | Likely Benign | 0.17 | Likely Benign | -0.81 | Neutral | 0.000 | Benign | 0.001 | Benign | -1.25 | Pathogenic | 0.25 | Tolerated | 3.37 | 35 | 1 | 1 | 0.1 | 14.03 | 201.3 | -17.3 | -0.1 | 0.7 | -0.2 | 0.1 | X | Potentially Benign | Ser535 is located near the terminal end of an α-helix (res. Ala533-Val560) close to the membrane interface. In the WT simulations, the hydroxyl side chain of Ser535 forms hydrogen bonds with nearby residues (e.g., His539, Glu538) without any specific interactions. These hydrogen bonds disrupt the structure of the terminal end of the α-helix (Ala533-Ser535), causing it to weaken or unfold during the WT simulations. In the variant simulations, Thr535, a hydrophilic residue with a hydroxyl group of almost the same size as Ser, interacts more frequently with the preceding loop residues (e.g., Thr532, Cys531) due to its longer side chain. Regardless, the residue swap is tolerated in the simulations with no negative effects. However, due to its location near the SynGAP-membrane interface, the effect of the residue swap cannot be fully addressed using the SynGAP solvent-only simulations. | 10.1016/j.ajhg.2020.11.011 | |||||||
| c.3567G>C | E1189D 2D  AIThe SynGAP1 missense variant E1189D (gnomAD ID 6-33444602‑G‑C) is listed in ClinVar as Benign (ClinVar ID 833989.0). In silico predictors that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Predictors that indicate a pathogenic effect are PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. The high‑accuracy AlphaMissense‑Optimized tool classifies the variant as benign, and the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also favors a benign outcome. No Foldetta stability assessment is available for this residue. Overall, the majority of evidence points to a benign impact, and this conclusion aligns with the ClinVar designation, showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Likely Benign | 1 | 6-33444602-G-C | 3 | 1.86e-6 | -3.582 | Likely Benign | 0.461 | Ambiguous | Likely Benign | 0.359 | Likely Benign | -1.42 | Neutral | 0.992 | Probably Damaging | 0.989 | Probably Damaging | 5.30 | Benign | 0.25 | Tolerated | 3.82 | 4 | 3 | 2 | 0.0 | -14.03 | ||||||||||||||||||||||||||
| c.962G>A | R321H 2D 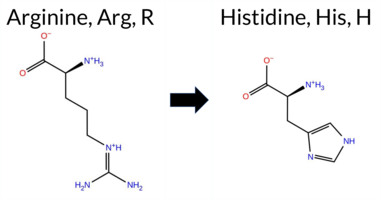 3DClick to see structure in 3D Viewer AISynGAP1 missense variant R321H is listed in ClinVar with an uncertain significance and is present in gnomAD (variant ID 6‑33437867‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, PROVEAN, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic outcome are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and FATHMM, while premPS remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign, Foldetta as benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive due to a 2‑to‑2 split. Overall, the majority of predictions support a benign impact, and this consensus does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | Uncertain | 1 | 6-33437867-G-A | 8 | 4.96e-6 | -8.751 | Likely Pathogenic | 0.136 | Likely Benign | Likely Benign | 0.323 | Likely Benign | 0.48 | Likely Benign | 0.1 | -0.36 | Likely Benign | 0.06 | Likely Benign | 0.59 | Ambiguous | -1.43 | Neutral | 1.000 | Probably Damaging | 0.998 | Probably Damaging | 1.92 | Pathogenic | 0.25 | Tolerated | 3.38 | 23 | 2 | 0 | 1.3 | -19.05 | 218.5 | 86.9 | 1.1 | 0.0 | 0.3 | 0.0 | X | Potentially Benign | The guanidinium group of Arg321, located in a β hairpin loop linking two anti-parallel β sheet strands (res. Thr305-Asn315, res. Ala322-Asp330), faces outward without forming any stable interactions in the WT simulations. Similarly, in the variant simulations, the imidazole ring of His321 also points outward without making any stable intra-protein interactions. Thus, the residue swap does not seem to cause adverse effects on the protein structure based on the simulations. However, β hairpins are potential nucleation sites during the initial stages of protein folding, so even minor changes in them could be significant. | |||||||||
| c.1131G>A | M377I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M377I (ClinVar ID 3803473.0, status = Uncertain) is present in gnomAD (ID = 6‑33438036‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign,” while Foldetta (combining FoldX‑MD and Rosetta outputs) is inconclusive. Overall, the computational evidence strongly favors a benign classification, which does not contradict the ClinVar status of Uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 1 | 6-33438036-G-A | 1 | 6.23e-7 | -2.895 | Likely Benign | 0.212 | Likely Benign | Likely Benign | 0.227 | Likely Benign | 0.76 | Ambiguous | 0.3 | 0.54 | Ambiguous | 0.65 | Ambiguous | 0.24 | Likely Benign | -0.41 | Neutral | 0.000 | Benign | 0.001 | Benign | 5.46 | Benign | 0.26 | Tolerated | 4.32 | 12 | 1 | 2 | 2.6 | -18.03 | |||||||||||||||||
| c.2623G>A | A875T 2D  AIThe SynGAP1 missense variant A875T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6‑33443175‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports the variant as “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact, which is consistent with the ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443175-G-A | 1 | 6.20e-7 | -3.793 | Likely Benign | 0.179 | Likely Benign | Likely Benign | 0.110 | Likely Benign | -1.56 | Neutral | 0.972 | Probably Damaging | 0.864 | Possibly Damaging | 2.72 | Benign | 0.26 | Tolerated | 3.77 | 5 | 0 | 1 | -2.5 | 30.03 | |||||||||||||||||||||||||||
| c.1027G>A | V343I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V343I is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33437932‑G‑A). Prediction tools that uniformly indicate a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, resolves to “Likely Benign” (3 benign vs. 1 pathogenic). High‑accuracy assessments are consistent: AlphaMissense‑Optimized is benign; the SGM‑Consensus is likely benign; and Foldetta, combining FoldX‑MD and Rosetta outputs, is benign. Overall, the collective evidence strongly supports a benign classification, and this does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 2 | 6-33437932-G-A | 1 | 6.20e-7 | -6.020 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.020 | Likely Benign | -0.27 | Likely Benign | 0.0 | -0.04 | Likely Benign | -0.16 | Likely Benign | -0.39 | Likely Benign | -0.14 | Neutral | 0.159 | Benign | 0.084 | Benign | 1.98 | Pathogenic | 0.27 | Tolerated | 3.37 | 25 | 4 | 3 | 0.3 | 14.03 | 240.2 | -26.9 | -0.2 | 0.2 | -0.2 | 0.2 | X | Potentially Benign | The iso-propyl side chain of Val343, located in an anti-parallel β sheet strand (res. Gly341-Pro349), is packing against multiple hydrophobic residues of the C2 domain (e.g., Leu327, Leu274, Val365). In the variant simulations, the sec-butyl side chain of Ile343 is basically able to form the same interactions as valine due to its similar hydrophobic profile. The residue swap also does not seem to cause negative effects on the protein structure based on the simulations. | ||||||||
| c.1231A>G | I411V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I411V is reported in ClinVar as benign (ClinVar ID 1654508.0) and is not found in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Two tools predict a pathogenic outcome: PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. Predictions that are inconclusive or unavailable are AlphaMissense‑Default, FoldX, Rosetta, premPS, and Foldetta. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta) is uncertain. Overall, the preponderance of evidence points to a benign effect for I411V, which is consistent with its ClinVar classification and does not contradict the reported status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Likely Benign | 1 | -6.290 | Likely Benign | 0.385 | Ambiguous | Likely Benign | 0.212 | Likely Benign | 0.74 | Ambiguous | 0.0 | 0.82 | Ambiguous | 0.78 | Ambiguous | 0.99 | Ambiguous | -0.86 | Neutral | 0.935 | Possibly Damaging | 0.858 | Possibly Damaging | 3.90 | Benign | 0.27 | Tolerated | 3.38 | 28 | 4 | 3 | -0.3 | -14.03 | 233.3 | 28.2 | -0.2 | 0.0 | -0.2 | 0.0 | X | Potentially Benign | The sec-butyl side chain of Ile411, located in the hydrophobic space between an anti-parallel β sheet strand (res. Pro398-Ile411) and an α helix (res. Asp684-Gln702), packs against multiple residues (e.g., Met409, Arg259). In the variant simulations, the side chain of Val411 is able to favorably fill the same hydrophobic niche despite its slightly smaller size. In short, the residue swap has no apparent negative effect on the structure based on the simulations. | |||||||||||
| c.2596G>A | V866I 2D  AIThe SynGAP1 missense variant V866I is listed in ClinVar with an “Uncertain” status (ClinVar ID 536995.0) and is present in gnomAD (6‑33443148‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 3 | 6-33443148-G-A | 5 | 3.10e-6 | -4.652 | Likely Benign | 0.118 | Likely Benign | Likely Benign | 0.059 | Likely Benign | -0.39 | Neutral | 0.957 | Probably Damaging | 0.541 | Possibly Damaging | 2.69 | Benign | 0.27 | Tolerated | 3.82 | 4 | 4 | 3 | 0.3 | 14.03 | |||||||||||||||||||||||||||
| c.1635G>A | M545I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M545I is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Prediction tools cluster into two groups: benign predictions arise from FoldX, Rosetta, and SIFT, whereas pathogenic predictions come from REVEL, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, and AlphaMissense‑Default; premPS remains inconclusive. High‑accuracy methods provide mixed evidence: AlphaMissense‑Optimized indicates pathogenicity, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also suggests likely pathogenic, while Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, predicts a benign effect. Overall, the preponderance of conventional tools and the SGM Consensus lean toward pathogenicity, whereas the Foldetta result is an outlier. Thus, the variant is most likely pathogenic based on the available predictions, and this assessment does not contradict its ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -8.348 | Likely Pathogenic | 0.999 | Likely Pathogenic | Likely Pathogenic | 0.592 | Likely Pathogenic | 0.47 | Likely Benign | 0.1 | 0.14 | Likely Benign | 0.31 | Likely Benign | 0.63 | Ambiguous | -3.61 | Deleterious | 0.935 | Possibly Damaging | 0.941 | Probably Damaging | -1.27 | Pathogenic | 0.28 | Tolerated | 3.37 | 35 | 1 | 2 | 2.6 | -18.03 | ||||||||||||||||||||
| c.2669G>C | R890P 2D  AIThe SynGAP1 missense variant R890P is listed in ClinVar (ID 575680.0) as Benign and is present in gnomAD (6‑33443221‑G‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a Likely Benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion aligns with the ClinVar status, showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Likely Benign | 2 | 6-33443221-G-C | 28 | 1.74e-5 | -1.931 | Likely Benign | 0.301 | Likely Benign | Likely Benign | 0.191 | Likely Benign | -1.21 | Neutral | 0.999 | Probably Damaging | 0.977 | Probably Damaging | 4.02 | Benign | 0.28 | Tolerated | 4.32 | 4 | 0 | -2 | 2.9 | -59.07 | |||||||||||||||||||||||||||
| c.3170G>A | S1057N 2D  AIThe SynGAP1 missense variant S1057N is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. All available in‑silico predictors classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” No tool predicts pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, while Foldetta (combining FoldX‑MD and Rosetta outputs) has no available result for this variant. Overall, the computational evidence overwhelmingly supports a benign effect, and this is consistent with the ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -6.386 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.218 | Likely Benign | -0.41 | Neutral | 0.451 | Benign | 0.129 | Benign | 5.25 | Benign | 0.28 | Tolerated | 1 | 1 | -2.7 | 27.03 | ||||||||||||||||||||||||||||||||
| c.3394T>C | S1132P 2D  AIThe SynGAP1 missense variant S1132P is listed in ClinVar with an uncertain significance (ClinVar ID 1341927.0) and is present in the gnomAD database (gnomAD ID 6‑33443946‑T‑C). All available in‑silico predictors uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign or likely benign outcomes. No tool in the dataset predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign effect, which is consistent with the ClinVar uncertain classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 3 | 6-33443946-T-C | 1 | 6.74e-7 | -1.423 | Likely Benign | 0.144 | Likely Benign | Likely Benign | 0.301 | Likely Benign | 0.38 | Neutral | 0.003 | Benign | 0.006 | Benign | 5.40 | Benign | 0.28 | Tolerated | 4.32 | 4 | 1 | -1 | -0.8 | 10.04 | |||||||||||||||||||||||||||
| c.1832T>C | M611T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant M611T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33440884‑T‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. Four tools (FoldX, Rosetta, Foldetta, premPS) return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact, and this does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | 6-33440884-T-C | 1 | 6.19e-7 | -5.696 | Likely Benign | 0.101 | Likely Benign | Likely Benign | 0.240 | Likely Benign | 1.98 | Ambiguous | 0.2 | 0.94 | Ambiguous | 1.46 | Ambiguous | 0.87 | Ambiguous | -2.40 | Neutral | 0.034 | Benign | 0.038 | Benign | -1.19 | Pathogenic | 0.29 | Tolerated | 3.37 | 35 | -1 | -1 | -2.6 | -30.09 | |||||||||||||||||
| c.2858C>A | P953Q 2D  AIThe SynGAP1 missense variant P953Q is listed in ClinVar with an uncertain significance (ClinVar ID 1176820.0) and is not found in gnomAD. Functional prediction tools uniformly classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all report benign effects, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus also reports likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the consensus of all available predictions points to a benign impact, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -6.038 | Likely Benign | 0.079 | Likely Benign | Likely Benign | 0.086 | Likely Benign | -0.78 | Neutral | 0.058 | Benign | 0.015 | Benign | 2.78 | Benign | 0.29 | Tolerated | 3.77 | 5 | 0 | -1 | -1.9 | 31.01 | ||||||||||||||||||||||||||||||
| c.3136C>G | P1046A 2D  AIThe SynGAP1 missense variant P1046A is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443688‑C‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote). Only FATHMM predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443688-C-G | 1 | 6.20e-7 | -3.246 | Likely Benign | 0.048 | Likely Benign | Likely Benign | 0.041 | Likely Benign | -1.67 | Neutral | 0.001 | Benign | 0.008 | Benign | 2.39 | Pathogenic | 0.29 | Tolerated | 3.77 | 5 | -1 | 1 | 3.4 | -26.04 | |||||||||||||||||||||||||||
| c.2567A>G | N856S 2D  AIThe SynGAP1 missense variant N856S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443119‑A‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Those that predict a pathogenic outcome are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote) also benign; Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign effect, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443119-A-G | 2 | 1.24e-6 | -2.104 | Likely Benign | 0.064 | Likely Benign | Likely Benign | 0.040 | Likely Benign | -1.54 | Neutral | 0.901 | Possibly Damaging | 0.535 | Possibly Damaging | 4.16 | Benign | 0.30 | Tolerated | 3.88 | 3 | 1 | 1 | 2.7 | -27.03 | |||||||||||||||||||||||||||
| c.2591C>T | A864V 2D  AIThe SynGAP1 missense variant A864V is listed in ClinVar with an uncertain significance (ClinVar ID 655662.0) and is observed in gnomAD (ID 6‑33443143‑C‑T). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Tools that predict a pathogenic outcome are polyPhen‑2 HumDiv and FATHMM. High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign, and Foldetta results are unavailable. Based on the preponderance of evidence, the variant is most likely benign, which does not contradict its current ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 2 | 6-33443143-C-T | 6 | 3.72e-6 | -4.749 | Likely Benign | 0.126 | Likely Benign | Likely Benign | 0.038 | Likely Benign | -1.35 | Neutral | 0.767 | Possibly Damaging | 0.119 | Benign | 2.45 | Pathogenic | 0.30 | Tolerated | 3.82 | 4 | 0 | 0 | 2.4 | 28.05 | |||||||||||||||||||||||||||
| c.2684G>A | S895N 2D  AIThe SynGAP1 missense variant S895N is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is “Likely Benign.” High‑accuracy assessments show AlphaMissense‑Optimized as benign, and the SGM‑Consensus (majority vote) also leans benign. No Foldetta (protein‑folding stability) result is available, so it does not influence the assessment. Overall, the preponderance of predictions indicates the variant is most likely benign, which is consistent with its ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -6.399 | Likely Benign | 0.604 | Likely Pathogenic | Likely Benign | 0.118 | Likely Benign | -0.85 | Neutral | 0.991 | Probably Damaging | 0.988 | Probably Damaging | 2.64 | Benign | 0.30 | Tolerated | 4.32 | 4 | 1 | 1 | -2.7 | 27.03 | ||||||||||||||||||||||||||||||
| c.1436G>A | R479Q 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant R479Q is listed in ClinVar with an “Uncertain” significance and is present in gnomAD (variant ID 6‑33438468‑G‑A). Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, SIFT, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). In contrast, polyPhen‑2 HumDiv and HumVar both predict a pathogenic impact. Uncertain or inconclusive results come from FoldX, Rosetta, Foldetta, and ESM1b. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus also as likely benign, while Foldetta remains uncertain. Overall, the majority of evidence points to a benign effect, and this consensus does not contradict the ClinVar “Uncertain” status; thus the variant is most likely benign based on current predictions. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | 6-33438468-G-A | 7 | 4.34e-6 | -7.109 | In-Between | 0.259 | Likely Benign | Likely Benign | 0.191 | Likely Benign | 0.54 | Ambiguous | 0.1 | 0.57 | Ambiguous | 0.56 | Ambiguous | 0.49 | Likely Benign | -1.16 | Neutral | 1.000 | Probably Damaging | 0.991 | Probably Damaging | 3.42 | Benign | 0.31 | Tolerated | 3.39 | 32 | 1 | 1 | 1.0 | -28.06 | |||||||||||||||||
| c.1730C>G | A577G 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A577G is listed in ClinVar as Benign (ClinVar ID 1010280.0) and is present in gnomAD (ID 6‑33440782‑C‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. Predictions that are inconclusive (FoldX, Rosetta, Foldetta, premPS) are treated as unavailable. High‑accuracy methods give a benign verdict: AlphaMissense‑Optimized is benign, the SGM‑Consensus is Likely Benign, and Foldetta is uncertain. Overall, the majority of reliable predictions support a benign impact, which is consistent with the ClinVar status and does not contradict it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Benign/Likely benign | 2 | 6-33440782-C-G | 1 | 6.20e-7 | -5.717 | Likely Benign | 0.268 | Likely Benign | Likely Benign | 0.443 | Likely Benign | 0.83 | Ambiguous | 0.0 | 1.02 | Ambiguous | 0.93 | Ambiguous | 0.86 | Ambiguous | -1.84 | Neutral | 0.997 | Probably Damaging | 0.990 | Probably Damaging | -1.31 | Pathogenic | 0.31 | Tolerated | 3.37 | 34 | 1 | 0 | -2.2 | -14.03 | 158.7 | 23.6 | 0.0 | 0.0 | 0.0 | 0.0 | X | Potentially Benign | Ala577 is located near the end and outer surface of an α-helix (res. Arg563-Glu578), where its methyl group does not form any particular interactions in the WT simulations. The introduced residue, glycine, is known as an “α-helix breaker.” However, the residue swap caused only minor helix shortening in one of the replica simulations for the variant system. Regardless, the residue swap seems to be well tolerated based on the variant simulations. | ||||||||
| c.2743G>A | G915S 2D  AIThe SynGAP1 missense variant G915S is listed in ClinVar as Benign (ClinVar ID 652083.0) and is present in the gnomAD database (gnomAD ID 6‑33443295‑G‑A). Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only polyPhen‑2 HumDiv reports a pathogenic prediction, representing the sole discordant signal. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is Likely Benign. No Foldetta stability result is available, so it does not influence the assessment. Overall, the majority of evidence indicates that the variant is most likely benign, and this conclusion is consistent with its ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33443295-G-A | 9 | 5.58e-6 | -3.557 | Likely Benign | 0.083 | Likely Benign | Likely Benign | 0.050 | Likely Benign | -0.88 | Neutral | 0.801 | Possibly Damaging | 0.201 | Benign | 2.73 | Benign | 0.31 | Tolerated | 3.77 | 5 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||
| c.3487C>G | H1163D 2D  AISynGAP1 missense variant H1163D is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools show a split: benign calls come from REVEL, SIFT, ESM1b, and FATHMM, while pathogenic calls come from PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. High‑accuracy assessments are inconclusive: AlphaMissense‑Optimized rates the variant as uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields a tie and is therefore unavailable, and Foldetta folding‑stability analysis is not provided. With an equal number of benign and pathogenic predictions and no decisive high‑accuracy evidence, the variant remains ambiguous. Thus, it is most likely neither clearly benign nor pathogenic, and this uncertainty aligns with its ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Uncertain | 1 | -2.107 | Likely Benign | 0.949 | Likely Pathogenic | Ambiguous | 0.476 | Likely Benign | -2.60 | Deleterious | 0.991 | Probably Damaging | 0.991 | Probably Damaging | 5.44 | Benign | 0.31 | Tolerated | 3.88 | 3 | 1 | -1 | -0.3 | -22.05 | |||||||||||||||||||||||||||||||
| c.1918A>T | T640S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T640S is listed in ClinVar as Benign (ClinVar ID 2980241.0) and is present in the gnomAD database (gnomAD ID 6‑33441177‑A‑T). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive result is from FoldX, which is treated as unavailable. High‑accuracy assessments confirm benignity: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, is benign. Overall, the variant is most likely benign, and this conclusion is consistent with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Benign | 1 | 6-33441177-A-T | 1 | 6.20e-7 | -2.371 | Likely Benign | 0.062 | Likely Benign | Likely Benign | 0.088 | Likely Benign | -0.78 | Ambiguous | 0.1 | 0.43 | Likely Benign | -0.18 | Likely Benign | -0.30 | Likely Benign | 0.92 | Neutral | 0.000 | Benign | 0.001 | Benign | 3.60 | Benign | 0.33 | Tolerated | 3.37 | 30 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||
| c.2369C>G | T790S 2D  AIThe SynGAP1 missense variant T790S is listed in ClinVar (ID 1020340.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). In contrast, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM predict a pathogenic impact. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus also as likely benign; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this is consistent with the ClinVar “Uncertain” status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | Uncertain | 1 | -3.914 | Likely Benign | 0.123 | Likely Benign | Likely Benign | 0.134 | Likely Benign | -1.83 | Neutral | 0.997 | Probably Damaging | 0.989 | Probably Damaging | 2.39 | Pathogenic | 0.33 | Tolerated | 3.64 | 6 | 1 | 1 | -0.1 | -14.03 | |||||||||||||||||||||||||||||
| c.3134C>G | A1045G 2D  AIThe SynGAP1 missense variant A1045G is reported in ClinVar as Benign (ClinVar ID 416778.0) and is present in the gnomAD database (gnomAD ID 6‑33443686‑C‑G). Prediction tools that assess pathogenicity all converge on a benign outcome: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the variant as benign. No tool in the dataset predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not available. Overall, the variant is most likely benign, and this prediction aligns with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign/Likely benign | 7 | 6-33443686-C-G | 1407 | 8.72e-4 | -3.246 | Likely Benign | 0.075 | Likely Benign | Likely Benign | 0.024 | Likely Benign | -1.21 | Neutral | 0.224 | Benign | 0.066 | Benign | 2.64 | Benign | 0.33 | Tolerated | 3.77 | 5 | 1 | 0 | -2.2 | -14.03 | 10.1016/j.ajhg.2020.11.011 | ||||||||||||||||||||||||||
| c.3179G>T | G1060V 2D  AIThe SynGAP1 missense variant G1060V is listed in ClinVar as benign (ClinVar ID 1345112.0) and is observed in gnomAD (6‑33443731‑G‑T). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Two tools, polyPhen‑2 HumDiv and HumVar, predict a pathogenic effect. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) classifies the variant as likely benign, and AlphaMissense‑Optimized also reports a benign outcome. No Foldetta stability assessment is available for this variant. Overall, the majority of evidence points to a benign impact, aligning with the ClinVar designation and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33443731-G-T | 1 | 6.22e-7 | -6.966 | Likely Benign | 0.103 | Likely Benign | Likely Benign | 0.369 | Likely Benign | -0.73 | Neutral | 0.986 | Probably Damaging | 0.728 | Possibly Damaging | 2.63 | Benign | 0.33 | Tolerated | 4.32 | 2 | -1 | -3 | 4.6 | 42.08 | |||||||||||||||||||||||||||
| c.3494C>T | S1165L 2D  AIThe SynGAP1 missense variant S1165L is listed in ClinVar with an uncertain significance (ClinVar ID 225899.0) and is not reported in gnomAD. Functional prediction tools show a split: benign predictions come from REVEL, PROVEAN, SIFT, ESM1b, and FATHMM, while pathogenic predictions arise from polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. Grouping by consensus, the benign‑predicted tools outnumber the pathogenic ones. High‑accuracy assessments further clarify the picture: the SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, leans toward benign (Likely Benign); AlphaMissense‑Optimized remains uncertain, and Foldetta data are unavailable. Overall, the majority of evidence points to a benign effect, aligning with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 2 | -2.984 | Likely Benign | 0.793 | Likely Pathogenic | Ambiguous | 0.166 | Likely Benign | -2.01 | Neutral | 0.998 | Probably Damaging | 0.992 | Probably Damaging | 2.60 | Benign | 0.33 | Tolerated | 3.88 | 3 | -3 | -2 | 4.6 | 26.08 | 10.1016/j.ajhg.2020.11.011 | |||||||||||||||||||||||||||||
| c.2277G>A | M759I 2D  AIThe SynGAP1 missense variant M759I is listed in ClinVar (ID 3686687.0) with an “Uncertain” status and is present in gnomAD (variant ID 6‑33441742‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Only polyPhen‑2 HumDiv predicts a pathogenic outcome, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this is consistent with the ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33441742-G-A | 1 | 6.20e-7 | -4.058 | Likely Benign | 0.393 | Ambiguous | Likely Benign | 0.075 | Likely Benign | -0.88 | Neutral | 0.454 | Possibly Damaging | 0.192 | Benign | 2.83 | Benign | 0.34 | Tolerated | 3.99 | 5 | 1 | 2 | 2.6 | -18.03 | |||||||||||||||||||||||||||
| c.3338G>A | G1113D 2D  AIThe SynGAP1 missense variant G1113D is listed in ClinVar with an uncertain significance (ClinVar ID 2766136.0) and is present in gnomAD (variant ID 6‑33443890‑G‑A). Functional prediction tools that agree on a benign outcome include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM—all of which classify the substitution as benign. AlphaMissense‑Optimized also predicts a benign effect, whereas AlphaMissense‑Default remains uncertain. No tool predicts a pathogenic effect. The high‑accuracy consensus methods reinforce this benign assessment: the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) reports “Likely Benign,” and AlphaMissense‑Optimized also indicates benign. Foldetta, a protein‑folding stability predictor combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the collective evidence strongly supports a benign impact, and this conclusion does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443890-G-A | -4.638 | Likely Benign | 0.354 | Ambiguous | Likely Benign | 0.061 | Likely Benign | -0.72 | Neutral | 0.029 | Benign | 0.017 | Benign | 2.58 | Benign | 0.34 | Tolerated | 4.32 | 2 | -1 | 1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||
| c.1888A>G | I630V 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I630V is listed in ClinVar as Benign and is present in gnomAD (variant ID 6‑33440940‑A‑G). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome; all other tools (FoldX, Rosetta, Foldetta, premPS, ESM1b) return uncertain or inconclusive results. The high‑accuracy consensus (SGM Consensus) derived from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN yields a benign majority (2 benign vs. 1 pathogenic, 1 uncertain). AlphaMissense‑Optimized also predicts benign. Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, is uncertain. Taken together, the overwhelming majority of predictions support a benign effect, and this conclusion aligns with the ClinVar designation. Thus, the variant is most likely benign, with no contradiction to the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Benign/Likely benign | 4 | 6-33440940-A-G | 59 | 3.66e-5 | -7.264 | In-Between | 0.145 | Likely Benign | Likely Benign | 0.143 | Likely Benign | 1.33 | Ambiguous | 0.0 | 0.94 | Ambiguous | 1.14 | Ambiguous | 0.64 | Ambiguous | -0.38 | Neutral | 0.018 | Benign | 0.011 | Benign | -1.37 | Pathogenic | 0.35 | Tolerated | 3.37 | 34 | 4 | 3 | -0.3 | -14.03 | 235.0 | 26.2 | -0.1 | 0.0 | -0.3 | 0.1 | X | Potentially Benign | The sec-butyl side chain of Ile630, located in an α helix (res. Glu617-Asn635), packs with hydrophobic residues (e.g., Phe594, Leu633, Ile626, Ile602) in the hydrophobic inter-helix space between two α helices (res. Glu617-Asn635 and res. Glu582-Met603).In the variant simulations, the iso-propyl side chain of Val630, which shares a similar size and physicochemical properties with Ile630 in the WT, maintains similar interactions in the inter-helix space. Although no negative structural effects are observed during the simulations, the implications of the residue swap on the complex formation with the GTPase, due to its location, cannot be investigated using solvent-only simulations. | |||||||||
| c.2270G>C | G757A 2D  AIThe SynGAP1 missense change G757A is catalogued in ClinVar (ID 3635272.0) with an uncertain significance designation and is not reported in gnomAD. Functional prediction algorithms uniformly classify the variant as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool in the dataset predicts pathogenicity. High‑accuracy consensus methods corroborate this view: the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign effect, and AlphaMissense‑Optimized also predicts benign. The Foldetta stability assessment is unavailable for this variant. Taken together, the evidence overwhelmingly supports a benign interpretation, which is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.626 | Likely Benign | 0.091 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -0.45 | Neutral | 0.267 | Benign | 0.127 | Benign | 2.73 | Benign | 0.35 | Tolerated | 1 | 0 | 2.2 | 14.03 | ||||||||||||||||||||||||||||||||
| c.3250C>G | P1084A 2D  AIThe SynGAP1 missense variant P1084A is listed in ClinVar (ID 2827308.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (which itself is “Likely Benign”). In contrast, PROVEAN and polyPhen‑2 HumDiv predict a pathogenic impact. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; a Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -3.928 | Likely Benign | 0.066 | Likely Benign | Likely Benign | 0.114 | Likely Benign | -2.54 | Deleterious | 0.649 | Possibly Damaging | 0.157 | Benign | 4.05 | Benign | 0.35 | Tolerated | 3.77 | 5 | -1 | 1 | 3.4 | -26.04 | ||||||||||||||||||||||||||||||
| c.382C>A | P128T 2D  AIThe SynGAP1 missense variant P128T is listed in ClinVar with an “Uncertain” status (ClinVar ID 2801315.0) and is present in gnomAD (ID 6‑33432247‑C‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote) as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33432247-C-A | 1 | 6.20e-7 | -4.217 | Likely Benign | 0.267 | Likely Benign | Likely Benign | 0.075 | Likely Benign | -0.96 | Neutral | 0.952 | Possibly Damaging | 0.500 | Possibly Damaging | 4.19 | Benign | 0.35 | Tolerated | 3.74 | 4 | -1 | 0 | 0.9 | 3.99 | |||||||||||||||||||||||||||
| c.2651G>A | R884Q 2D  AIThe SynGAP1 missense variant R884Q is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443203‑G‑A). All available in silico predictors agree on a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are not reported, so they are unavailable. Overall, the computational evidence strongly supports a benign classification, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 2 | 6-33443203-G-A | 5 | 3.10e-6 | -3.785 | Likely Benign | 0.128 | Likely Benign | Likely Benign | 0.055 | Likely Benign | -0.42 | Neutral | 0.012 | Benign | 0.004 | Benign | 2.62 | Benign | 0.36 | Tolerated | 4.32 | 4 | 1 | 1 | 1.0 | -28.06 | |||||||||||||||||||||||||||
| c.2812G>A | G938R 2D  AIThe SynGAP1 missense variant G938R is listed in ClinVar (ID 1019898.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence (seven benign versus three pathogenic predictions) supports a benign classification. This consensus does not contradict the ClinVar “Uncertain” designation, which remains unresolved. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -5.271 | Likely Benign | 0.732 | Likely Pathogenic | Likely Benign | 0.141 | Likely Benign | -1.11 | Neutral | 0.999 | Probably Damaging | 0.985 | Probably Damaging | 2.74 | Benign | 0.36 | Tolerated | 3.77 | 5 | -3 | -2 | -4.1 | 99.14 | ||||||||||||||||||||||||||||||
| c.2900G>A | R967Q 2D  AIThe SynGAP1 missense variant R967Q is listed in ClinVar as Benign (ClinVar ID 536992.0) and is present in gnomAD (6‑33443452‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Tools that predict a pathogenic effect are PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, aligning with the ClinVar classification and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign/Likely benign | 2 | 6-33443452-G-A | 31 | 1.92e-5 | -3.057 | Likely Benign | 0.080 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -0.01 | Neutral | 0.994 | Probably Damaging | 0.626 | Possibly Damaging | 4.21 | Benign | 0.36 | Tolerated | 4.32 | 2 | 1 | 1 | 1.0 | -28.06 | |||||||||||||||||||||||||||
| c.3293G>A | S1098N 2D  AIThe SynGAP1 missense variant S1098N is listed in ClinVar (ID 864704.0) with an “Uncertain” clinical significance and is present in gnomAD (variant ID 6‑33443845‑G‑A). All evaluated in‑silico predictors agree on a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign classification, which is consistent with the ClinVar “Uncertain” status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 2 | 6-33443845-G-A | 6 | 3.89e-6 | -5.120 | Likely Benign | 0.156 | Likely Benign | Likely Benign | 0.063 | Likely Benign | -0.58 | Neutral | 0.369 | Benign | 0.120 | Benign | 2.76 | Benign | 0.36 | Tolerated | 3.77 | 5 | 1 | 1 | -2.7 | 27.03 | |||||||||||||||||||||||||||
| c.3161G>A | G1054D 2D  AISynGAP1 missense variant G1054D is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and ESM1b, while AlphaMissense‑Default remains uncertain. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized scores benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields benign. Foldetta, a protein‑folding stability method, has no available result for this variant. Overall, the preponderance of evidence indicates the variant is most likely benign, which does not contradict the current ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Uncertain | 1 | -10.385 | Likely Pathogenic | 0.351 | Ambiguous | Likely Benign | 0.279 | Likely Benign | -0.26 | Neutral | 0.818 | Possibly Damaging | 0.266 | Benign | 4.07 | Benign | 0.37 | Tolerated | 3.77 | 5 | 1 | -1 | -3.1 | 58.04 | |||||||||||||||||||||||||||||||
| c.1973G>A | G658D 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant G658D is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6-33441232‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, FoldX, premPS, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Only PROVEAN predicts a pathogenic outcome, while Rosetta, Foldetta, ESM1b, and AlphaMissense‑Default are inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, and Foldetta, which integrates FoldX‑MD and Rosetta, is also inconclusive. Overall, the preponderance of evidence points to a benign effect, and this does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Uncertain | 1 | 6-33441232-G-A | 3 | 1.86e-6 | -7.786 | In-Between | 0.442 | Ambiguous | Likely Benign | 0.144 | Likely Benign | -0.40 | Likely Benign | 0.1 | -0.59 | Ambiguous | -0.50 | Ambiguous | 0.46 | Likely Benign | -2.64 | Deleterious | 0.008 | Benign | 0.005 | Benign | 3.53 | Benign | 0.38 | Tolerated | 3.39 | 24 | 1 | -1 | -3.1 | 58.04 | 219.8 | -84.3 | 0.0 | 0.0 | 0.2 | 0.1 | X | Potentially Pathogenic | Gly658, located on the outer surface of an α helix (res. Ser641-Glu666), weakens the helix integrity at that spot, which is necessary for the kink in the middle of the long helix. In the variant simulations, the carboxylic acid side chain of Asp658 is on the surface of the α helix and is not involved in any interactions. However, aspartate is not as effective a breaker of the secondary structure element as glycine, which may lead to misfolding. | |||||||||
| c.2282G>C | R761P 2D  AIThe SynGAP1 missense variant R761P is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33441747‑G‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of predictions point to a benign impact, and this is consistent with the ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 3 | 6-33441747-G-C | 1 | 6.20e-7 | -5.091 | Likely Benign | 0.640 | Likely Pathogenic | Likely Benign | 0.201 | Likely Benign | -1.89 | Neutral | 0.999 | Probably Damaging | 0.968 | Probably Damaging | 2.69 | Benign | 0.38 | Tolerated | 3.99 | 5 | 0 | -2 | 2.9 | -59.07 | |||||||||||||||||||||||||||
| c.1260T>G | F420L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F420L is listed in ClinVar (ID 1397885.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM. Those that predict a pathogenic effect comprise premPS, PROVEAN, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Stability‑based methods (FoldX, Rosetta, Foldetta) yield inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, the SGM Consensus also as pathogenic, while Foldetta remains uncertain. Overall, the majority of evidence points toward a pathogenic impact, which does not contradict the ClinVar “Uncertain” status but suggests the variant is more likely pathogenic rather than benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -8.432 | Likely Pathogenic | 0.998 | Likely Pathogenic | Likely Pathogenic | 0.146 | Likely Benign | 1.76 | Ambiguous | 0.0 | 1.41 | Ambiguous | 1.59 | Ambiguous | 1.04 | Destabilizing | -5.39 | Deleterious | 0.009 | Benign | 0.005 | Benign | 4.22 | Benign | 0.39 | Tolerated | 3.37 | 29 | 2 | 0 | 1.0 | -34.02 | 231.1 | 13.2 | 0.0 | 0.0 | -0.1 | 0.0 | X | Potentially Benign | In the WT, the phenyl ring of the Phe420 side chain, located on an α helix (res. Met414-Glu436), packs against hydrophobic residues in the interhelix area of the GAP domain (e.g., Leu689, Leu714, Leu717, Leu718). In the variant simulations, the iso-butyl side chain of Leu420 also packs into the hydrophobic inter-helix niche, but due to its smaller size, the resulting steric interactions are not as favorable as with phenylalanine. In short, the residue swap does not cause severe effects on the protein structure based on the variant simulations. | |||||||||||
| c.2506A>G | S836G 2D  AIThe SynGAP1 missense variant S836G is listed in ClinVar (ID 537003.0) with an uncertain significance annotation and is observed in the gnomAD database (variant ID 6‑33443058‑A‑G). Consensus from multiple in silico predictors indicates a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all classify the substitution as benign. No tool in the dataset predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized reports a benign outcome, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is labeled Likely Benign. Foldetta, a protein‑folding stability predictor, did not return a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign classification, which does not conflict with the ClinVar uncertain designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443058-A-G | 4 | 2.48e-6 | -4.749 | Likely Benign | 0.112 | Likely Benign | Likely Benign | 0.066 | Likely Benign | -1.65 | Neutral | 0.006 | Benign | 0.019 | Benign | 2.54 | Benign | 0.39 | Tolerated | 3.77 | 5 | 1 | 0 | 0.4 | -30.03 | |||||||||||||||||||||||||||
| c.3152G>A | G1051D 2D  AISynGAP1 missense variant G1051D is listed in ClinVar as Benign and is present in gnomAD (variant ID 6‑33443704‑G‑A). Prediction tools that classify the variant as benign include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict pathogenicity are polyPhen‑2 HumDiv, ESM1b, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive (two benign versus two pathogenic votes), and Foldetta stability analysis is unavailable. Overall, the balance of evidence favors a benign effect, consistent with the ClinVar annotation and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Benign | 1 | 6-33443704-G-A | 2 | 1.24e-6 | -9.379 | Likely Pathogenic | 0.311 | Likely Benign | Likely Benign | 0.445 | Likely Benign | -0.31 | Neutral | 0.761 | Possibly Damaging | 0.239 | Benign | -0.74 | Pathogenic | 0.39 | Tolerated | 3.77 | 5 | -1 | 1 | -3.1 | 58.04 | ||||||||||||||||||||||||||||
| c.3846G>C | E1282D 2D  AIThe SynGAP1 missense variant E1282D is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6-33447894-G-C). All available in silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic prediction. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also indicates a likely benign outcome. High‑accuracy assessments confirm this: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus (majority vote) is likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant, so its status is unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, which is consistent with the ClinVar “Uncertain” classification rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33447894-G-C | 1 | 6.44e-7 | -3.879 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.104 | Likely Benign | -1.26 | Neutral | 0.112 | Benign | 0.036 | Benign | 2.70 | Benign | 0.39 | Tolerated | 3.77 | 5 | 3 | 2 | 0.0 | -14.03 | |||||||||||||||||||||||||||
| c.2282G>A | R761Q 2D  AIThe SynGAP1 missense variant R761Q is listed in ClinVar (ID 2882770.0) with an “Uncertain” status and is present in gnomAD (6‑33441747‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are not available. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33441747-G-A | 11 | 6.81e-6 | -4.187 | Likely Benign | 0.202 | Likely Benign | Likely Benign | 0.191 | Likely Benign | -0.63 | Neutral | 0.996 | Probably Damaging | 0.878 | Possibly Damaging | 2.75 | Benign | 0.40 | Tolerated | 3.99 | 5 | 1 | 1 | 1.0 | -28.06 | |||||||||||||||||||||||||||
| c.745G>A | A249T 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant A249T is listed in ClinVar (ID 1031675.0) with an uncertain significance annotation and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, SIFT, ESM1b, and FATHMM, whereas polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default predict a pathogenic outcome. Predictions that are inconclusive are FoldX, Rosetta, Foldetta, and AlphaMissense‑Optimized. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as likely benign, and Foldetta (combining FoldX‑MD and Rosetta) as uncertain. Overall, the balance of evidence favors a benign interpretation, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | Uncertain | 1 | -3.564 | Likely Benign | 0.805 | Likely Pathogenic | Ambiguous | 0.487 | Likely Benign | 1.50 | Ambiguous | 0.6 | 1.39 | Ambiguous | 1.45 | Ambiguous | 0.30 | Likely Benign | -0.96 | Neutral | 0.990 | Probably Damaging | 0.815 | Possibly Damaging | 5.65 | Benign | 0.40 | Tolerated | 3.39 | 15 | 1 | 0 | -2.5 | 30.03 | 214.5 | -43.3 | 0.0 | 0.0 | 0.5 | 0.2 | X | Potentially Benign | The methyl group of Ala249, located on the surface of an α helix (res. Ala236-Val250) facing an anti-parallel β sheet strand (res. Ile205-Val209), packs against nearby hydrophobic residues such as Leu200, Leu246, and Val250. In the variant simulations, the hydroxyl group of Thr249, which is not suitable for hydrophobic packing, forms a stable hydrogen bond with the backbone carbonyl of Asn245 in the same helix. Although this interaction could theoretically weaken the structural integrity of the α helix, this destabilizing effect is not observed in the variant simulations. | |||||||||||
| c.971G>A | R324Q 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant R324Q is listed in ClinVar with an uncertain significance (ClinVar ID 2572558.0) and is present in gnomAD (ID 6‑33437876‑G‑A). Prediction tools that classify the variant as benign include REVEL, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict pathogenicity are premPS, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. The high‑accuracy AlphaMissense‑Optimized score is benign, and the SGM Consensus—derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN—also indicates a likely benign outcome. Protein‑stability predictions from FoldX, Rosetta, and the combined Foldetta method are all uncertain. Overall, the consensus of available computational evidence points to a benign effect for R324Q, which is consistent with its ClinVar status of uncertain significance rather than a pathogenic designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 3 | 6-33437876-G-A | 3 | 1.86e-6 | -5.001 | Likely Benign | 0.173 | Likely Benign | Likely Benign | 0.307 | Likely Benign | 0.56 | Ambiguous | 0.1 | 0.63 | Ambiguous | 0.60 | Ambiguous | 1.02 | Destabilizing | -1.17 | Neutral | 0.999 | Probably Damaging | 0.994 | Probably Damaging | 1.92 | Pathogenic | 0.41 | Tolerated | 3.39 | 22 | 1 | 1 | 1.0 | -28.06 | |||||||||||||||||
| c.1405G>A | A469T 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant A469T is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools cluster into two groups: benign predictions come from premPS, PROVEAN, SIFT, and AlphaMissense‑Optimized; pathogenic predictions arise from REVEL, FoldX, polyPhen‑2 (HumDiv and HumVar), ESM1b, FATHMM, AlphaMissense‑Default, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN), and the Foldetta stability assessment (combining FoldX‑MD and Rosetta). The high‑accuracy subset shows AlphaMissense‑Optimized as benign, whereas SGM Consensus and Foldetta both predict pathogenic. Overall, the majority of evidence supports a pathogenic effect, and this conclusion does not conflict with the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Uncertain | 1 | -9.540 | Likely Pathogenic | 0.723 | Likely Pathogenic | Likely Benign | 0.527 | Likely Pathogenic | 2.26 | Destabilizing | 0.1 | 1.90 | Ambiguous | 2.08 | Destabilizing | 0.34 | Likely Benign | -1.46 | Neutral | 0.994 | Probably Damaging | 0.986 | Probably Damaging | -1.21 | Pathogenic | 0.42 | Tolerated | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||
| c.2818G>A | G940S 2D  AIThe SynGAP1 missense variant G940S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443370‑G‑A). All available in silico predictors agree on a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” High‑accuracy tools reinforce this view: AlphaMissense‑Optimized predicts benign, and the SGM‑Consensus likewise indicates a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no reported result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign classification, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443370-G-A | 1 | 6.20e-7 | -5.451 | Likely Benign | 0.084 | Likely Benign | Likely Benign | 0.135 | Likely Benign | 0.45 | Neutral | 0.409 | Benign | 0.253 | Benign | 2.77 | Benign | 0.44 | Tolerated | 3.77 | 5 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||
| c.958G>A | V320I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V320I is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that uniformly indicate a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while Rosetta remains inconclusive. High‑accuracy assessments corroborate the benign trend: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is “Likely Benign”; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, also predicts benign. Overall, the preponderance of evidence points to a benign effect for V320I, and this conclusion does not conflict with the current ClinVar designation of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 1 | -5.220 | Likely Benign | 0.111 | Likely Benign | Likely Benign | 0.027 | Likely Benign | -0.27 | Likely Benign | 0.2 | 0.66 | Ambiguous | 0.20 | Likely Benign | 0.01 | Likely Benign | -0.21 | Neutral | 0.198 | Benign | 0.114 | Benign | 1.77 | Pathogenic | 0.45 | Tolerated | 3.38 | 23 | 3 | 4 | 0.3 | 14.03 | ||||||||||||||||||||
| c.2300T>C | I767T 2D  AIThe SynGAP1 missense variant I767T is listed in ClinVar (ID 1044161.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only polyPhen‑2 HumDiv flags the variant as pathogenic, creating a single discordant prediction. The high‑accuracy consensus from SGM (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields a “Likely Benign” classification, and AlphaMissense‑Optimized also reports benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -3.749 | Likely Benign | 0.252 | Likely Benign | Likely Benign | 0.138 | Likely Benign | -0.78 | Neutral | 0.625 | Possibly Damaging | 0.249 | Benign | 4.12 | Benign | 0.46 | Tolerated | 3.64 | 6 | 0 | -1 | -5.2 | -12.05 | ||||||||||||||||||||||||||||||
| c.1729G>A | A577T 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant A577T is listed in ClinVar as benign (ClinVar ID 2195056.0) and is present in gnomAD (ID 6‑33440781‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign, SGM‑Consensus as likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as uncertain. No other high‑confidence stability predictions are available. Overall, the consensus of the available predictions indicates that the variant is most likely benign, which aligns with its ClinVar classification and does not contradict the reported status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Benign | 1 | 6-33440781-G-A | 6 | 3.72e-6 | -5.311 | Likely Benign | 0.322 | Likely Benign | Likely Benign | 0.427 | Likely Benign | 0.86 | Ambiguous | 0.1 | 0.54 | Ambiguous | 0.70 | Ambiguous | 0.54 | Ambiguous | -1.47 | Neutral | 0.999 | Probably Damaging | 0.987 | Probably Damaging | -1.31 | Pathogenic | 0.47 | Tolerated | 3.37 | 34 | 1 | 0 | -2.5 | 30.03 | 191.9 | -43.4 | 0.0 | 0.0 | 0.7 | 0.1 | X | Potentially Benign | Ala577 is located near the end and outer surface of an α-helix (res. Arg563-Glu578), where its methyl group does not form any particular interactions in the WT simulations. In the variant simulations, the hydroxyl group of the Thr577 side chain hydrogen bonds with the backbone atoms of Arg573 and Lys574 within the same helix, which has the potential to weaken the stability of the secondary structure element. Regardless, the residue swap seems to be well tolerated based on the variant simulations. | ||||||||
| c.2401G>A | G801S 2D  AIThe SynGAP1 missense variant G801S is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. All available in‑silico predictors classify the change as benign: SGM‑Consensus (Likely Benign), REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this benign prediction: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is also Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no reported result for this variant. Overall, the evidence strongly supports a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | Uncertain | 1 | -3.665 | Likely Benign | 0.087 | Likely Benign | Likely Benign | 0.039 | Likely Benign | -0.41 | Neutral | 0.009 | Benign | 0.019 | Benign | 2.76 | Benign | 0.48 | Tolerated | 4.32 | 2 | 0 | 1 | -0.4 | 30.03 | |||||||||||||||||||||||||||||
| c.2932C>T | P978S 2D  AIThe SynGAP1 missense variant P978S is listed in ClinVar (ID 3379672.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign. Only polyPhen‑2 HumDiv indicates a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, classifies the variant as “Likely Benign.” High‑accuracy assessments further support a benign interpretation: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is likely benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the preponderance of predictions points to a benign effect, which is consistent with the ClinVar “Uncertain” status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -3.913 | Likely Benign | 0.151 | Likely Benign | Likely Benign | 0.085 | Likely Benign | -1.07 | Neutral | 0.481 | Possibly Damaging | 0.220 | Benign | 4.22 | Benign | 0.48 | Tolerated | 1 | -1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||||
| c.3178G>A | G1060S 2D  AIThe SynGAP1 missense variant G1060S is listed in ClinVar with an uncertain significance (ClinVar ID 1512003.0) and is present in gnomAD (variant ID 6‑33443730‑G‑A). All evaluated in‑silico predictors classify the change as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this benign view: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence overwhelmingly supports a benign effect, which is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443730-G-A | -4.759 | Likely Benign | 0.082 | Likely Benign | Likely Benign | 0.376 | Likely Benign | -0.08 | Neutral | 0.271 | Benign | 0.054 | Benign | 2.69 | Benign | 0.49 | Tolerated | 4.32 | 2 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||||
| c.2116G>A | E706K 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant E706K is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, and FATHMM all classify the change as benign. In contrast, ESM1b and AlphaMissense‑Default predict a pathogenic impact. Tools that return uncertain results—FoldX, Rosetta, Foldetta, and AlphaMissense‑Optimized—do not provide decisive evidence. The SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is inconclusive (two pathogenic versus two benign calls). High‑accuracy assessments are likewise ambiguous: AlphaMissense‑Optimized is uncertain, Foldetta is uncertain, and the SGM Consensus remains inconclusive. Overall, the preponderance of evidence points to a benign effect, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Uncertain | 1 | -10.519 | Likely Pathogenic | 0.833 | Likely Pathogenic | Ambiguous | 0.080 | Likely Benign | 1.17 | Ambiguous | 0.1 | 0.51 | Ambiguous | 0.84 | Ambiguous | 0.08 | Likely Benign | -1.51 | Neutral | 0.345 | Benign | 0.028 | Benign | 4.15 | Benign | 0.52 | Tolerated | 3.47 | 10 | 0 | 1 | -0.4 | -0.94 | 187.1 | 49.2 | 0.0 | 0.0 | 0.4 | 0.1 | X | Uncertain | The carboxylate side chain of Glu706, located at the end and outer surface of an α-helix (res. Thr704-Gly712), forms a salt bridge with Lys710 and a hydrogen bond with its own backbone amino group at the helix end in the WT simulations. Although Lys706 is unable to make these transient interactions in the variant simulations, there is no apparent negative effect on the protein structure due to the residue swap. However, because the model ends abruptly at the C-terminus, no definite conclusions can be drawn based on the simulations. | ||||||||||||
| c.2928T>G | F976L 2D  AIThe SynGAP1 missense variant F976L is listed in ClinVar (ID 624245.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, and FATHMM. Only AlphaMissense‑Default predicts a pathogenic outcome. The high‑accuracy consensus predictor SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. AlphaMissense‑Optimized is inconclusive, and no Foldetta (FoldX‑MD/Rosetta stability) result is available. Overall, the majority of evidence points to a benign impact, and this is consistent with the ClinVar designation of uncertainty rather than a definitive pathogenic claim. Thus, the variant is most likely benign, and its predictions do not contradict the current ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.432 | Likely Benign | 0.825 | Likely Pathogenic | Ambiguous | 0.212 | Likely Benign | -0.87 | Neutral | 0.264 | Benign | 0.102 | Benign | 4.20 | Benign | 0.53 | Tolerated | 4.32 | 2 | 2 | 0 | 1.0 | -34.02 | ||||||||||||||||||||||||||||||
| c.3508A>G | S1170G 2D  AIThe SynGAP1 missense variant S1170G is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on benign impact include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts pathogenicity, so the pathogenic group is empty. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also predicts benign, while Foldetta results are unavailable. Based on the collective predictions, the variant is most likely benign, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Uncertain | 1 | -4.288 | Likely Benign | 0.221 | Likely Benign | Likely Benign | 0.349 | Likely Benign | -0.81 | Neutral | 0.241 | Benign | 0.229 | Benign | 5.31 | Benign | 0.54 | Tolerated | 4.32 | 4 | 1 | 0 | 0.4 | -30.03 | |||||||||||||||||||||||||||||
| c.1586T>C | I529T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant I529T is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus “Likely Benign” call. Tools that predict a pathogenic outcome are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) also yields a benign prediction; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, predicts benign. Overall, the majority of evidence points to a benign effect, and this is consistent with the ClinVar “Uncertain” classification—there is no contradiction between the predictions and the current ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | -0.539 | Likely Benign | 0.336 | Likely Benign | Likely Benign | 0.343 | Likely Benign | 0.22 | Likely Benign | 0.2 | 0.16 | Likely Benign | 0.19 | Likely Benign | 0.17 | Likely Benign | 0.24 | Neutral | 0.872 | Possibly Damaging | 0.820 | Possibly Damaging | -1.23 | Pathogenic | 0.55 | Tolerated | 3.37 | 35 | 0 | -1 | -5.2 | -12.05 | 207.2 | 29.8 | 0.2 | 0.0 | 0.2 | 0.1 | X | Potentially Benign | Ile529 is located on an α-α loop between the two α-helices (res. Gly502-Tyr518 and Ala533-Val560). In the WT simulations, the sec-butyl side chain of Ile529 faces the membrane interface and shows no specific interactions. In the variant simulations, the hydroxyl group of Thr529 forms a hydrogen bond with the carboxylate side chain of Asp527, but no negative structural changes are observed. However, due to its location near the SynGAP-membrane interface, the effect of the residue swap cannot be fully addressed using the SynGAP solvent-only simulations. | |||||||||||
| c.2881C>T | H961Y 2D  AIThe SynGAP1 missense variant H961Y is listed in ClinVar with an uncertain significance (ClinVar ID 862637.0) and is present in gnomAD (ID 6‑33443433‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and ESM1b. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this is not in conflict with the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 2 | 6-33443433-C-T | 3 | 1.86e-6 | -8.051 | Likely Pathogenic | 0.157 | Likely Benign | Likely Benign | 0.102 | Likely Benign | -1.07 | Neutral | 0.716 | Possibly Damaging | 0.147 | Benign | 4.10 | Benign | 0.55 | Tolerated | 3.77 | 5 | 0 | 2 | 1.9 | 26.03 | |||||||||||||||||||||||||||
| c.3484C>T | P1162S 2D  AIThe SynGAP1 missense variant P1162S is listed in ClinVar (ID 2287942.0) with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, and FATHMM. Those that predict a pathogenic effect are PolyPhen‑2 HumDiv, PolyPhen‑2 HumVar, and AlphaMissense‑Default. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. AlphaMissense‑Optimized is uncertain, and Foldetta (a protein‑folding stability method combining FoldX‑MD and Rosetta outputs) has no available result for this variant. Overall, the majority of high‑accuracy and consensus predictions lean toward a benign impact. Thus, the variant is most likely benign, which is consistent with its ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.118 | Likely Benign | 0.913 | Likely Pathogenic | Ambiguous | 0.215 | Likely Benign | -1.93 | Neutral | 1.000 | Probably Damaging | 0.999 | Probably Damaging | 2.73 | Benign | 0.55 | Tolerated | 3.88 | 3 | 1 | -1 | 0.8 | -10.04 | ||||||||||||||||||||||||||||||
| c.1622C>G | A541G 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant A541G is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33438865‑C‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. The remaining tools (FoldX, Rosetta, Foldetta, premPS, AlphaMissense‑Default, and ESM1b) return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, and Foldetta is also inconclusive. Overall, the balance of evidence leans toward a benign impact, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Uncertain | 1 | 6-33438865-C-G | 2 | 1.24e-6 | -7.233 | In-Between | 0.341 | Ambiguous | Likely Benign | 0.421 | Likely Benign | 0.67 | Ambiguous | 0.0 | 0.94 | Ambiguous | 0.81 | Ambiguous | 0.76 | Ambiguous | -1.48 | Neutral | 0.999 | Probably Damaging | 0.995 | Probably Damaging | -1.31 | Pathogenic | 0.57 | Tolerated | 3.37 | 35 | 1 | 0 | -2.2 | -14.03 | 170.1 | 23.6 | 0.0 | 0.0 | 0.0 | 0.0 | X | Potentially Pathogenic | Ala541 is located on the outer surface of an α-helix (res. Ala533-Val560). The methyl group of Ala541 is on the surface and does not form any interactions. Glycine, known as an “α-helix breaker,” weakens the integrity of the helix. Indeed, in the variant simulations, the hydrogen bond formation between Gly541 and the backbone carbonyl of Ala537 is disrupted. | |||||||||
| c.670A>G | T224A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T224A is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33435521‑A‑G). Prediction tools that agree on a benign effect include REVEL, FoldX, polyPhen‑2 (HumDiv and HumVar), SIFT, FATHMM, and AlphaMissense‑Optimized. Those that predict a pathogenic effect are PROVEAN and AlphaMissense‑Default. The remaining tools (Rosetta, Foldetta, premPS, ESM1b) return uncertain or inconclusive results. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as pathogenic, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | PH | Uncertain | 3 | 6-33435521-A-G | 2 | 1.24e-6 | -7.379 | In-Between | 0.651 | Likely Pathogenic | Likely Benign | 0.464 | Likely Benign | 0.33 | Likely Benign | 0.1 | 1.05 | Ambiguous | 0.69 | Ambiguous | 0.91 | Ambiguous | -2.96 | Deleterious | 0.243 | Benign | 0.079 | Benign | 5.57 | Benign | 0.57 | Tolerated | 3.41 | 13 | 1 | 0 | 2.5 | -30.03 | 169.0 | 41.4 | -0.5 | 1.1 | -0.4 | 0.0 | X | X | Uncertain | The introduced residue Ala224 is located on the outer surface of an anti-parallel β sheet strand (res. Cys219-Thr224). Unlike the hydroxyl group of the Thr224 side chain in the WT model, the methyl side chain of Ala224 cannot form hydrogen bonds with nearby residues Ser204, Ser226, and Gly227. Without these hydrogen-bonding interactions at the β sheet surface, the secondary structure element becomes unstable and unfolds during the variant simulations. However, since the model ends abruptly at the N-terminus, no definite conclusions can be drawn from the simulations. | ||||||||
| c.866T>C | M289T 2D  AIThe SynGAP1 missense variant M289T is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools that classify the variant as benign include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and FATHMM. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized scores the variant as benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is labeled likely benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, also predicts a benign effect. Taken together, the majority of evidence indicates that M289T is most likely benign, and this conclusion does not contradict the current ClinVar status of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 1 | -4.668 | Likely Benign | 0.238 | Likely Benign | Likely Benign | 0.222 | Likely Benign | 0.73 | Ambiguous | 0.1 | 0.17 | Likely Benign | 0.45 | Likely Benign | -0.01 | Likely Benign | -0.47 | Neutral | 0.801 | Possibly Damaging | 0.315 | Benign | 1.83 | Pathogenic | 0.57 | Tolerated | -1 | -1 | -2.6 | -30.09 | ||||||||||||||||||||||
| c.1286G>A | R429Q 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant R429Q is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6‑33438191‑G‑A). Functional prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, PROVEAN, SIFT, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and ESM1b, while premPS is inconclusive. The high‑accuracy consensus (SGM Consensus) – a majority vote among AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN – yields a “Likely Benign” result. AlphaMissense‑Optimized itself predicts benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also predicts benign. Taken together, the preponderance of evidence points to a benign impact for R429Q, which does not contradict the current ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 2 | 6-33438191-G-A | 10 | 6.20e-6 | -8.227 | Likely Pathogenic | 0.143 | Likely Benign | Likely Benign | 0.156 | Likely Benign | 0.45 | Likely Benign | 0.1 | 0.36 | Likely Benign | 0.41 | Likely Benign | 0.98 | Ambiguous | -1.25 | Neutral | 1.000 | Probably Damaging | 0.979 | Probably Damaging | 3.47 | Benign | 0.58 | Tolerated | 3.38 | 25 | 1 | 1 | 1.0 | -28.06 | 235.8 | 59.5 | 0.0 | 0.0 | -0.3 | 0.4 | X | Potentially Pathogenic | The guanidinium group of the Arg429 side chain, located in an α helix (res. Met414-Glu436), either forms a salt bridge with the carboxylate group of an acidic residue (Asp474, Asp467) or an H-bond with the hydroxyl group of Ser471 in an opposing α helix (res. Ala461-Phe476). In the variant simulations, Gln429 cannot form ionic interactions with the acidic residues; however, the carboxamide group can form multiple H-bonds. The H-bonding coordination of the Asn429 side chain varied between the replica simulations. In one simulation, three H-bonds were formed simultaneously with the Asp467 side chain, the backbone carbonyl group of Asn426, and the amide group of Met430 at the end of the same α helix. The residue swap could affect the tertiary structure assembly during folding due to weaker bond formation, but no large-scale negative effects were seen during the simulations. | ||||||||
| c.3397A>G | I1133V 2D  AIThe SynGAP1 missense variant I1133V is listed in ClinVar as Benign (ClinVar ID 999690.0) and is present in the gnomAD database (gnomAD ID 6‑33443949‑A‑G). All evaluated in‑silico predictors classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is Likely Benign. Foldetta results are unavailable. Consequently, the variant is most likely benign, and this prediction aligns with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33443949-A-G | 22 | 1.48e-5 | -3.362 | Likely Benign | 0.067 | Likely Benign | Likely Benign | 0.180 | Likely Benign | 0.06 | Neutral | 0.007 | Benign | 0.007 | Benign | 5.47 | Benign | 0.58 | Tolerated | 4.32 | 3 | 4 | 3 | -0.3 | -14.03 | 10.1016/j.ajhg.2020.11.011 | ||||||||||||||||||||||||||
| c.1441C>T | H481Y 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant H481Y is listed in ClinVar as benign (ClinVar ID 1543764.0) and is present in the gnomAD database (gnomAD ID 6‑33438473‑C‑T). Prediction tools that classify the variant as benign include REVEL, Rosetta, premPS, SIFT, FATHMM, and AlphaMissense‑Optimized. Those that predict pathogenicity are PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, and AlphaMissense‑Default. FoldX and Foldetta report uncertain stability effects. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as likely pathogenic, and Foldetta as uncertain. Taking all available evidence together, the variant is most likely benign, which is consistent with its ClinVar benign annotation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Pathogenic | GAP | Likely Benign | 1 | 6-33438473-C-T | 16 | 9.91e-6 | -10.910 | Likely Pathogenic | 0.565 | Likely Pathogenic | Likely Benign | 0.256 | Likely Benign | -0.53 | Ambiguous | 0.1 | -0.46 | Likely Benign | -0.50 | Ambiguous | 0.20 | Likely Benign | -3.32 | Deleterious | 0.988 | Probably Damaging | 0.979 | Probably Damaging | 3.40 | Benign | 0.59 | Tolerated | 3.37 | 33 | 0 | 2 | 1.9 | 26.03 | 256.5 | -44.4 | 0.0 | 0.0 | 0.2 | 0.2 | X | X | Uncertain | The imidazole ring of the His481 side chain is located in a short helical structure (res. Glu480-Leu482) within an α-α loop connecting the two α-helices (res. Ala461-Phe476 and Leu489-Glu519) at the GAP-Ras interface. In the WT simulations, His481 alternately stacks against Arg485, Arg587, and Glu480 without a definite role. In the variant simulations, Tyr481 also alternately stacks with nearby arginine residues, including Arg485, Arg587, and Arg479. The interaction between Tyr481 and Arg479 affects the α-α loop, causing it to fold into a distorted helical structure, an effect that might be more pronounced during protein folding. Finally, the potential effect of the residue swap on SynGAP-Ras complex formation or GTPase activation cannot be fully addressed using the SynGAP solvent-only simulations. | |||||||
| c.1040C>A | T347N 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant T347N is listed in ClinVar with an uncertain significance (ClinVar ID 3672484.0) and is present in the gnomAD database (gnomAD ID 6‑33437945‑C‑A). Prediction tools that uniformly indicate a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The SGM‑Consensus, which aggregates the majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, resolves to “Likely Benign” (3 benign vs. 1 pathogenic). High‑accuracy assessments are consistent: AlphaMissense‑Optimized is benign, the SGM‑Consensus is likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) is benign. Overall, the collective evidence points to a benign effect, aligning with the ClinVar designation of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 1 | 6-33437945-C-A | 9 | 5.58e-6 | -5.545 | Likely Benign | 0.165 | Likely Benign | Likely Benign | 0.059 | Likely Benign | 0.41 | Likely Benign | 0.1 | 0.46 | Likely Benign | 0.44 | Likely Benign | -0.06 | Likely Benign | 1.96 | Neutral | 0.001 | Benign | 0.001 | Benign | 1.67 | Pathogenic | 0.60 | Tolerated | 3.37 | 25 | 0 | 0 | -2.8 | 13.00 | |||||||||||||||||
| c.1428C>G | F476L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant F476L is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33438460‑C‑G). Functional prediction tools cluster into two groups: benign predictions come from REVEL, PROVEAN, SIFT, and FATHMM, while pathogenic predictions arise from polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Tools that returned uncertain results—FoldX, Rosetta, Foldetta, and premPS—do not contribute to the assessment. High‑accuracy methods give the following: AlphaMissense‑Optimized predicts pathogenic; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is inconclusive, with two pathogenic and two benign calls; Foldetta also reports an uncertain stability change. Overall, the balance of evidence favors a pathogenic effect for F476L, which contrasts with the ClinVar designation of uncertain significance. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Uncertain | 2 | 6-33438460-C-G | 4 | 2.48e-6 | -10.109 | Likely Pathogenic | 0.994 | Likely Pathogenic | Likely Pathogenic | 0.180 | Likely Benign | 1.00 | Ambiguous | 0.1 | 1.04 | Ambiguous | 1.02 | Ambiguous | 0.75 | Ambiguous | -1.10 | Neutral | 0.997 | Probably Damaging | 0.978 | Probably Damaging | 3.53 | Benign | 0.60 | Tolerated | 3.40 | 22 | 2 | 0 | 1.0 | -34.02 | 235.9 | 16.1 | 0.0 | 0.1 | -0.2 | 0.0 | X | Potentially Benign | In the WT simulations, the phenyl ring of Phe476, located at the end of an α-helix (res. Ala461-Phe476), packs with the hydrophobic side chains of Leu482 and Ile483. Additionally, Phe476 stacks with the Arg475 side chain on the preceding α-α loop connecting the two α-helices (res. Ala461-Phe476 and res. Leu489-Glu519) near the GAP-Ras interface.In the variant simulations, Leu476 can maintain hydrophobic packing with neighboring residues, although not as efficiently as the phenylalanine in the WT system. The absence of Phe476/Arg475 stacking weakens the integrity of the α-helix end in the variant simulations. Nonetheless, no large-scale adverse effects are observed in the simulations. Lastly, the potential effect of the residue swap on SynGAP-Ras complex formation or GTPase activation cannot be fully addressed using the SynGAP solvent-only simulations. | |||||||||
| c.3238G>A | A1080T 2D  AIThe SynGAP1 missense variant A1080T (ClinVar ID 1473274.0) is listed as “Uncertain” in ClinVar and is present in gnomAD (ID 6‑33443790‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the computational evidence strongly suggests the variant is most likely benign, which does not contradict the current ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 2 | 6-33443790-G-A | 17 | 1.06e-5 | -3.928 | Likely Benign | 0.133 | Likely Benign | Likely Benign | 0.144 | Likely Benign | -0.19 | Neutral | 0.253 | Benign | 0.042 | Benign | 4.10 | Benign | 0.60 | Tolerated | 3.77 | 5 | 1 | 0 | -2.5 | 30.03 | |||||||||||||||||||||||||||
| c.2291A>G | N764S 2D  AIThe SynGAP1 missense variant N764S is listed in ClinVar as Benign (ClinVar ID 1948460.0) and is not reported in gnomAD. Prediction tools that agree on a benign effect include SGM‑Consensus (Likely Benign), REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. In contrast, polyPhen‑2 (HumDiv and HumVar) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, consistent with the ClinVar classification, and there is no contradiction between the predictions and the reported ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | -3.149 | Likely Benign | 0.159 | Likely Benign | Likely Benign | 0.058 | Likely Benign | -0.84 | Neutral | 0.992 | Probably Damaging | 0.846 | Possibly Damaging | 2.65 | Benign | 0.61 | Tolerated | 3.64 | 6 | 1 | 1 | 2.7 | -27.03 | ||||||||||||||||||||||||||||||
| c.3160G>A | G1054S 2D  AIThe SynGAP1 missense variant G1054S is listed in ClinVar (ID 699126.0) as Benign and is present in gnomAD (variant ID 6‑33443712‑G‑A). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic or likely pathogenic outcome. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts Benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) yields Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence strongly supports a benign effect, consistent with the ClinVar designation and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33443712-G-A | 32 | 1.99e-5 | -5.294 | Likely Benign | 0.075 | Likely Benign | Likely Benign | 0.160 | Likely Benign | 0.21 | Neutral | 0.121 | Benign | 0.013 | Benign | 4.04 | Benign | 0.63 | Tolerated | 3.77 | 5 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||
| c.1198G>C | V400L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V400L is listed in ClinVar as Benign (ClinVar ID 1166313.0) and is present in gnomAD (variant ID 6‑33438103‑G‑C). Prediction tools that agree on a benign effect include REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive result is from FoldX, which is treated as unavailable. High‑accuracy assessments confirm benignity: AlphaMissense‑Optimized is benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) is benign; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, is benign. Overall, the computational evidence strongly supports a benign classification, consistent with the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Benign | 1 | 6-33438103-G-C | 22 | 1.36e-5 | -1.000 | Likely Benign | 0.137 | Likely Benign | Likely Benign | 0.325 | Likely Benign | -0.71 | Ambiguous | 0.2 | 0.39 | Likely Benign | -0.16 | Likely Benign | -0.29 | Likely Benign | -0.60 | Neutral | 0.001 | Benign | 0.001 | Benign | 5.33 | Benign | 0.64 | Tolerated | 3.38 | 27 | 2 | 1 | -0.4 | 14.03 | 251.0 | -30.1 | 0.0 | 0.0 | 0.7 | 0.1 | X | Potentially Benign | The iso-propyl side chain of Val400, located in an anti-parallel β sheet strand (res. Ala399-Ile411), hydrophobically packs against hydrophobic residues within the anti-parallel β sheet of the C2 domain (e.g., Ile268, Ala404, Leu325, Leu402). Val400 is swapped for another hydrophobic residue, leucine, whose branched hydrocarbon side chain is of a comparable size and thus packs favorably within the C2 domain. In short, the residue swap has no apparent negative effect on the structure based on the variant simulations. | 10.1016/j.ajhg.2020.11.011 | |||||||
| c.1964T>A | L655Q 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant L655Q is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Foldetta, premPS, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) all indicate benign or likely benign. Only the two polyPhen‑2 implementations (HumDiv and HumVar) predict pathogenicity, while Rosetta remains inconclusive. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign, the SGM‑Consensus is likely benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) predicts benign stability. Overall, the majority of evidence supports a benign impact for L655Q, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | -5.278 | Likely Benign | 0.144 | Likely Benign | Likely Benign | 0.139 | Likely Benign | -0.01 | Likely Benign | 0.0 | 0.69 | Ambiguous | 0.34 | Likely Benign | -0.15 | Likely Benign | 0.61 | Neutral | 0.955 | Possibly Damaging | 0.602 | Possibly Damaging | 3.59 | Benign | 0.65 | Tolerated | 3.39 | 24 | -2 | -2 | -7.3 | 14.97 | 229.9 | -8.6 | 0.0 | 0.0 | 0.4 | 0.0 | X | Potentially Benign | The iso-butyl side chain of Leu655, located on the surface of an α helix (res. Ser641-Glu666), is not involved in any interactions in the WT simulations. In the variant simulations, the carboxamide side chain of Gln655 dynamically interacts with neighboring residues (e.g., Glu651, Glu656, Arg544) on the protein surface, with no negative structural effects. | |||||||||||
| c.3048C>A | D1016E 2D  AIThe SynGAP1 missense variant D1016E is reported in ClinVar (ID 3803472.0) as benign and is present in gnomAD (variant ID 6‑33443600‑C‑A). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, aligning with the ClinVar benign classification and showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Likely Benign | 1 | 6-33443600-C-A | 2 | 1.24e-6 | -3.422 | Likely Benign | 0.216 | Likely Benign | Likely Benign | 0.017 | Likely Benign | -0.37 | Neutral | 0.008 | Benign | 0.028 | Benign | 2.64 | Benign | 0.65 | Tolerated | 3.77 | 5 | 2 | 3 | 0.0 | 14.03 | |||||||||||||||||||||||||||
| c.886T>G | S296A 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant S296A (ClinVar ID 469166.0) is listed as ClinVar status Uncertain and is not present in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign or neutral. In contrast, polyPhen‑2 (HumDiv and HumVar) and FATHMM predict pathogenic. Grouping by consensus, the benign‑predicting tools outnumber the pathogenic ones. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is also benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, reports a benign effect. No evidence from the available tools suggests a deleterious impact. Therefore, the variant is most likely benign, and this conclusion is consistent with its ClinVar status of Uncertain rather than pathogenic. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 1 | -6.847 | Likely Benign | 0.247 | Likely Benign | Likely Benign | 0.209 | Likely Benign | 0.50 | Ambiguous | 0.3 | -0.26 | Likely Benign | 0.12 | Likely Benign | 0.35 | Likely Benign | -1.79 | Neutral | 0.992 | Probably Damaging | 0.987 | Probably Damaging | 1.97 | Pathogenic | 0.65 | Tolerated | 3.40 | 16 | 1 | 1 | 2.6 | -16.00 | 182.5 | 26.6 | -0.2 | 0.1 | -0.5 | 0.0 | X | Potentially Pathogenic | The hydroxyl group of the Ser296 side chain, located in an anti-parallel β sheet strand (res. Met289-Pro298), stably hydrogen bonds with the carboxylate group of Asp330 in a neighboring β strand (res. Ala322-Asp332). The backbone carbonyl group of Ser296 also hydrogen bonds with the guanidinium group of Arg279 in another nearby β strand (res. Arg279-Cys285). In the variant simulations, the methyl group of the Ala296 side chain cannot hydrogen bond with Asp330, causing the carboxylate group positioning to fluctuate more than in the WT simulations.Although the residue swap does not seem to affect the anti-parallel β sheet assembly during the simulations, it is possible that the Ser296-Asp330 hydrogen bond plays a crucial role in maintaining the C2 domain fold. Notably, because Ser296 is located near the membrane interface, the potential effect of the residue swap on the SynGAP-membrane association cannot be addressed by solvent-only simulations. | |||||||||||
| c.2924C>T | T975I 2D  AIThe SynGAP1 missense variant T975I is listed in ClinVar with an “Uncertain” status and is present in the gnomAD database (ID 6‑33443476‑C‑T). Prediction tools that agree on benign impact include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized; no tool predicts pathogenicity. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign, while Foldetta (combining FoldX‑MD and Rosetta outputs) has no available result for this variant. Overall, the computational evidence overwhelmingly supports a benign effect, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443476-C-T | 6 | 3.72e-6 | -3.912 | Likely Benign | 0.164 | Likely Benign | Likely Benign | 0.068 | Likely Benign | -1.66 | Neutral | 0.411 | Benign | 0.239 | Benign | 4.11 | Benign | 0.66 | Tolerated | 4.32 | 2 | 0 | -1 | 5.2 | 12.05 | |||||||||||||||||||||||||||
| c.3638A>G | N1213S 2D  AIThe SynGAP1 missense variant N1213S is listed in ClinVar as Benign (ClinVar ID 708250.0) and is present in the gnomAD database (gnomAD ID 6‑33446630‑A‑G). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). In contrast, PolyPhen‑2 (HumDiv and HumVar) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus also as benign; the Foldetta protein‑folding stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, aligning with the ClinVar classification and indicating no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Benign | 1 | 6-33446630-A-G | 13 | 8.05e-6 | -4.086 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.094 | Likely Benign | -0.56 | Neutral | 0.906 | Possibly Damaging | 0.551 | Possibly Damaging | 2.82 | Benign | 0.68 | Tolerated | 3.77 | 5 | 1 | 1 | 2.7 | -27.03 | 10.1016/j.ajhg.2020.11.011 | |||||||||||||||||||||||||
| c.1195G>A | A399T 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant A399T is listed in ClinVar (ID 1990638.0) as Benign and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts a pathogenic outcome; the only inconclusive results come from FoldX, Rosetta, and Foldetta, which are treated as unavailable evidence. High‑accuracy assessments show AlphaMissense‑Optimized as Benign, the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign, and Foldetta as Uncertain. Overall, the variant is most likely benign, and this conclusion aligns with its ClinVar benign classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Benign | 1 | -5.236 | Likely Benign | 0.114 | Likely Benign | Likely Benign | 0.272 | Likely Benign | 1.24 | Ambiguous | 0.1 | 0.91 | Ambiguous | 1.08 | Ambiguous | 0.49 | Likely Benign | -0.40 | Neutral | 0.131 | Benign | 0.039 | Benign | 5.41 | Benign | 0.69 | Tolerated | 3.38 | 26 | 1 | 0 | -2.5 | 30.03 | 211.4 | -41.4 | 0.0 | 0.0 | 0.6 | 0.4 | X | Potentially Pathogenic | The methyl group of Ala399, located in an anti-parallel β sheet strand (res. Ala399-Ile411), is swapped for a hydroxyl-containing threonine. In the variant simulations, the hydroxyl group of Thr399 can form H-bonds with the backbone atoms of the residues in the membrane-facing loops (e.g., Gly382) in the C2 domain. Consequently, the ability of the Thr399 side chain to form H-bonds with the membrane-facing loops could adversely affect the dynamics and stability of the SynGAP-membrane association. However, since the effects on the dynamics of the membrane-facing loops can only be studied through the SynGAP-membrane complex, no definite conclusions can be drawn. | |||||||||||
| c.3961C>T | P1321S 2D  AIThe SynGAP1 missense variant P1321S is listed in ClinVar (ID 1806027.0) with an “Uncertain” clinical significance and is present in the gnomAD database (gnomAD ID 6‑33451835‑C‑T). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic prediction. High‑accuracy assessments corroborate this benign trend: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign effect, which is consistent with the ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 2 | 6-33451835-C-T | 10 | 6.46e-6 | -4.897 | Likely Benign | 0.077 | Likely Benign | Likely Benign | 0.049 | Likely Benign | 0.68 | Neutral | 0.028 | Benign | 0.004 | Benign | 4.27 | Benign | 0.71 | Tolerated | 3.77 | 5 | 1 | -1 | 0.8 | -10.04 | 10.1016/j.ajhg.2020.11.011 | ||||||||||||||||||||||||||
| c.3979C>T | P1327S 2D  AIThe SynGAP1 missense variant P1327S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33451853‑C‑T). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). In contrast, polyPhen‑2 (both HumDiv and HumVar models) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta protein‑folding stability analysis is unavailable for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33451853-C-T | -4.744 | Likely Benign | 0.131 | Likely Benign | Likely Benign | 0.092 | Likely Benign | 0.28 | Neutral | 0.980 | Probably Damaging | 0.857 | Possibly Damaging | 4.25 | Benign | 0.71 | Tolerated | 3.77 | 5 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||||
| c.3049T>C | F1017L 2D  AIThe SynGAP1 missense variant F1017L is listed in ClinVar (ID 3719654.0) as benign and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and the SGM‑Consensus score (derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as uncertain, while the SGM‑Consensus (majority vote) is benign. No Foldetta stability prediction is available for this variant. Overall, the preponderance of evidence points to a benign impact, aligning with the ClinVar classification and showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | -2.048 | Likely Benign | 0.934 | Likely Pathogenic | Ambiguous | 0.157 | Likely Benign | -2.38 | Neutral | 0.798 | Possibly Damaging | 0.373 | Benign | 2.65 | Benign | 0.72 | Tolerated | 3.77 | 5 | 0 | 2 | 1.0 | -34.02 | ||||||||||||||||||||||||||||||
| c.3631A>G | M1211V 2D  AIThe SynGAP1 missense variant M1211V is listed in ClinVar as Benign (ClinVar ID 3674914.0) and is present in gnomAD (ID 6‑33446623‑A‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus as Likely Benign, and Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, aligning with the ClinVar classification and not contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Benign | 1 | 6-33446623-A-G | 3 | 1.86e-6 | -2.101 | Likely Benign | 0.258 | Likely Benign | Likely Benign | 0.412 | Likely Benign | -0.29 | Neutral | 0.932 | Possibly Damaging | 0.949 | Probably Damaging | 5.43 | Benign | 0.72 | Tolerated | 3.77 | 5 | 1 | 2 | 2.3 | -32.06 | ||||||||||||||||||||||||||
| c.2935T>C | F979L 2D  AIThe SynGAP1 missense variant F979L (ClinVar ID 1000410.0, status Uncertain, not found in gnomAD) has been evaluated by multiple in silico predictors. Benign predictions come from REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, and FATHMM, while pathogenic predictions are reported by polyPhen‑2 HumDiv and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign. High‑accuracy assessments show AlphaMissense‑Optimized as Uncertain, whereas the SGM‑Consensus (majority vote) supports a benign outcome. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta, has no available result for this variant. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar status of Uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.341 | Likely Benign | 0.870 | Likely Pathogenic | Ambiguous | 0.228 | Likely Benign | -1.00 | Neutral | 0.625 | Possibly Damaging | 0.430 | Benign | 4.22 | Benign | 0.73 | Tolerated | 4.32 | 2 | 2 | 0 | 1.0 | -34.02 | ||||||||||||||||||||||||||||||
| c.2900G>T | R967L 2D  AIThe SynGAP1 missense variant R967L is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443452‑G‑T). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). In contrast, polyPhen‑2 (both HumDiv and HumVar models) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the preponderance of evidence points to a benign impact for R967L, which does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443452-G-T | 1 | 6.20e-7 | -3.496 | Likely Benign | 0.164 | Likely Benign | Likely Benign | 0.123 | Likely Benign | -0.99 | Neutral | 0.959 | Probably Damaging | 0.586 | Possibly Damaging | 4.15 | Benign | 0.75 | Tolerated | 4.32 | 2 | -2 | -3 | 8.3 | -43.03 | |||||||||||||||||||||||||||
| c.1108G>A | G370S 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant G370S is listed in ClinVar with an uncertain significance and is present in gnomAD (variant ID 6‑33438013‑G‑A). Consensus predictions from standard in silico tools cluster into two groups: benign (REVEL, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized) and pathogenic (FoldX, FATHMM). Two tools report uncertainty: Rosetta and Foldetta. High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized predicts benign; the SGM Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is Likely Benign; Foldetta remains uncertain. Overall, the preponderance of evidence points to a benign effect, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Uncertain | 1 | 6-33438013-G-A | 15 | 9.31e-6 | -3.533 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.282 | Likely Benign | 2.83 | Destabilizing | 2.0 | 1.05 | Ambiguous | 1.94 | Ambiguous | -0.02 | Likely Benign | 0.47 | Neutral | 0.000 | Benign | 0.000 | Benign | 1.33 | Pathogenic | 0.77 | Tolerated | 3.42 | 19 | 1 | 0 | -0.4 | 30.03 | 196.6 | -49.6 | 0.9 | 2.2 | -0.1 | 0.4 | Uncertain | Gly370 is located in the Gly-rich Ω loop (res. Pro364- Pro398) between two anti-parallel β sheet strands (res. Thr359-Pro364, res. Ala399-Ile411). Because, the Ω loop is assumed to be directly interacting with the membrane, it is only seen to move arbitrarily throughout the WT solvent simulations. The Ω loop is potentially playing a crucial loop in the SynGAP-membrane complex association, stability and dynamics, regardless, this aspect cannot be addressed through the solvent simulations only. The Ω-loops are known to have a major role in protein functions that requires flexibility and thus, they are rich in glycines, prolines and to a lesser extent, hydrophilic residues to ensure maximum flexibility. Thus, Ser370 in the variant is potentially tolerated in the Ω loop. However, since the effect on the Gly-rich Ω loop dynamics can only be well-studied through the SynGAP-membrane complex, no definite conclusions can be withdrawn. | |||||||||
| c.2493G>C | E831D 2D  AIThe SynGAP1 missense variant E831D is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443045‑G‑C). All available in‑silico predictors classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic or likely‑pathogenic outcome. Grouping by agreement, the benign‑predicting tools comprise the entire set, while no pathogenic predictions are present. High‑accuracy assessments reinforce this: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also yields a benign classification. Foldetta results are unavailable. Overall, the computational evidence strongly supports a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443045-G-C | 1 | 6.19e-7 | -3.055 | Likely Benign | 0.063 | Likely Benign | Likely Benign | 0.073 | Likely Benign | 1.23 | Neutral | 0.002 | Benign | 0.002 | Benign | 2.64 | Benign | 0.77 | Tolerated | 3.77 | 5 | 3 | 2 | 0.0 | -14.03 | |||||||||||||||||||||||||||
| c.2113A>C | K705Q 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 K705Q missense variant (ClinVar ID 3699560.0) is listed as “Uncertain” in ClinVar and is present in gnomAD (variant ID 6‑33441372‑A‑C). Prediction tools that uniformly indicate a benign effect include REVEL, FoldX, Rosetta, Foldetta, premPS, PROVEAN, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. In contrast, polyPhen‑2 (both HumDiv and HumVar models) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign; the SGM Consensus—derived from a majority vote of AlphaMissense‑Default (uncertain), ESM1b (benign), FATHMM (benign), and PROVEAN (benign)—also yields a benign classification; Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, predicts benign. Overall, the preponderance of evidence supports a benign impact for K705Q, and this conclusion does not contradict the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | 6-33441372-A-C | 1 | 6.20e-7 | -5.787 | Likely Benign | 0.436 | Ambiguous | Likely Benign | 0.142 | Likely Benign | -0.10 | Likely Benign | 0.0 | 0.33 | Likely Benign | 0.12 | Likely Benign | -0.02 | Likely Benign | -0.24 | Neutral | 0.997 | Probably Damaging | 0.969 | Probably Damaging | 3.42 | Benign | 0.78 | Tolerated | 3.47 | 10 | 1 | 1 | 0.4 | -0.04 | |||||||||||||||||
| c.484C>G | R162G 2D  AIThe SynGAP1 missense variant R162G is listed in ClinVar (ID 2703066.0) with an uncertain significance status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is classified as Likely Benign (three benign votes versus one pathogenic). High‑accuracy assessments show AlphaMissense‑Optimized as Benign and the SGM‑Consensus as Likely Benign; Foldetta stability analysis is unavailable. Overall, the majority of predictions support a benign impact, and this is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -6.985 | Likely Benign | 0.664 | Likely Pathogenic | Likely Benign | 0.190 | Likely Benign | -0.73 | Neutral | 0.487 | Possibly Damaging | 0.272 | Benign | 4.09 | Benign | 0.78 | Tolerated | 3.74 | 4 | -2 | -3 | 4.1 | -99.14 | ||||||||||||||||||||||||||||||
| c.2998A>G | I1000V 2D  AIThe SynGAP1 missense variant I1000V is listed in ClinVar (ID 2572013.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Functional prediction tools that assess evolutionary conservation and structural impact (REVEL, SIFT, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, ESM1b, FATHMM, PROVEAN, AlphaMissense‑Default) all converge on a benign outcome. No tool in the dataset predicts pathogenicity. High‑accuracy predictors reinforce this consensus: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) indicates a likely benign classification. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Based on the collective evidence, the variant is most likely benign, and this assessment does not contradict the ClinVar status, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 2 | -4.102 | Likely Benign | 0.098 | Likely Benign | Likely Benign | 0.086 | Likely Benign | -0.20 | Neutral | 0.437 | Benign | 0.170 | Benign | 2.76 | Benign | 0.81 | Tolerated | 4.32 | 4 | 3 | 4 | -0.3 | -14.03 | ||||||||||||||||||||||||||||||
| c.458C>A | T153N 2D  AIThe SynGAP1 missense variant T153N is listed in ClinVar (ID 984906.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all predict benign, while the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates a likely benign outcome. In contrast, polyPhen‑2 (HumDiv and HumVar) predict pathogenic. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence supports a benign classification, and this is consistent with the ClinVar “Uncertain” designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 3 | -0.739 | Likely Benign | 0.226 | Likely Benign | Likely Benign | 0.161 | Likely Benign | 0.88 | Neutral | 0.888 | Possibly Damaging | 0.537 | Possibly Damaging | 4.23 | Benign | 0.81 | Tolerated | 3.61 | 5 | 0 | 0 | -2.8 | 13.00 | ||||||||||||||||||||||||||||||
| c.3502A>G | I1168V 2D  AIThe SynGAP1 missense variant I1168V is listed in ClinVar (ID 936001.0) with an “Uncertain” status and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. AlphaMissense‑Default is uncertain, while the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) reports a “Likely Benign” outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign impact, and this consensus does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -3.263 | Likely Benign | 0.524 | Ambiguous | Likely Benign | 0.363 | Likely Benign | -0.14 | Neutral | 0.876 | Possibly Damaging | 0.643 | Possibly Damaging | 5.47 | Benign | 0.84 | Tolerated | 3.88 | 3 | 4 | 3 | -0.3 | -14.03 | ||||||||||||||||||||||||||||||
| c.892C>T | P298S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant P298S is listed in ClinVar as Benign (ClinVar ID 2965590.0) and is present in gnomAD (ID 6‑33437797‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and FATHMM. High‑accuracy assessments show AlphaMissense‑Optimized as Benign, the SGM‑Consensus as Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as Uncertain. No evidence from FoldX, Rosetta, or premPS is available to support either outcome. Overall, the majority of predictions support a benign impact, aligning with the ClinVar designation. Thus, the variant is most likely benign and does not contradict the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Benign | 1 | 6-33437797-C-T | 5 | 3.10e-6 | -6.342 | Likely Benign | 0.144 | Likely Benign | Likely Benign | 0.189 | Likely Benign | 1.38 | Ambiguous | 0.2 | 1.41 | Ambiguous | 1.40 | Ambiguous | 0.58 | Ambiguous | -1.20 | Neutral | 0.991 | Probably Damaging | 0.898 | Possibly Damaging | 2.03 | Pathogenic | 0.85 | Tolerated | 3.39 | 20 | -1 | 1 | 0.8 | -10.04 | |||||||||||||||||
| c.526A>C | S176R 2D  AIThe SynGAP1 missense variant S176R is listed in ClinVar with an uncertain significance and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, and FATHMM, whereas polyPhen‑2 HumDiv, AlphaMissense‑Default, and AlphaMissense‑Optimized predict a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a likely benign classification. High‑accuracy assessments show AlphaMissense‑Optimized as pathogenic, while the SGM‑Consensus remains likely benign; Foldetta results are unavailable. Overall, the balance of evidence favors a benign interpretation, and this assessment does not conflict with the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -6.492 | Likely Benign | 0.987 | Likely Pathogenic | Likely Pathogenic | 0.247 | Likely Benign | 0.94 | Neutral | 0.718 | Possibly Damaging | 0.168 | Benign | 4.16 | Benign | 0.87 | Tolerated | 0 | -1 | -3.7 | 69.11 | ||||||||||||||||||||||||||||||||
| c.1851G>T | E617D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant E617D is listed in ClinVar with an uncertain significance (ID 2584916.0) and is not reported in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, FoldX, Foldetta, premPS, PROVEAN, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score all indicate benign or likely benign. In contrast, polyPhen‑2 (HumDiv and HumVar) and FATHMM predict a pathogenic impact, while Rosetta remains inconclusive. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized is benign; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is likely benign; and Foldetta, which integrates FoldX‑MD and Rosetta outputs, also predicts benign. Overall, the preponderance of evidence supports a benign classification, which does not contradict the current ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | -1.349 | Likely Benign | 0.241 | Likely Benign | Likely Benign | 0.322 | Likely Benign | 0.12 | Likely Benign | 0.1 | 0.80 | Ambiguous | 0.46 | Likely Benign | 0.07 | Likely Benign | -0.01 | Neutral | 0.994 | Probably Damaging | 0.979 | Probably Damaging | -1.35 | Pathogenic | 0.88 | Tolerated | 3.37 | 35 | 2 | 3 | 0.0 | -14.03 | ||||||||||||||||||||
| c.3434A>G | N1145S 2D  AIThe SynGAP1 missense variant N1145S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33444469‑A‑G). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). In contrast, PolyPhen‑2 (both HumDiv and HumVar models) predict a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; the Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33444469-A-G | 2 | 1.24e-6 | -0.989 | Likely Benign | 0.126 | Likely Benign | Likely Benign | 0.308 | Likely Benign | -1.15 | Neutral | 0.997 | Probably Damaging | 0.989 | Probably Damaging | 5.55 | Benign | 0.89 | Tolerated | 4.32 | 4 | 1 | 1 | 2.7 | -27.03 | |||||||||||||||||||||||||||
| c.1339G>C | V447L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V447L is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that classify the variant as benign include REVEL, Foldetta, premPS, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). Tools that predict pathogenicity are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. Uncertain results are reported by FoldX, Rosetta, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as Benign, the SGM‑Consensus as Likely Benign, and Foldetta as Benign. Overall, the majority of evidence points to a benign effect, and this consensus does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | -5.136 | Likely Benign | 0.491 | Ambiguous | Likely Benign | 0.180 | Likely Benign | -1.13 | Ambiguous | 0.1 | 0.54 | Ambiguous | -0.30 | Likely Benign | 0.03 | Likely Benign | -0.29 | Neutral | 0.947 | Possibly Damaging | 0.851 | Possibly Damaging | 3.61 | Benign | 0.90 | Tolerated | 3.37 | 32 | 1 | 2 | -0.4 | 14.03 | ||||||||||||||||||||
| c.3943T>C | W1315R 2D  AIThe SynGAP1 missense variant W1315R is listed in ClinVar (ID 1029092.0) with an “Uncertain” clinical significance and is not reported in gnomAD. Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus score (which is a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). Only AlphaMissense‑Default predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as benign; Foldetta results are not available for this variant. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 0.205 | Likely Benign | 0.660 | Likely Pathogenic | Likely Benign | 0.114 | Likely Benign | 1.31 | Neutral | 0.000 | Benign | 0.001 | Benign | 4.37 | Benign | 0.91 | Tolerated | 3.77 | 5 | 2 | -3 | -3.6 | -30.03 | ||||||||||||||||||||||||||||||
| c.2695A>G | I899V 2D  AIThe SynGAP1 missense variant I899V is listed in ClinVar as a benign alteration (ClinVar ID 1003653.0) and is present in the gnomAD database (gnomAD ID 6‑33443247‑A‑G). All evaluated in‑silico predictors classify the variant as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity, so the pathogenic‑prediction group is empty. High‑accuracy assessments further support a benign effect: AlphaMissense‑Optimized reports benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant. Overall, the computational evidence strongly suggests the variant is benign, consistent with its ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33443247-A-G | 6 | 3.72e-6 | -2.569 | Likely Benign | 0.074 | Likely Benign | Likely Benign | 0.040 | Likely Benign | 0.09 | Neutral | 0.220 | Benign | 0.078 | Benign | 2.75 | Benign | 0.92 | Tolerated | 4.32 | 4 | 4 | 3 | -0.3 | -14.03 | |||||||||||||||||||||||||||
| c.3364G>A | G1122S 2D  AIThe SynGAP1 missense variant G1122S is listed in ClinVar as a benign alteration (ClinVar ID 643187.0) and is present in gnomAD (gnomAD ID 6‑33443916‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates likely benign, while Foldetta’s protein‑folding stability result is unavailable. Overall, the variant is most likely benign, and this assessment is consistent with its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign/Likely benign | 2 | 6-33443916-G-A | 27 | 1.79e-5 | -4.880 | Likely Benign | 0.072 | Likely Benign | Likely Benign | 0.189 | Likely Benign | -0.08 | Neutral | 0.022 | Benign | 0.006 | Benign | 4.89 | Benign | 0.92 | Tolerated | 3.77 | 5 | 1 | 0 | -0.4 | 30.03 | |||||||||||||||||||||||||||
| c.958G>C | V320L 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V320L is listed in ClinVar with an uncertain significance and is present in gnomAD (ID 6‑33437863‑G‑C). Functional prediction tools that agree on a benign effect include REVEL, FoldX, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and FATHMM. Predictions that are inconclusive are Rosetta, Foldetta, premPS, and AlphaMissense‑Default. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign impact. This conclusion does not contradict the ClinVar status, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | C2 | Uncertain | 1 | 6-33437863-G-C | 6 | 3.72e-6 | -6.207 | Likely Benign | 0.362 | Ambiguous | Likely Benign | 0.096 | Likely Benign | -0.26 | Likely Benign | 0.2 | 1.33 | Ambiguous | 0.54 | Ambiguous | 0.51 | Ambiguous | -1.02 | Neutral | 0.900 | Possibly Damaging | 0.373 | Benign | 1.78 | Pathogenic | 0.92 | Tolerated | 3.38 | 23 | 2 | 1 | -0.4 | 14.03 | 245.8 | -10.2 | 0.3 | 0.9 | 0.1 | 0.3 | X | Potentially Benign | The isopropyl side chain of Val310, located in a β hairpin loop linking two anti-parallel β sheet strands (res. Thr305-Asn315, res. Ala322-Asp330), hydrophobically packs with the side chains of nearby residues (e.g., Leu286, Val350, Pro318). The hydrophobic Leu320 side chain mostly forms the same interactions; hence, the residue swap does not seem to negatively affect the protein structure based on the variant simulations. | |||||||||
| c.1322T>C | V441A 2D  3DClick to see structure in 3D Viewer AISynGAP1 variant V441A is listed in ClinVar as uncertain and is present in gnomAD (ID 6‑33438227‑T‑C). Consensus from most in silico predictors favors a benign effect: REVEL, FoldX, Rosetta, Foldetta, polyPhen‑2 HumVar, SIFT, FATHMM, and AlphaMissense‑Optimized all report benign. Pathogenic predictions come from PROVEAN, polyPhen‑2 HumDiv, and ESM1b, while premPS and AlphaMissense‑Default remain uncertain. High‑accuracy assessments give a mixed picture: AlphaMissense‑Optimized predicts benign; the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) yields pathogenic; Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, reports benign. Overall, the preponderance of evidence points to a benign impact, aligning with the ClinVar uncertain designation rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | GAP | Conflicting | 2 | 6-33438227-T-C | 3 | 1.86e-6 | -9.439 | Likely Pathogenic | 0.359 | Ambiguous | Likely Benign | 0.053 | Likely Benign | -0.14 | Likely Benign | 0.0 | 0.33 | Likely Benign | 0.10 | Likely Benign | 0.95 | Ambiguous | -2.92 | Deleterious | 0.513 | Possibly Damaging | 0.214 | Benign | 3.44 | Benign | 0.93 | Tolerated | 3.37 | 29 | 0 | 0 | -2.4 | -28.05 | 195.0 | 44.6 | 0.0 | 0.1 | 0.5 | 0.0 | X | X | Uncertain | The iso-propyl side chain of Val441, located on the outer surface of an α helix (res. Asn440-Thr458), does not interact with other residues in the WT simulations. In the variant simulations, the methyl side chain of Ala441 is similarly hydrophobic and does not form any interactions on the outer helix surface. Although the residue swap does not negatively affect the protein structure based on the simulations, it is noteworthy that the residue faces the RasGTPase interface. Thus, the effect of the residue swap on the SynGAP-Ras complex formation or GTPase activation cannot be fully addressed using the SynGAP solvent-only simulations. | ||||||||
| c.2186A>G | N729S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant N729S is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). No tool in the dataset predicts a pathogenic outcome. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM Consensus also as benign, while Foldetta (combining FoldX‑MD and Rosetta outputs) is inconclusive. Based on the collective predictions, the variant is most likely benign, and this assessment does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | -1.578 | Likely Benign | 0.066 | Likely Benign | Likely Benign | 0.063 | Likely Benign | 0.14 | Likely Benign | 0.1 | 1.34 | Ambiguous | 0.74 | Ambiguous | -0.36 | Likely Benign | -0.42 | Neutral | 0.221 | Benign | 0.027 | Benign | 3.38 | Benign | 0.93 | Tolerated | 3.59 | 7 | 1 | 1 | 2.7 | -27.03 | ||||||||||||||||||||
| c.3929C>T | T1310M 2D  AIThe SynGAP1 missense variant T1310M is listed in ClinVar (ID 2160201.0) as Benign and is present in gnomAD (gnomAD ID 6‑33451803‑C‑T). All evaluated in‑silico predictors report a benign outcome: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the computational evidence overwhelmingly supports a benign classification, which aligns with the ClinVar status and does not contradict it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | 6-33451803-C-T | 17 | 1.05e-5 | -4.822 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.069 | Likely Benign | 2.19 | Neutral | 0.021 | Benign | 0.005 | Benign | 2.98 | Benign | 0.93 | Tolerated | 3.77 | 5 | -1 | -1 | 2.6 | 30.09 | |||||||||||||||||||||||||||
| c.2434C>T | P812S 2D  AIThe SynGAP1 missense variant P812S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33442986‑C‑T). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are polyPhen‑2 HumDiv and polyPhen‑2 HumVar. AlphaMissense‑Default remains uncertain, and no Foldetta stability result is available. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta data are missing. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | Uncertain | 1 | 6-33442986-C-T | 1 | 6.20e-7 | -5.689 | Likely Benign | 0.456 | Ambiguous | Likely Benign | 0.162 | Likely Benign | -0.62 | Neutral | 0.999 | Probably Damaging | 0.966 | Probably Damaging | 2.89 | Benign | 0.95 | Tolerated | 4.32 | 4 | 1 | -1 | 0.8 | -10.04 | ||||||||||||||||||||||||||
| c.3304G>A | A1102T 2D  AIThe SynGAP1 missense variant A1102T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443856‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443856-G-A | 11 | 7.17e-6 | -3.540 | Likely Benign | 0.070 | Likely Benign | Likely Benign | 0.044 | Likely Benign | -0.30 | Neutral | 0.001 | Benign | 0.001 | Benign | 2.32 | Pathogenic | 0.95 | Tolerated | 3.77 | 5 | 1 | 0 | -2.5 | 30.03 | |||||||||||||||||||||||||||
| c.2302G>A | D768N 2D  AIThe SynGAP1 missense variant D768N is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33442460‑G‑A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN). No tool predicts a pathogenic outcome; AlphaMissense‑Default is uncertain. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus is “Likely Benign,” and Foldetta data are unavailable. Overall, the consensus of available predictions indicates that the variant is most likely benign, which does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33442460-G-A | 2 | 2.57e-6 | -6.892 | Likely Benign | 0.453 | Ambiguous | Likely Benign | 0.048 | Likely Benign | -0.77 | Neutral | 0.106 | Benign | 0.009 | Benign | 4.07 | Benign | 0.96 | Tolerated | 3.64 | 6 | 1 | 2 | 0.0 | -0.98 | |||||||||||||||||||||||||||
| c.2420A>T | Y807F 2D  AIThe SynGAP1 missense variant Y807F is listed in ClinVar (ID 1491782.0) with an “Uncertain” status and is not reported in gnomAD. All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) also indicates a likely benign effect. No tool in the set predicts pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus (derived from the four high‑accuracy predictors) is benign. Foldetta, which integrates FoldX‑MD and Rosetta stability calculations, has no available result for this variant. Overall, the computational evidence strongly supports a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | SH3-binding motif | Uncertain | 1 | -3.667 | Likely Benign | 0.073 | Likely Benign | Likely Benign | 0.057 | Likely Benign | 0.14 | Neutral | 0.012 | Benign | 0.022 | Benign | 2.92 | Benign | 0.98 | Tolerated | 3.77 | 5 | 7 | 3 | 4.1 | -16.00 | |||||||||||||||||||||||||||||
| c.1663G>A | V555I 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant V555I is listed in ClinVar with an “Uncertain” status and is not reported in gnomAD. Prediction tools that uniformly indicate a benign effect include REVEL, Rosetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus score (Likely Benign). Only FATHMM predicts a pathogenic outcome, while FoldX, Foldetta, and premPS are inconclusive. High‑accuracy assessments show AlphaMissense‑Optimized as benign, the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN) as Likely Benign, and Foldetta as uncertain. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | -4.544 | Likely Benign | 0.084 | Likely Benign | Likely Benign | 0.253 | Likely Benign | -0.82 | Ambiguous | 0.0 | -0.41 | Likely Benign | -0.62 | Ambiguous | -0.55 | Ambiguous | 0.45 | Neutral | 0.002 | Benign | 0.002 | Benign | -1.26 | Pathogenic | 1.00 | Tolerated | 4 | 3 | 0.3 | 14.03 | ||||||||||||||||||||||
| c.2101C>T | P701S 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant P701S (ClinVar ID 2995856.0) is listed as “Uncertain” in ClinVar and is present in gnomAD (ID 6‑33441360‑C‑T). Prediction tools that agree on a benign effect include REVEL, Rosetta, premPS, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (Likely Benign). No tool in the dataset predicts a pathogenic outcome; all remaining predictions are either benign or uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as Benign, the SGM‑Consensus as Likely Benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) as Uncertain. Based on the collective evidence, the variant is most likely benign, and this conclusion does not contradict the ClinVar status, which remains uncertain. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | GAP | Uncertain | 1 | 6-33441360-C-T | 3 | 1.86e-6 | -4.375 | Likely Benign | 0.221 | Likely Benign | Likely Benign | 0.132 | Likely Benign | 1.33 | Ambiguous | 0.0 | 0.12 | Likely Benign | 0.73 | Ambiguous | -0.36 | Likely Benign | 0.78 | Neutral | 0.044 | Benign | 0.025 | Benign | 3.48 | Benign | 1.00 | Tolerated | 3.47 | 10 | -1 | 1 | 0.8 | -10.04 | 10.1016/j.ajhg.2020.11.011 | ||||||||||||||||
| c.2275A>C | M759L 2D  AIThe SynGAP1 missense variant M759L is listed in ClinVar with an uncertain significance (ClinVar ID 942432.0) and is present in gnomAD (gnomAD ID 6‑33441740‑A‑C). All evaluated in‑silico predictors agree on a benign effect: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign classifications. No tool predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the computational evidence strongly supports a benign impact, which is consistent with the ClinVar uncertain status rather than contradicting it. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33441740-A-C | 2 | 1.24e-6 | -2.431 | Likely Benign | 0.093 | Likely Benign | Likely Benign | 0.048 | Likely Benign | -0.53 | Neutral | 0.002 | Benign | 0.005 | Benign | 2.84 | Benign | 1.00 | Tolerated | 3.99 | 5 | 4 | 2 | 1.9 | -18.03 | |||||||||||||||||||||||||||
| c.2299A>G | I767V 2D  AIThe SynGAP1 missense variant I767V is listed in ClinVar (ID 1402700.0) with an “Uncertain” clinical significance and is not reported in gnomAD. All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. The SGM‑Consensus, which aggregates the majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, also reports a “Likely Benign” outcome. No tool predicts pathogenicity. High‑accuracy assessments confirm this: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, while Foldetta (combining FoldX‑MD and Rosetta stability predictions) has no available result for this variant. Overall, the computational evidence strongly supports a benign effect, and this conclusion does not contradict the current ClinVar status of uncertainty. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -2.791 | Likely Benign | 0.064 | Likely Benign | Likely Benign | 0.096 | Likely Benign | 0.10 | Neutral | 0.072 | Benign | 0.029 | Benign | 4.21 | Benign | 1.00 | Tolerated | 3.64 | 6 | 4 | 3 | -0.3 | -14.03 | ||||||||||||||||||||||||||||||
| c.2343G>A | M781I 2D  AIThe SynGAP1 missense variant M781I is listed in ClinVar (ID 2802065.0) as Benign and is not reported in gnomAD. All available in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized predicts benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates Likely Benign. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the predictions strongly support a benign effect, consistent with the ClinVar designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | -2.484 | Likely Benign | 0.323 | Likely Benign | Likely Benign | 0.101 | Likely Benign | 0.05 | Neutral | 0.000 | Benign | 0.001 | Benign | 2.89 | Benign | 1.00 | Tolerated | 3.64 | 6 | 1 | 2 | 2.6 | -18.03 | ||||||||||||||||||||||||||||||
| c.2350G>A | A784T 2D  AIThe SynGAP1 missense variant A784T is listed in ClinVar (ID 962668.0) as Benign and is not reported in gnomAD. Across the available in‑silico predictors, every tool examined—REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized—consistently classifies the substitution as benign. No tool predicts pathogenicity. High‑accuracy assessments reinforce this consensus: AlphaMissense‑Optimized reports a benign effect, and the SGM Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) indicates a likely benign outcome. A Foldetta stability analysis is unavailable, so it does not influence the overall interpretation. Based on the unanimous benign predictions and the ClinVar designation, the variant is most likely benign, with no contradiction to the ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Benign | 1 | -3.579 | Likely Benign | 0.089 | Likely Benign | Likely Benign | 0.046 | Likely Benign | 1.23 | Neutral | 0.001 | Benign | 0.006 | Benign | 2.92 | Benign | 1.00 | Tolerated | 3.64 | 6 | 1 | 0 | -2.5 | 30.03 | ||||||||||||||||||||||||||||||
| c.2860C>T | P954S 2D  AIThe SynGAP1 missense variant P954S is listed in ClinVar as Benign (ClinVar ID 833606.0) and is present in gnomAD (ID 6‑33443412‑C‑T). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). Tools that predict a pathogenic effect are PolyPhen‑2 HumDiv and PolyPhen‑2 HumVar. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus as likely benign; Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. Overall, the majority of evidence points to a benign impact, which is consistent with the ClinVar classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Likely Benign | 1 | 6-33443412-C-T | 1 | 6.20e-7 | -3.525 | Likely Benign | 0.062 | Likely Benign | Likely Benign | 0.143 | Likely Benign | -0.25 | Neutral | 0.954 | Possibly Damaging | 0.812 | Possibly Damaging | 2.87 | Benign | 1.00 | Tolerated | 3.77 | 5 | 1 | -1 | 0.8 | -10.04 | |||||||||||||||||||||||||||
| c.3092T>C | M1031T 2D  AIThe SynGAP1 missense variant M1031T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33443644‑T‑C). In silico prediction tools that agree on a benign effect include REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote from AlphaMissense‑Default, ESM1b, FATHMM, PROVEAN). No tool predicts a pathogenic outcome; the only inconclusive result is AlphaMissense‑Default, which is treated as unavailable. High‑accuracy assessments further support a benign classification: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus is “Likely Benign,” and Foldetta data are not available. **Thus, the variant is most likely benign, and this conclusion does not contradict the ClinVar “Uncertain” status.** Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33443644-T-C | 2 | 1.24e-6 | -1.863 | Likely Benign | 0.540 | Ambiguous | Likely Benign | 0.085 | Likely Benign | -0.24 | Neutral | 0.002 | Benign | 0.005 | Benign | 2.67 | Benign | 1.00 | Tolerated | 3.77 | 5 | -1 | -1 | -2.6 | -30.09 | |||||||||||||||||||||||||||
| c.3223C>A | Q1075K 2D  AIThe SynGAP1 missense variant Q1075K (ClinVar ID 2762879.0) is listed as “Uncertain” in ClinVar and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are polyPhen‑2 HumDiv, polyPhen‑2 HumVar, and AlphaMissense‑Default. The SGM‑Consensus, which aggregates AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is “Likely Benign” because three of the four contributing tools predict benign. High‑accuracy assessments show AlphaMissense‑Optimized as benign and the SGM‑Consensus (majority vote) as benign; Foldetta stability analysis is unavailable. Overall, the majority of evidence points to a benign impact, and this conclusion does not contradict the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | -5.135 | Likely Benign | 0.728 | Likely Pathogenic | Likely Benign | 0.134 | Likely Benign | -0.67 | Neutral | 0.963 | Probably Damaging | 0.959 | Probably Damaging | 2.75 | Benign | 1.00 | Tolerated | 3.77 | 5 | 1 | 1 | -0.4 | 0.04 | ||||||||||||||||||||||||||||||
| c.323A>G | K108R 2D  AIThe SynGAP1 missense variant K108R is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33432188‑A‑G). Functional prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. In contrast, polyPhen‑2 (HumDiv and HumVar) predict a pathogenic impact. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support benignity: AlphaMissense‑Optimized is benign, and the SGM‑Consensus itself is benign; Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33432188-A-G | 6 | 3.72e-6 | -2.892 | Likely Benign | 0.148 | Likely Benign | Likely Benign | 0.184 | Likely Benign | 0.37 | Neutral | 0.993 | Probably Damaging | 0.956 | Probably Damaging | 4.22 | Benign | 1.00 | Tolerated | 3.61 | 5 | 3 | 2 | -0.6 | 28.01 | |||||||||||||||||||||||||||
| c.3380G>C | G1127A 2D  AIThe SynGAP1 missense variant G1127A is listed in ClinVar (ID 426748.0) with an uncertain significance status and is present in gnomAD (variant ID 6‑33443932‑G‑C). Functional prediction tools uniformly indicate a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all score the variant as benign. No tool predicts pathogenicity. The high‑accuracy consensus methods corroborate this: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability predictor combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the evidence strongly supports a benign classification, which does not contradict the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 4 | 6-33443932-G-C | 4 | 2.68e-6 | -5.949 | Likely Benign | 0.080 | Likely Benign | Likely Benign | 0.164 | Likely Benign | -0.43 | Neutral | 0.001 | Benign | 0.002 | Benign | 4.83 | Benign | 1.00 | Tolerated | 4.32 | 4 | 1 | 0 | 2.2 | 14.03 | |||||||||||||||||||||||||||
| c.3633G>A | M1211I 2D  AIThe SynGAP1 missense variant M1211I is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6-33446625-G-A). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized. Tools that predict a pathogenic effect are AlphaMissense‑Default, polyPhen‑2 HumDiv, and polyPhen‑2 HumVar. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, has no available result for this variant. High‑accuracy predictions therefore point to a benign outcome: AlphaMissense‑Optimized is benign, and the SGM‑Consensus also indicates benign. Based on the aggregate predictions, the variant is most likely benign, which does not contradict the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Uncertain | 1 | 6-33446625-G-A | 3 | 1.86e-6 | -1.537 | Likely Benign | 0.764 | Likely Pathogenic | Likely Benign | 0.298 | Likely Benign | -0.42 | Neutral | 0.969 | Probably Damaging | 0.968 | Probably Damaging | 5.40 | Benign | 1.00 | Tolerated | 3.77 | 5 | 1 | 2 | 2.6 | -18.03 | ||||||||||||||||||||||||||
| c.3638A>C | N1213T 2D  AIThe SynGAP1 missense variant N1213T is listed in ClinVar with an “Uncertain” status and is present in gnomAD (ID 6‑33446630‑A‑C). Prediction tools that agree on a benign effect include REVEL, PROVEAN, SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. In contrast, polyPhen‑2 (HumDiv and HumVar) predict a pathogenic outcome. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, reports a “Likely Benign” classification. High‑accuracy assessments further support benignity: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, while Foldetta results are unavailable. Overall, the majority of evidence points to a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Coiled-coil | Conflicting | 2 | 6-33446630-A-C | 46 | 2.85e-5 | -5.428 | Likely Benign | 0.266 | Likely Benign | Likely Benign | 0.097 | Likely Benign | -1.08 | Neutral | 0.959 | Probably Damaging | 0.721 | Possibly Damaging | 2.74 | Benign | 1.00 | Tolerated | 3.77 | 5 | 0 | 0 | 2.8 | -13.00 | ||||||||||||||||||||||||||
| c.3858A>T | E1286D 2D  AIThe SynGAP1 missense variant E1286D is listed in ClinVar (ID 469159.0) with an “Uncertain” clinical significance and is present in gnomAD (variant ID 6‑33447906‑A‑T). All evaluated in‑silico predictors classify the substitution as benign: REVEL, PROVEAN, PolyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores. No tool in the set predicts pathogenicity. High‑accuracy assessments reinforce this view: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta, a protein‑folding stability method combining FoldX‑MD and Rosetta outputs, did not provide a result for this variant, so its status is unavailable. Overall, the computational evidence strongly supports a benign effect, and this conclusion does not contradict the current ClinVar “Uncertain” designation. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 4 | 6-33447906-A-T | 143 | 9.22e-5 | -4.010 | Likely Benign | 0.081 | Likely Benign | Likely Benign | 0.036 | Likely Benign | 1.02 | Neutral | 0.001 | Benign | 0.004 | Benign | 2.96 | Benign | 1.00 | Tolerated | 3.77 | 5 | 3 | 2 | 0.0 | -14.03 | 10.1016/j.ajhg.2020.11.011 | ||||||||||||||||||||||||||
| c.3913A>G | T1305A 2D  AIThe SynGAP1 missense variant T1305A is listed in ClinVar (ID 411587.0) with an “Uncertain” clinical significance and is present in the gnomAD database (variant ID 6‑33451787‑A‑G). All evaluated in‑silico predictors classify the change as benign: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized. No tool reports a pathogenic or likely pathogenic outcome. High‑accuracy assessments reinforce this benign prediction: AlphaMissense‑Optimized is benign, and the SGM Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” Foldetta results are unavailable. Overall, the computational evidence overwhelmingly supports a benign effect, and this conclusion does not contradict the ClinVar “Uncertain” status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Conflicting | 4 | 6-33451787-A-G | 30 | 1.86e-5 | -2.692 | Likely Benign | 0.055 | Likely Benign | Likely Benign | 0.069 | Likely Benign | 1.74 | Neutral | 0.000 | Benign | 0.001 | Benign | 3.24 | Benign | 1.00 | Tolerated | 3.77 | 5 | 1 | 0 | 2.5 | -30.03 | |||||||||||||||||||||||||||
| c.4021G>T | A1341S 2D  AIThe SynGAP1 missense variant A1341S is listed in ClinVar with an “Uncertain” status and is present in gnomAD (variant ID 6-33451895-G-T). All available in silico predictors agree on a benign effect: REVEL, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, FATHMM, AlphaMissense‑Default, and AlphaMissense‑Optimized all return benign scores, and the SGM‑Consensus (majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN) is “Likely Benign.” No tool predicts pathogenicity. High‑accuracy assessments corroborate this: AlphaMissense‑Optimized is benign, and the SGM‑Consensus is benign; Foldetta results are not available. Based on the collective predictions, the variant is most likely benign, and this conclusion does not contradict the ClinVar “Uncertain” classification. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | Uncertain | 1 | 6-33451895-G-T | -2.867 | Likely Benign | 0.078 | Likely Benign | Likely Benign | 0.099 | Likely Benign | 0.80 | Neutral | 0.000 | Benign | 0.001 | Benign | 4.40 | Benign | 1.00 | Tolerated | 3.77 | 5 | 1 | 1 | -2.6 | 16.00 | |||||||||||||||||||||||||||||
| c.603T>A | D201E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 D201E missense variant (ClinVar ID 3004688.0) is classified as **Benign** in ClinVar and is not reported in gnomAD. Prediction tools that agree on a benign effect include REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, AlphaMissense‑Optimized, and the SGM‑Consensus (majority vote). Only polyPhen‑2 HumDiv predicts a pathogenic outcome, while Rosetta, Foldetta, and AlphaMissense‑Default are uncertain. High‑accuracy assessments show AlphaMissense‑Optimized as **Benign**, the SGM‑Consensus as **Likely Benign**, and Foldetta as **Uncertain**. Taken together, the overwhelming majority of evidence points to a benign impact, and this conclusion aligns with the ClinVar designation. Thus, the variant is most likely benign, with no contradiction to its ClinVar status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | Benign | 1 | -2.640 | Likely Benign | 0.406 | Ambiguous | Likely Benign | 0.165 | Likely Benign | 0.42 | Likely Benign | 0.2 | 1.99 | Ambiguous | 1.21 | Ambiguous | 0.23 | Likely Benign | -0.69 | Neutral | 0.633 | Possibly Damaging | 0.108 | Benign | 4.30 | Benign | 1.00 | Tolerated | 3.46 | 9 | 3 | 2 | 0.0 | 14.03 | 258.7 | -24.8 | 0.9 | 0.1 | -0.3 | 0.2 | X | Uncertain | Asp201, an acidic residue located in the N-terminal loop before the first anti-parallel β sheet strand (res. Ile205-Pro208), is replaced by another acidic residue, glutamate. The carboxylate groups of both Asp201 and Glu201 side chains form hydrogen bonds with the hydroxyl group of Ser221 in the simulations. Due to its shorter side chain, Asp201 can also hydrogen bond with the backbone amide groups of neighboring loop residues Ser204 and Asp203. However, since the model ends abruptly at the N-terminus, no definite conclusions can be drawn from the simulations. | |||||||||||
| c.603T>G | D201E 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 D201E missense variant (gnomAD ID 6‑33435245‑T‑G) is listed in ClinVar with an uncertain significance. Across a broad panel of in silico predictors, the majority indicate a benign effect: REVEL, FoldX, premPS, PROVEAN, polyPhen‑2 HumVar, SIFT, ESM1b, FATHMM, and AlphaMissense‑Optimized all score benign. Only polyPhen‑2 HumDiv predicts pathogenicity, while Rosetta, Foldetta, and AlphaMissense‑Default remain inconclusive. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, resolves to “likely benign.” High‑accuracy assessments further support a benign outcome: AlphaMissense‑Optimized is benign, SGM‑Consensus is likely benign, and Foldetta is inconclusive. Taken together, the preponderance of evidence points to a benign impact, which does not conflict with the ClinVar uncertain status. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | PH | Conflicting | 2 | 6-33435245-T-G | 20 | 1.24e-5 | -2.640 | Likely Benign | 0.406 | Ambiguous | Likely Benign | 0.165 | Likely Benign | 0.42 | Likely Benign | 0.2 | 1.99 | Ambiguous | 1.21 | Ambiguous | 0.23 | Likely Benign | -0.69 | Neutral | 0.633 | Possibly Damaging | 0.108 | Benign | 4.30 | Benign | 1.00 | Tolerated | 3.46 | 9 | 3 | 2 | 0.0 | 14.03 | 258.7 | -24.8 | 0.9 | 0.1 | -0.3 | 0.2 | X | Uncertain | Asp201, an acidic residue located in the N-terminal loop before the first anti-parallel β sheet strand (res. Ile205-Pro208), is replaced by another acidic residue, glutamate. The carboxylate groups of both Asp201 and Glu201 side chains form hydrogen bonds with the hydroxyl group of Ser221 in the simulations. Due to its shorter side chain, Asp201 can also hydrogen bond with the backbone amide groups of neighboring loop residues Ser204 and Asp203. However, since the model ends abruptly at the N-terminus, no definite conclusions can be drawn from the simulations. | ||||||||
| c.819G>T | E273D 2D  3DClick to see structure in 3D Viewer AIThe SynGAP1 missense variant E273D is listed in ClinVar as Benign (ClinVar ID 1471608.0) and is present in gnomAD (variant ID 6‑33437724‑G‑T). Prediction tools that agree on a benign effect include REVEL, FoldX, Rosetta, Foldetta, PROVEAN, polyPhen‑2 (HumDiv and HumVar), SIFT, ESM1b, AlphaMissense‑Default, and AlphaMissense‑Optimized. Only FATHMM predicts a pathogenic outcome, while premPS is uncertain. The SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is Likely Benign. High‑accuracy assessments are consistent: AlphaMissense‑Optimized predicts benign, the SGM‑Consensus (majority vote) is benign, and Foldetta (combining FoldX‑MD and Rosetta outputs) also predicts benign. No prediction contradicts the ClinVar benign status; overall, the evidence strongly supports that E273D is most likely benign. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Benign | 1 | 6-33437724-G-T | 2 | 1.24e-6 | -1.811 | Likely Benign | 0.058 | Likely Benign | Likely Benign | 0.092 | Likely Benign | 0.26 | Likely Benign | 0.1 | -0.48 | Likely Benign | -0.11 | Likely Benign | -0.63 | Ambiguous | 1.99 | Neutral | 0.004 | Benign | 0.010 | Benign | 2.00 | Pathogenic | 1.00 | Tolerated | 3.38 | 18 | 3 | 2 | 0.0 | -14.03 | 223.1 | 22.1 | 0.2 | 0.0 | 0.0 | 0.1 | X | Potentially Benign | The negatively charged residue Glu273, located in a β hairpin loop (res. Glu273-Lys278) that connects two anti-parallel β sheet strands, is replaced with another negatively charged residue, aspartate. Because the C2 domain loop faces the membrane surface, the potentially crucial role of the carboxylate group of Glu273 or Asp273 on SynGAP-membrane association cannot be fully explored via solvent-only simulations.As a minor note, the neighboring residue Arg272, which stacks with the indole ring of the Trp362 side chain and directly faces RasGTPase, forms a salt bridge more often with Asp273 than with the non-mutated Glu273 in the simulations. Regardless, due to the similar physicochemical properties of the WT and variant residues at the membrane interface, the residue swap is likely to be well tolerated. | ||||||||
| c.865A>G | M289V 2D  3DClick to see structure in 3D Viewer AISynGAP1 missense variant M289V is reported in ClinVar as Benign (ClinVar ID 2122760.0) and is not present in gnomAD. Functional prediction tools largely agree on a benign effect: REVEL, Rosetta, Foldetta, premPS, PROVEAN, polyPhen‑2 HumDiv, polyPhen‑2 HumVar, SIFT, ESM1b, AlphaMissense‑Default, AlphaMissense‑Optimized, and the SGM‑Consensus all predict benign, while only FATHMM predicts pathogenic. High‑accuracy assessments reinforce the benign consensus: AlphaMissense‑Optimized scores benign; the SGM‑Consensus, derived from a majority vote of AlphaMissense‑Default, ESM1b, FATHMM, and PROVEAN, is benign; and Foldetta, which integrates FoldX‑MD and Rosetta stability outputs, also indicates benign. No prediction or stability result is inconclusive. Overall, the computational evidence strongly supports a benign classification, consistent with the ClinVar status and showing no contradiction. Disclaimer: This summary was generated using AI and should be interpreted alongside expert review. | Likely Benign | C2 | Benign | 1 | -4.239 | Likely Benign | 0.117 | Likely Benign | Likely Benign | 0.150 | Likely Benign | 1.09 | Ambiguous | 0.1 | -0.27 | Likely Benign | 0.41 | Likely Benign | 0.24 | Likely Benign | -0.36 | Neutral | 0.136 | Benign | 0.054 | Benign | 1.80 | Pathogenic | 1.00 | Tolerated | 3.38 | 23 | 2 | 1 | 2.3 | -32.06 | 204.2 | 51.0 | 0.0 | 0.0 | 0.2 | 0.0 | X | Potentially Benign | The hydrophobic residue Met289, located in a β hairpin linking two anti-parallel β sheet strands (res. Met289-Arg299, res. Arg272-Leu286), is swapped for another hydrophobic residue, valine. In the variant simulations, the branched hydrocarbon side chain of Val289 packs against the phenol group of the Tyr291 side chain but is unable to form methionine-aromatic interactions. β hairpins are potential nucleation sites during the initial stages of protein folding, so even minor changes in them could be significant. However, based on the simulations, the residue swap does not cause adverse effects on the structure. | |||||||||||
Found 757 rows. Show 200 rows per page. Page 4/4 « Previous |